LPA Quality Assurance: Proficiency Testing
- Read more about LPA Quality Assurance: Proficiency Testing
- Log in to post comments
Steps Involved in Proficiency Testing (PT):
Steps Involved in Proficiency Testing (PT):
A batch of samples is defined as a group of samples that are processed, amplified and hybridized:
QC during the DNA extraction procedure
The quality concepts of the Line Probe Assay (LPA) laboratory include:
Issue #1:
Absence of Conjugate Control (CC) and Amplification Control (AC) in a negative control (Figure 1).

Figure 1: Absence of CC and AC in a Negative Control
Table 1 below lists the reasons and troubleshooting for the absence of CC and AC in a negative control.
There could be overall weak or no signals on the strip, including or except, the Conjugate Control (CC) zone.

Figure: LPA showing No Signal Except CC
Reasons and Troubleshooting for No Signal Except CC are given in the table below:
|
REASON FOR ERROR |
SOLUTION |
|---|
The limitations of Second Line Probe Assay (SL-LPA) are:
he examples of banding patterns for Second Line - Line Probe Assay (SL - LPA) are shown in the figure below.
Description
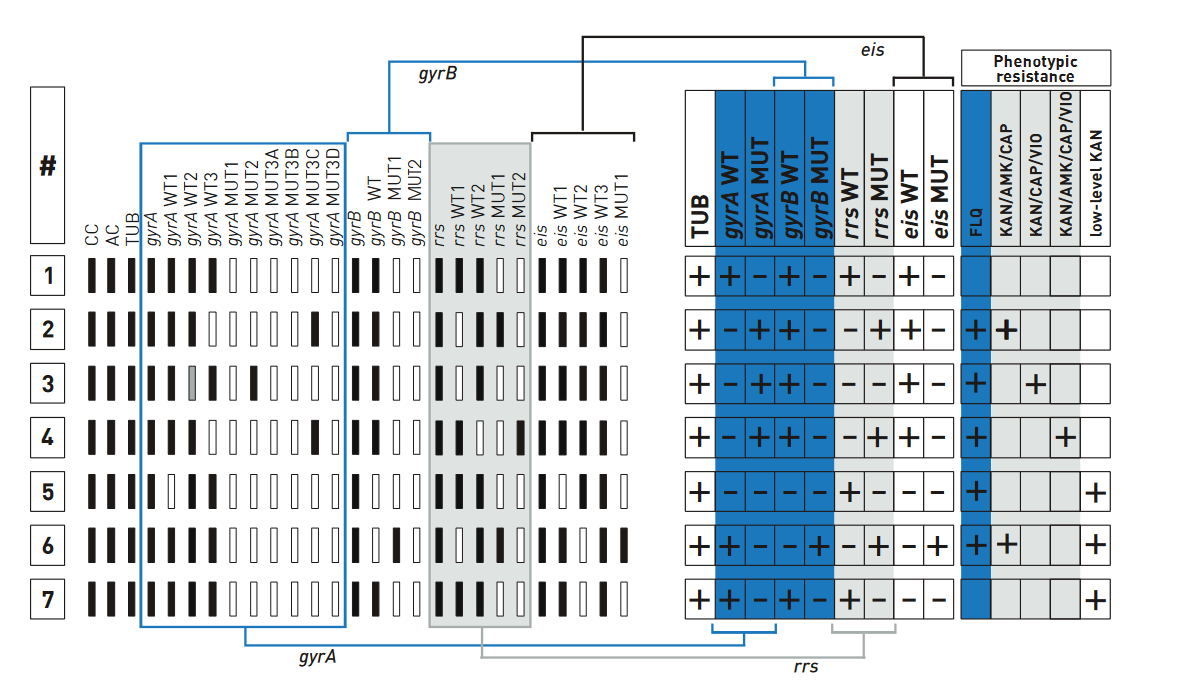
Notes
Special Scenarios
Zone developed/ not developed (failing) in Wild Type (WT) and Mutation (MUT) probes are used to predict drugs resistance in First Line - Line Probe Assay (FL-LPA).
For Rifampicin
Table 1 shows mutations in rpoB gene and corresponding WT, MUT and responsible codons and mutations.
Table 1: Mutations in the rpoB gene and corresponding wild type and mutation band; Source: GenoType MTBDR plus ver 2.0 kit, Instructions for Use.
Reporting Drug-resistance in Special Cases