CBNAAT Cartridge Loading
- Read more about CBNAAT Cartridge Loading
- Log in to post comments
Specimen Processing for CBNAAT
Persons with cough of more than 2 weeks, with or without other symptoms suggestive of TB, should be promptly identified as presumptive pulmonary TB patients.
Under NTEP, they are to be referred to the designated microscopy centre (DMC) for sputum examination using the Request form for examination of biological specimen.
Patients belonging to the key population EPTB, HIV and Paediatrics groups (after X-ray screening in case of children) can be directly referred for NAAT.
Autoclaves are key biosafety equipment in tuberculosis (TB) laboratories.
It allows decontamination of used instruments and articles and makes it safe for reuse (or discarding).
Autoclaves kill all pathogens on the articles which will be added to it for sterilization. Hence the articles autoclaved will be sterile (no biohazard pathogens on them).
Principle of Procedure
Principle of procedure
Staff working with patient specimens and live mycobacterial cultures must operate under appropriate biosafety conditions with adequate infection control measures in place, including staff health checks.
For the safety of personnel working in TB labs, the following factors must be considered to avoid the risk associated with pathogenicity and transmission of TB bacilli.
The 4 key factors that must be considered during risk assessment of TB labs include:
Risk assessment is the process that enables the appropriate selection of microbiological practices, safety equipment, and facility safeguards that can prevent laboratory-associated infections (LAIs).
The figure below shows a flowchart that presents the risk assessment process for a biological hazard.
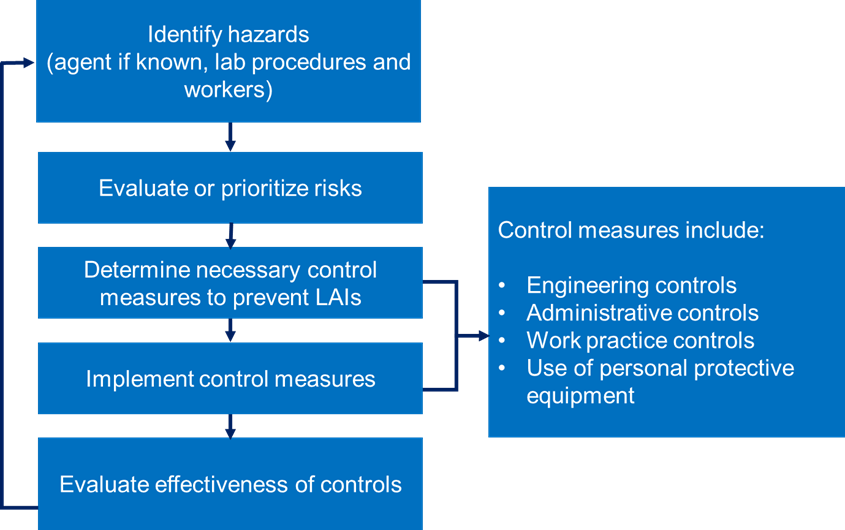
Figure: Risk assessment process for a biological hazard
Risk assessments for TB laboratories should consider: