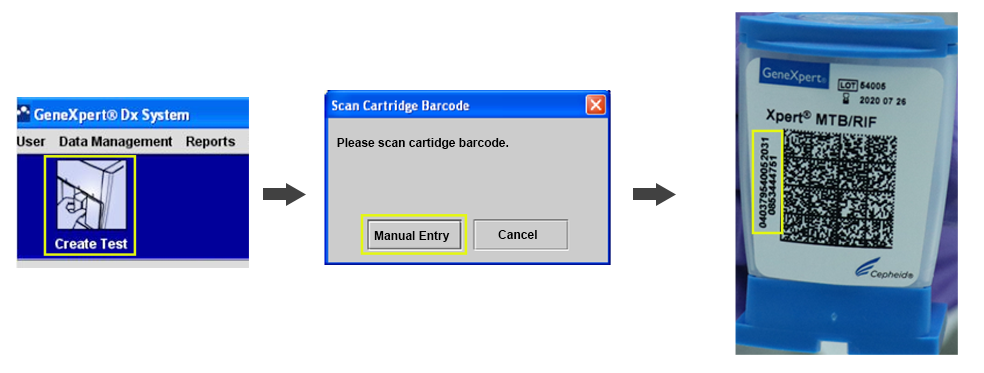
CBNAAT Hardware Problem: Modules Not Detected
- Read more about CBNAAT Hardware Problem: Modules Not Detected
- Log in to post comments
One of the common problems encountered while using the CBNAAT machine is the non-detection of the module.
Usually, there is a loss of communication between modules and software, leading to non-detection.
Origins:
- Ethernet connection between Personal Computer (PC) and CBNAAT is bad
- Power supply issue (main power or Universal Power Supply (UPS) fluctuations)
- Bad connection points between gateway board and modules
- Too high room temperature
Solutions: