-
CDST_LT: Introduction to biosafety
FullscreenBiosafety in TB Laboratories
ContentWhat is Biosafety?
Biosafety is safe handling and containment of infectious microorganisms and hazardous biological materials.
Why Biosafety?
Infections which are obtained through laboratory or laboratory-related activities are known as Laboratory Acquired Infections (LAI). These infections are major occupational health hazards and are a cause of concern for the safety of the staff working in laboratories.
Routes of entry of LAI
There are multiple routes of entry and transfer of toxic material through the body and its transfer to various organs and systems.
The most predominant routes of LAI are:
- Nose - Inhalation of infectious aerosols
- Mouth - Ingestion or exposure through mouth pipetting or touching mouth or eyes with contaminated fingers or contaminated object
- Skin and mucous membrane:
- Spills and splashes onto skin and mucous membranes
- Parenteral inoculations with syringes or other contaminated sharps
- Animal bites and scratches from research laboratories or activities
Biosafety guidelines are prepared to promote:
- Safe microbiological practices
- Safety equipment and facility safeguards for reducing LAIs
- To protect public health and the environment
Biosafety in Tuberculosis TB laboratory (lab)
In a TB lab, the primary risk for infection transmission is the aerosol contaminated with TB bacilli when:
- TB patients walk into the laboratory for specimen collection or other testing and may be a source of droplet infection if they cough during laboratory visits
- Procedures such as centrifugation, vortexing, and vigorous shaking generate significant aerosols that are biohazardous
Key points to consider for M. TB as a biological hazard are given in the table below.
MYCOBACTERIUM TUBERCULOSIS (M. TB) AS BIOLOGICAL HAZARD
RISK FACTORS
Pathogenicity
M.TB exposure may lead to infection
5-10% of infected persons will develop TB disease
Primary route of transmission
Inhalation
Stability
Tubercle bacilli can remain viable for extended periods in the environment
Infectious dose
As little as 10 bacilli can infect humans
Effective Vaccine (for adults)
No vaccine is available for adults
Effective treatment for strain susceptibility to different medicines
Yes
Effective treatment for MUltiple Drug Resistant (MDR), Extensively Drug Resistant (XDR) strains
Yes, but more difficult to treat than susceptible strains
Standards for TB Lab Biosafety
- Standards are prepared based on the recommendations from the WHO expert group in 2012.
- Standards use a procedural approach for the assessment of risk.
- It establishes minimum requirements necessary to ensure biosafety during TB microscopy, culture, drug-susceptibility testing (DST) and molecular testing in different countries and epidemiological settings.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Risk Assessment Process
ContentRisk assessment is the process that enables the appropriate selection of microbiological practices, safety equipment, and facility safeguards that can prevent laboratory-associated infections (LAIs).
The figure below shows a flowchart that presents the risk assessment process for a biological hazard.
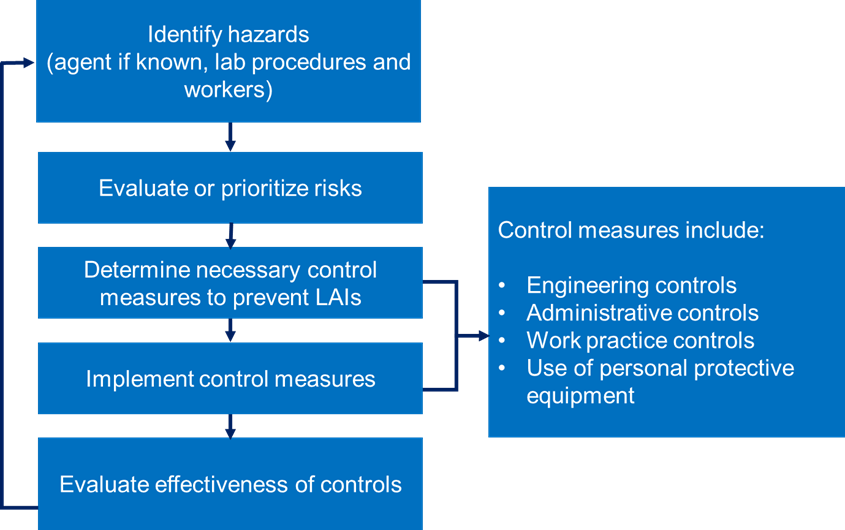
Figure: Risk assessment process for a biological hazard
Risk assessments for TB laboratories should consider:
- Bacterial load of materials (such as specimens and cultures)
- Viability of the bacilli
- Whether the material handled is prone to generate infectious aerosols during different procedures
- Laboratory’s workload and procedures; it is important that laboratories working with drug resistant strains should establish higher level precautions and biosafety measures
- Epidemiology of the disease and the patient population served by the laboratory
- Health/immune status of laboratory workers
- Experience and competency of laboratory staff in using good laboratory practices while handling infectious agents
- Equipment biosafety features required to prevent spill/breakage/aerosol generation when handling potentially infectious material
- Maintenance of equipment
- Biomedical waste management
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Risk Monitoring
ContentRisk monitoring is a regular inspection of the laboratory environment to monitor risks and control measures.
Stages of Risk Monitoring
1. Thorough investigation of lab incidents/lab accidents to understand the reason for such accidents
2. Review of corrective actions taken after the lab incident/lab accident
3. Implementation of preventive measures post-incident to prevent the future occurrence of such events
4. Documenting the process of risk monitoring
Importance of Risk Monitoring
Risk monitoring is important to ensure that selected and implemented biosafety measures are constantly improved.
Development of a new procedural risk assessment/ review of an existing one is required in the following situations:
- If there is an initiation of new work or there is a change in the work environment
- If there are some alterations to the workflow or volume of work done
- Construction of a new lab or modification in the lab structure
- Introduction of new equipment into the lab
- Alterations in staffing arrangements
- Alterations in standard operating procedures or working practices
- Major incidents in the laboratory like a major spill
- Evidence of, or suspicion of, any staff acquiring a Laboratory Acquired Infection
- Emergency responses and contingency planning requirements
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety in TB Laboratory: Factors to be Considered in TB Laboratory Risk Assessment
ContentStaff working with patient specimens and live mycobacterial cultures must operate under appropriate biosafety conditions with adequate infection control measures in place, including staff health checks.
For the safety of personnel working in TB labs, the following factors must be considered to avoid the risk associated with pathogenicity and transmission of TB bacilli.
The 4 key factors that must be considered during risk assessment of TB labs include:
1. The organism i.e., M. tuberculosis: Different strains of TB carry different levels of individual and community hazards. The following should be considered:
- Route of transmission (Inhalation of aerosols)
- TB epidemiology and patient population served by the laboratory (drug-sensitive TB, drug-resistant TB, non-TB patient proportions): MDR and XDR strains carry higher risks and cause greater harm to the infected individual as treatments may be limited or less effective.
2. Specimen Handling: Procedures involving generation of aerosols are more hazardous. The following should be considered:
- Bacterial load in the environment
- Viability of bacilli in sputum specimens and cultures
- Manipulations (centrifugation, vortexing, and vigorous shaking) that are likely to generate infectious aerosols
3. Staff and Workload: Individuals in the laboratory differ in their susceptibility to TB, higher-risk individuals may be less experienced, over-worked or have an underlying health condition. The following should be considered:
Level of experience and competency of the laboratory staff:
- Training status of staff to perform laboratory procedures
- Staff’s competency procedures in handling potentially infectious material and response to emergencies
- Ability of the staff to use biosafety equipment properly
- Review of the technical proficiency of staff
- Staff proficiency in using aseptic techniques and Biological Safety Cabinets (BSCs)
- Staff willingness to accept responsibility for protecting themselves and co-workers
Health status of staff: Higher-risk groups include:
- Individuals with reduced immunity which may be caused by certain medications
- HIV-infection or pregnancy may increase the risk of becoming infected with TB
- Diabetic individuals
4. Location of the Laboratory: TB Laboratories are generally located away from the main health facilities in separate building with unrestricted entrance to the building. If the laboratory cannot be located in a separate, dedicated building, separation may be achieved by placing the laboratory at the blind end of a corridor. TB containment laboratories should have access only to authorized staff through an anteroom.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Biosafety measures for TB Laboratories
FullscreenTB Laboratory Design and Facilities as a Biosafety Measure
ContentThe basic recommended design features of TB laboratories to ensure appropriate biosafety are:
- Adequate ventilation and directional airflow; 6-12 air exchanges per hour (ACH) is adequate ventilation for TB laboratories.
- Ample space for the safe conduct of laboratory work and for cleaning and maintenance (Figure 1).
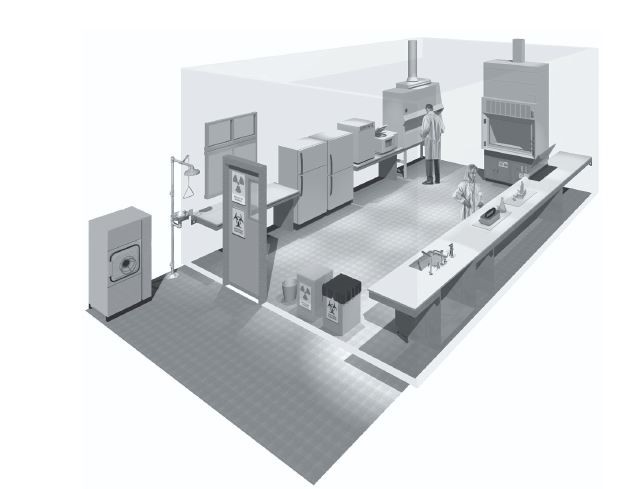
Figure 1: Ample and open spacing in an ideal TB lab

- Walls, ceilings and floors should be smooth and easy to clean.
- Floors should be slip-resistant.
- Benchtops should be resistant to the chemicals and disinfectants normally used in the laboratory.
- Proper and adequate illumination: Undesirable reflections and glare should be avoided; curtains must not be used.
- Open spaces between and under benches, cabinets and equipment to allow access for cleaning.
- Storage spaces should have the following features:
- Adequate to hold supplies for immediate use and prevent clutter on bench tops and in corridors outside the laboratory.
- Provision of additional space for long-term storage conveniently located outside work areas.
- Well-defined and specific area for safe preparation, handling and storage of acids, stains and solvents.
- Facilities for eating, drinking and resting should be provided outside work areas.
- Provisions for adequate handwashing, which includes:
- Sink and soap in every room of the laboratory, preferably located near the exit.
- Automated or hands-free taps are recommended.
- The dispenser for paper towels should be near the sink.
- Laboratory doors should have vision panels and appropriate fire ratings; they should be self-closing (Figure 2).
- There should be a reliable and adequate electricity supply.

Figure 2: Lab doors with fire rating shown

Laboratory furniture should be sturdy and made of impervious materials. It is important that the furniture can be decontaminated easily. Therefore, cloth-covered furniture should never be used in the laboratory (Figure 3)

Figure 3: Furniture, like chairs, must not be covered with cloth
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Ventilation in TB Laboratories as a Biosafety Measure
ContentVentilation is an essential biosafety design feature in a TB lab.
Laboratory ventilation has three basic elements:
1. Ventilation rate
Amount of outdoor air that flows into the laboratory.
2. Airflow direction
Overall direction of air flowing through the laboratory should be from functionally clean areas to dirty areas.
3. Airflow pattern
External air should be delivered to each area of the laboratory and the internal air be removed efficiently.
Adequate ventilation is the most important biosafety feature of a TB laboratory. The figure below shows the main recommendations when considering ventilation in TB labs.
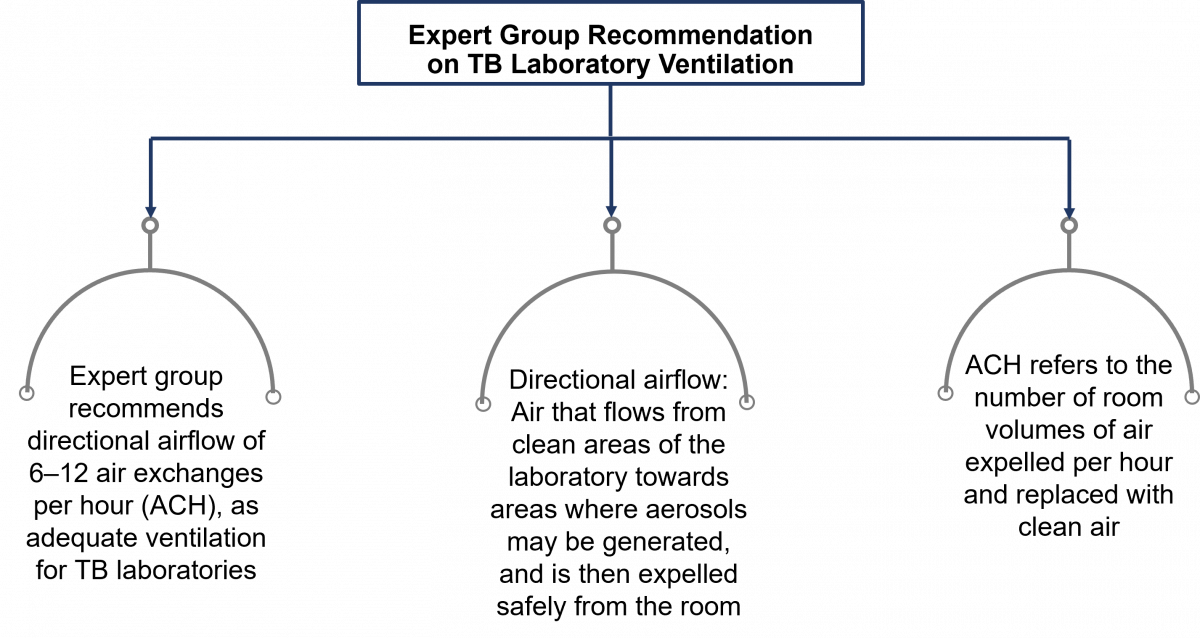
Figure: Expert Group Recommendations for TB Lab Ventilation

Ventilation in TB Containment Laboratories
- A ducted air ventilation system is required.
- This system must provide a sustained negative pressure, which maintains directional airflow by drawing air into the laboratory from “clean” areas toward “potentially contaminated” areas.
- The laboratory should be designed such that, under failure conditions, the airflow will not be reversed.
- Laboratory personnel must be able to verify directional airflow. A visual monitoring device, which confirms directional airflow, must be provided at the laboratory entry.
- Audible alarms should be considered to notify personnel of airflow disruption.
- The laboratory exhaust air must not re-circulate to any other area of the building.
- The laboratory building exhaust air should be dispersed away from occupied areas and from building air intake locations, or the exhaust air must be HEPA filtered.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Biosafety Measures for TB Laboratory Equipment
ContentAs part of the biosafety measures in TB labs, there are specific standards that need to be followed for lab equipment.
Key Features of Laboratory Equipment in TB Laboratories
The equipment should be:
- Designed to prevent or limit contact between the operator and infectious material.
- Constructed from materials impermeable to liquids and resistant to corrosion.
- Fabricated to be smooth, and without sharp edges and unguarded moving parts.
- Designed, constructed and installed to:
- Facilitate simple operations.
- Allow easy maintenance, cleaning, decontamination and certification testing.
- Not consisting of glassware and other breakable materials, whenever possible.
- Shock-proof and of standard materials, such as in case of electrical wires, plugs and other accessories.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: TB laboratory classification
FullscreenClassification of TB Laboratories
ContentBased on 2012 WHO expert group recommendations, TB labs can be classified into three types based on their levels of procedural risk:
- Low TB risk
- Moderate TB risk
- High TB risk (TB Containment Laboratory)
- Recommendations based on minimum requirements needed to limit or reduce risks of infection in laboratories for carrying out specific procedures.
- Additional measures may be deemed necessary following a site-specific risk assessment.
Based on the operational and diagnostic methods adopted, the laboratories can be classified as:
- The Designated Microscopy Centers (for microscopy only)
- The Nucleic Acid Amplification Testing (NAAT) Laboratories or the Molecular Test Laboratories
- The Culture and Drug Sesitivity Testing (C&DST) Laboratories or the Phenotypic Laboratories
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Low-risk TB Laboratories
ContentDepending on the procedural risk in the TB lab, the lab is classified into low, moderate and high-risk categories.
Low-risk TB laboratories work on sputum specimens for:
- Direct Acid-fast Bacilli (AFB) smear microscopy
- Cartridge-based Nucleic Acid Amplification Test (CBNAAT)/ Truenat


Figure 1: Procedures Conducted in a Low-Risk TB Lab
- Procedures that involve direct microscopy or killing of the mycobacteria before microscopy are taken up in these labs.
- These labs have a low risk of generating infectious aerosols from specimens and a low concentration of infectious particles.
Features and Essential Minimum Biosafety Measures of Low-risk TB Labs
1. Space requirements
- Bench spaces should be separated from areas used to receive specimens and administrative areas used for paperwork and telephones.
- Ensure adequate ventilation; it is important to maintain natural or mechanical, directional airflow with 6-12 air changes per hour (ACH). Workstations are optional solutions.
2. Procedural requirements
To minimize the generation of aerosols:
- Carefully open specimen containers.
- Air-dry smears and use a flame to fix them.
- Use disposable wooden sticks or transfer loops.
For the handling of leaking specimen containers: Discard and request a fresh sample.
3. Personal Protective Equipment (PPE) requirements
- Protective laboratory coats should be worn at all times in the laboratory.
- Gloves must be worn for all procedures that involve direct contact with potentially infectious materials.
- Gloves should not be reused.
- Staff should always wash their hands before leaving the laboratory.
- Respirators are optional.


Figure 2: Challenges in a Low-risk TB Lab that Increase the Risk of Infection
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Moderate-risk TB Laboratories
ContentModerate-risk TB Laboratories can:
- Process specimens for inoculation on primary solid-culture media
- Perform direct line-probe assays and direct Drug Sensitivity Testing (DST)
Essential Minimum Biosafety Measures
- Biological Safety Cabinets (BSCs): Class I or II
- Ventilation: 6-12 ACH
- Personal Protective Equipment: Laboratory gowns and gloves
- Laboratory design: Restricted traffic area
- Decontamination and waste disposal
- Procedures optimized for minimizing the generation of aerosols
Challenges of Moderate-risk TB Laboratories
- Staff may work in areas with poor ventilation and/ or poor illumination.
- BSCs may be poorly maintained and not certified or not be properly ducted.
- Careless manipulation of specimens may lead to aerosolization.
- Specimen containers may break or leak during centrifuging.
- Adequate warnings of biohazards may be lacking, and information on who should be contacted during an emergency may be inadequate.
- Cooling or heating systems may not work properly.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Personal Protective Equipment (PPE)
FullscreenPersonal Protective Equipment [PPE] Use in TB Lab Settings
ContentWhat is Personal Protective Equipment (PPE)?PPE refers to physical barriers which are used alone or in combination to protect an individual's mucous membranes, airways, skin and clothing from contact with infectious agents, hazardous material, conditions or processes.
Components of PPE
- Protective clothing: Surgical gowns and laboratory (lab) coats
- Respiratory devices: N95 respirators
- Hand protection: Gloves
- Head protection: Headgear/ hair covers/ caps
- Foot protection: Shoe cover, safety shoes/ closed-toe shoes
- Eye protection: Safety glasses/ goggles, shields
PPE Hazard Assessment
- Evaluation of the PPE requirements for a specific activity or work environment is carried out by the lab director.
- For TB labs it is based on the risk assessment. PPE requirements will thus differ according to whether activities are classified as low risk, medium risk or high risk.
PPE Usage in Different Sections of the TB Laboratory
The table below lists the different PPE requirements for different sections of the TB lab.
Table: PPE usage in different sections of TB labs Section/ RoomPPE used/ preferredSample collection/ reception roomLab coat, gloves Smear microscopy section/ room/ Designated Microscopy Centre (DMC)Lab coat, glovesCartridge-based Nucleic Acid Amplification Test (CBNAAT) sample processing sectionLab coat, gloves, Surgical maskSample opening area(in a higher laboratory) Surgical gown, gloves and N95 respirator Media preparation room Designated lab coat, gloves, hair cover, shoe covers/ closed-toe shoes Culture reading roomLab coat, gloves, N95 respiratorLine Probe Assay (LPA) clean roomsDesignated lab coat, gloves, hair cover, shoe covers/ closed-toe shoesWalk-in cold roomLab coat, insulating gloves, shoe covers/ closed-toe shoesWalk-in incubatorLab coat, respirator and gloves, shoe covers/ closed-toe shoesCorridor inside the labLab coat, shoe cover/ closed-toe shoesDeep freezersLab coat, cryo glovesDisinfection, washing and sterilisation roomLab coat, gloves (heavy duty), insulating gloves, closed-toe shoesResources
- National Guidelines for Infection Prevention and Control in Healthcare Facilities, 2020, MoHFW, GoI.
Resources
Use of a PPE in a DMC
ContentThe use of appropriate Personal Protective Equipment (PPE) in a TB laboratory is determined by risk assessment (according to the procedure and suspected pathogen).
Designated Microscopy Centres (DMCs) are low-risk TB laboratories, hence PPE should be used as follows:
- Laboratory coats should be worn at all times by the Laboratory Technician (LT) while working in the laboratory.
- Gloves and long-sleeved laboratory coats should be used during sample collection, opening sample transport boxes, handling leaky specimen containers, smear preparation and staining smears.
- Masks are not required for use during the preparation of sputum smears in well-ventilated areas.
- LTs and support staff handling biological waste should wear gloves.
- Patients that visit DMC to provide sputum samples should be advised to wear masks.
It is advised that the laboratory should maintain adequate ventilation by keeping the windows open all the time or installing exhaust for the personal protection of the laboratory staff.
Resources
- Guidelines on Airborne Infection Control in Healthcare and Other Settings, MoHFW, 2010.
- Infection Prevention and Control of Epidemic- and Pandemic-Prone Acute Respiratory Infections in Health Care, WHO, 2014.
- Training Modules (1-4) for Programme Managers and Medical Officers, NTEP, 2020.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
LTs and support staff handling biological waste should not wear gloves.
True
False
2
LTs and support staff handling biological waste should wear gloves.
Yes Yes Laboratory Gowns for Use by Lab Personnel
ContentLaboratory Gowns (Figure 1)
- Protect the skin and clothing from large droplets of infectious or hazardous materials that may be splattered in the laboratory.
- Gowns must be worn for specimen processing, tuberculosis (TB) culture and drug sensitivity testing (DST).
- The gown must have a solid front and close and tie in the back.
- The gown must have long-sleeves and cuffs.
- For maximum protection, gloves must be pulled over the cuff.
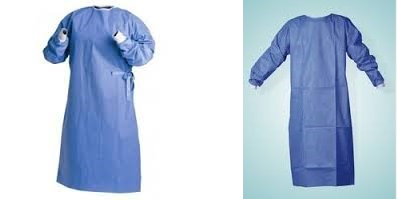
Figure 1: Laboratory Gowns
Laboratory Coats (Apron) (Figure 2)
- Lab coats must be worn when working on specimens and acid-fast bacilli (AFB) smears and not during TB culture work.
- Lab coats must be made from washable and autoclavable cloth or disposable materials.
- It must be closed up in the front (with snaps, buttons or velcro) and must have long sleeves.
- It must be buttoned till the top at all times in the lab.

Figure 2: Laboratory Coat
Do’s and Don’ts for Laboratory Gowns and Coats
- The laboratory gown must be laundered once a week and immediately after being overtly contaminated.
- If staff is using a reusable gown, it must be laundered and sterilized as required before reuse.
- Used gowns must be stored in separate cupboards or lockers.
- Hang the gown on designated hooks in the lab.
- Lab gowns must not be worn outside the laboratory, e.g., in communal areas, toilets, offices, etc.
- Staff must wear appropriately sized gowns and coats.
- Lab coats must not be taken home for washing. Laboratory personnel in-charge must arrange for cleaning from the worksite only. Before laundering, the gowns and lab coats must be disinfected or autoclaved.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Donning and Doffing of PPE
ContentSequence of Donning of Personal Protective Equipment (PPE): (How to put on PPE)
- Footwear
- Gown
- N-95 respirator
- Goggles and face shield
- Gloves
Donning must be done before entering the patient/ laboratory area.
Sequence of Doffing of PPE: (How to safely remove PPE)
- Gown and gloves
- Footwear
- Goggles and face shield
- N-95 respirator
- Wash hands or use an alcohol-based hand sanitizer immediately after removing all PPE.
Where to remove PPE?
- PPE must be removed at doorway, before leaving the patient room or in the anteroom.
- Staff must remove respirator outside the room, after the door has been closed.
Four Key Points to Remember about PPE Use
- Staff must don the PPE before any contact with the patient, generally before entering the room.
- After wearing the PPE on, staff must remain careful to prevent spreading contamination.
- After completing the tasks, staff must remove the PPE carefully and discard it in the receptacles provided.
- After removing the PPE, immediately hand hygiene must be performed before going on to the next patient.
Click the video below to see the donning and doffing of PPE.
Resources
- Manual for Donning and Doffing of PPE by Center for Disease Control and Prevention (CDC).
- Steps to put on Personal Protective Equipment (PPE), WHO Poster.
Kindly provide your valuable feedback on the page to the link provided HERE
N95 Respirators for TB Lab Personnel
ContentN95 respirators are Personal Protective Equipment (PPE) used as a biosafety measure in clinical and laboratory settings.
N95 Respirators
- N95 filtering facepiece respirators are air-purifying respirators.
- Certified to have a filter efficiency level of 95% or greater against particulate aerosols free of oil and 0.3 microns in size.
- N95 (United States Standard NIOSH N95) or program-specific guidelines should be followed in the selection of these respirators.

Figure: N95 Respirators
Use of N95 Respirators in TB Laboratories
- Not required in low- and moderate-risk TB laboratories but are required in high-risk TB laboratories
- Must be available in laboratories where culture manipulations are performed
- Should be included as part of a laboratory’s spill clean-up kit
- Should never be used as a substitute for a properly maintained and functioning Biosafety Cabinet (BSC) and other PPE
Approval Required for N95 Use
Personnel who are required to wear respirators, shall be approved for N95 use after completing the following:
- Medical Evaluation/ Clearance: To determine if users are physically fit to wear a respirator.
- Training: To ensure users are familiar with N95 respirators, their proper use and protective limitations.
- Fit-testing: To determine which respirator model/ size provides the proper fit for the user. Such a fit-test is required on an annual basis and when a different model is being used or there are changes in facial characteristics.
Table: Showing the difference between user seal check and fit-test
USER SEAL CHECK
FIT-TEST
Should be conducted each time a respirator is put on, to determine if it is properly sealed to the face.
- Evaluation of how a respirator fits, conducted by trained personnel. (Includes the use of a scented solution and the determination of whether the employee can detect the odour/ taste).
- Should be conducted prior to the use of a respirator and annually thereafter OR when a new respirator is being introduced.
Limitations of N95 Respirators
- N95 respirators ONLY filter out particulate contaminants. N95 respirators do not protect from chemical vapours/ gases, high-risk exposures such as those created by aerosol-generating procedures (i.e., bronchoscopy, autopsy) and asbestos handling.
- N95 respirators are generally single-use disposable. Considering the funding constraints for TB labs, the availability of N95 can pose a challenge.
Comparison between the Surgical Mask and N95 Respirators
- The purpose of a surgical mask is to protect the environment or the community from the wearer of the mask. It is usually worn by a patient infected with TB or other infectious diseases to protect others.
- Unlike the N-95 respirator, a surgical mask offers less protection to the wearer from aerosolized TB and is unable to form a tight seal; thus aerosols are able to come in through the gaps.
Disposal of N95 Respirators
- TB lab personnel must remove the mask using the appropriate technique (i.e., must not touch the front but remove the lace from behind), only after coming out of the laboratory.
- Staff must clean their hands by using an alcohol-based hand rub or soap and water, after removal or whenever inadvertently touching a used mask.
- N-95 mask must be discarded and collected in separate 'yellow colour-coded plastic bags' (suitable for biomedical waste collection).
Resources
- National Guidelines for Infection Prevention and Control in Healthcare Facilities, 2020. Ministry of Health and Family Welfare, Government of India.
- Importance of a User Seal Check, Manual from 3M Science.
- Frequently Asked Questions about Respiratory Protection by Center for Disease Control and Prevention (CDC).
Kindly provide your valuable feedback on the page to the link provided HERE
Gloves for TB Lab Personnel
Content- Gloves are made for protecting the hands of health professionals from contaminated and infectious surfaces.
- Lab staff must wear gloves for all the procedures that involve direct or accidental contact with infectious material.
- Gloves are made for single-use (disposable gloves) and should not be re-used (see Figure 1)
- Gloves are made from vinyl, latex or nitrile.
- For the staff who are allergic to latex and when performing PCR, nitrile gloves should be available for use.
Figure 1: Disposable Gloves
Do’s when using gloves
- Staff must select and use the right kind of glove for the job they are going to perform.
- Always use the correct size of gloves. Staff regularly need to check the fitness and size suitable for them.
- Gloves must be resistant to the chemical used during the procedure.
- Staff must inspect their gloves before using them.
- Before wearing gloves, staff must remove any rings, watches, or bracelets that might cut or tear the gloves.
- If gloves are torn during a procedure, replace them immediately (Figure 2).
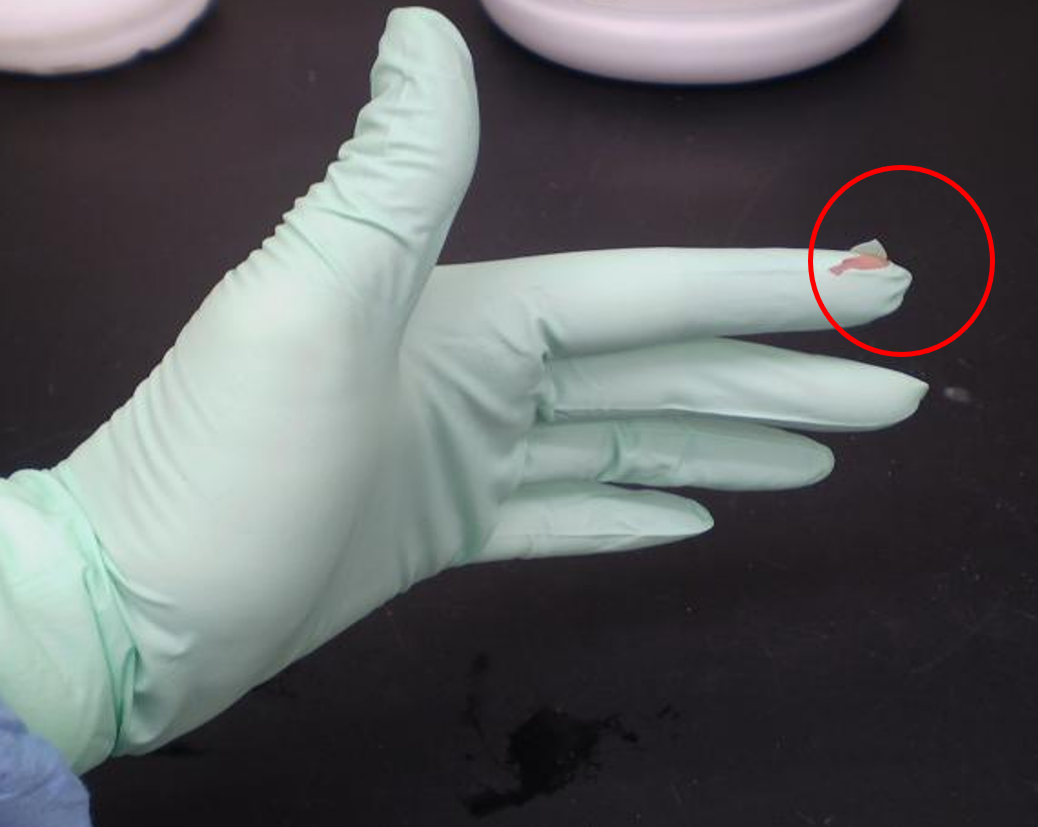
Figure 2: Torn gloves need immediate replacement
- Staff must wear double gloves when working with potentially hazardous materials in the biosafety cabinet.
- Staff must pull the gloves over the cuff of the lab gown for maximum protection.
- Staff must change the gloves:
- At the end of the task
- If gloves are soiled/punctured
- Soiled gloves must be discarded in the bag provided for waste disposal.
- Gloves guard against infection through cuts and abrasions on the hands. Cuts and abrasions must be covered with a sticking plaster and/or suitable barrier dressing at all times while in the laboratory
- Cryo gloves should be used while handling deep freezers and cotton gloves should be use while handling the autoclave
Don’ts when using gloves
- Contaminated gloves must not be used to handle or to operate equipment (e.g., microscope or telephone), that would otherwise never become contaminated.
- Staff must never wear gloves outside the laboratory.
- After wearing gloves, staff must avoid/limit touching any surface
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Emergency preparedness
FullscreenEmergency Preparedness Plan in TB Lab Settings
ContentThe emergency preparedness plan in a tuberculosis (TB) laboratory is a written document that should provide:
- Operational procedures for incidents and accidents while storing or dealing with tubercule bacteria isolates
- Precautions against natural disasters, for example, fire, flood, earthquakes and explosions

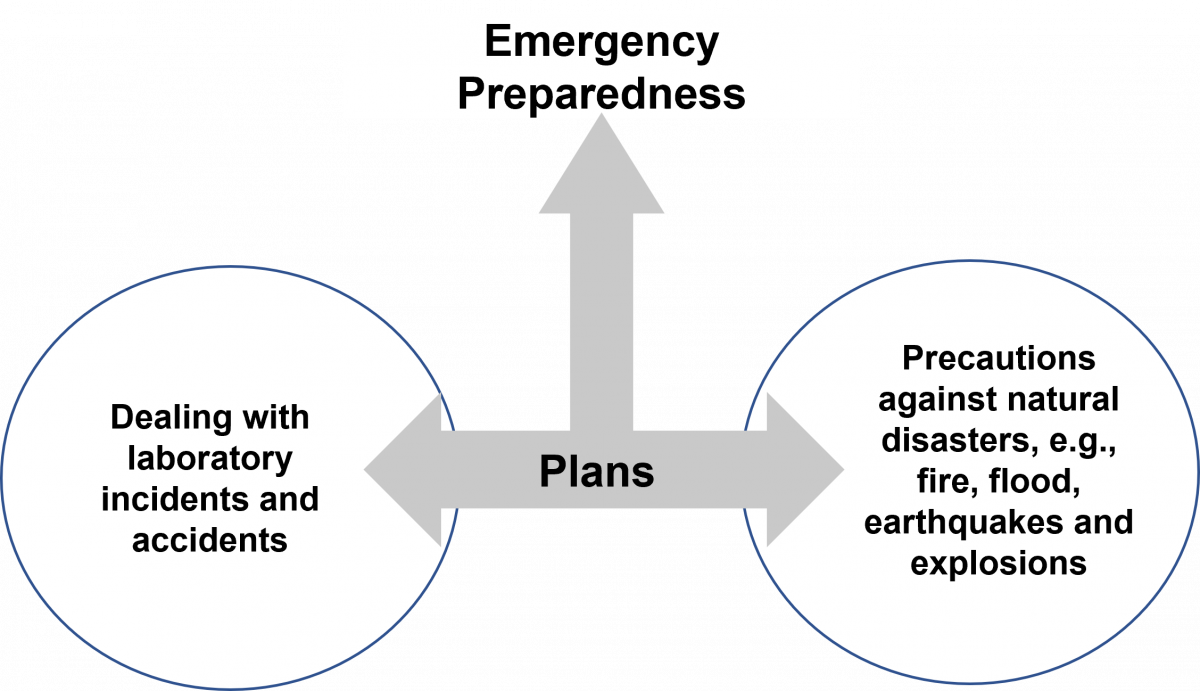
Figure: Composition of an Emergency Preparedness Plan
These protocols should follow standard protocols that are put together by organizations such as the World Health Organisation (WHO) and should be tailored to the needs of the laboratory.
The emergency preparedness plan should be prepared by the senior microbiologist/ laboratory technician who heads the laboratory, after due proceedings from the institutional infection control committee.
The plan can be revised from time to time based on the experiences during the course and should contain, who does what, the standard operating procedures for handling the emergency, checklists for operations and logistics, and emergency contact numbers.
The plan should provide operational procedures for the following scenarios:
- Responses to natural disasters such as fires, floods, earthquakes or explosions
- Risk assessments associated with any new or revised procedures
- Managing exposures and decontamination
- Emergency evacuation of people from the premises
- Emergency medical treatment of exposed and injured persons
- Medical surveillance and clinical management of persons exposed to an incident
- Epidemiological investigation and continuing operations after an incident
Considerations for the Emergency Plan
- Location of high-risk areas, such as laboratories and storage areas
- Identification of at-risk personnel and populations
- Identification of emergency procedures according to the level of risk
- Identification of responsible personnel and their duties, such as the biosafety officer, safety personnel, local health authority, clinicians, microbiologists, veterinarians, epidemiologists, fire services and police services
- Treatment and follow-up facilities that can receive exposed or infected persons
- Transport for exposed or infected persons
- How emergency equipment will be provided, such as protective clothing, disinfectants, chemical and biological spill kits, decontamination equipment and supplies
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
TB Laboratory Accidents Leading to Infectious Spillage
ContentThere are different types of accidents that can occur in the TB lab. These accidents and actions to be taken are elaborated below:
Tube Breaks or Infectious Spills inside the Biosafety Cabinet (BSC)
- Place absorbent tissue over the spill and apply the disinfectant for 30 mins to 1 hour.
- Carefully collect contaminated sharps material and place it in a puncture-resistant container for disposal.
- Clean the walls of BSC with a layer of absorbent paper towel liberally soaked in disinfectant solution.
- Electrical equipment should be checked carefully before it is used.
- Collect other contaminated material in a biohazard bag for appropriate disposal.
Infectious Spills outside the Biosafety Cabinet
- Everyone should immediately vacate the affected laboratory area.
- The laboratory manager should be informed of the incident immediately.
- Staff must be prevented from re-entering the laboratory for at least 1 hour, to allow aerosols to be removed through the laboratory’s ventilation system and allow time for heavier particles to settle.
- Signs should be posted indicating that entry is forbidden during the clean-up procedure (Figure below).


Figure: Door signs when a spill has occurred
- Appropriate protective clothing and respiratory protection MUST be worn during cleaning.
- Standard operating procedures for spill clean-up MUST be followed.
- The incident should be documented.
Spill Clean-up Procedure to be Used
- Put on gloves, a protective laboratory gown and a respirator.
- Re-enter the affected area.
- Cover spill with cloth or paper towels to contain it.
- Pour appropriate disinfectant over paper towels and the immediate surrounding area.
- Apply disinfectant concentrically, beginning at the outer margin of the spill and working towards the centre.
- Allow sufficient time for the disinfectant to act, before clearing away any material for disposal.
- Clean up the decontaminated area and place any contaminated material in a biohazard bag for disposal.
Tube Break or Spill Inside the Centrifuge: Clean-up Procedure
- The tube should be contained within the safety cup.
- Place the unopened safety cup in the BSC.
- Let sit undisturbed for 30 minutes.
- Disinfect all inside surfaces of the centrifuge.
- Carefully open the safety cup in which break occurred, place lid, top-down, next to the cup.
- Remove uncompromised tubes and disinfect the outer surface of tubes by wiping with disinfectant-soaked cotton/ gauze.
- Pour disinfectant into cup and lid, taking care not to splash.
- Let stand for 30 minutes, pour off disinfectant and place the broken tube into the discard container to be autoclaved.
- Disinfect the entire surface of the BSC.
- Wash the cup and lid, and insert in hot soapy water.
- Remove the O-ring to clean underneath, air-dry, and re-silicone the O-ring.
Resources
- World Health Organization Tuberculosis Manual 2012, p42.
- World Health Organization Lab Safety Manual, 3rd edition.
Kindly provide your valuable feedback on the page to the link provided HERE
Spill Clean-up Kit in TB Lab Settings
ContentThe spill response kits in a tuberculosis (TB) lab should be made available to deal with any contaminated or hazardous fluid spillage. Spill response kits should be made available:
- 1 kit placed outside the TB containment laboratory
- 1 kit placed inside the laboratory
The spill cleanup kit should include the items listed below:
- Hypochlorite solution (or 5% Phenol) stored in an opaque bottle
- Respirators (1 box)
- Gloves (1 box)
- Laboratory gowns (4-6 disposable gowns)
- Dustpan and brush (for disposal if necessary)
- Paper towels
- Soap
- Sharps container
- Biohazard bags
Note: Hypochlorite solution has a limited shelf life. For a large spill, it may be better to prepare the disinfectant solution at the time of clean up
Figure: BSL lab spill kit with spill incident logbook 
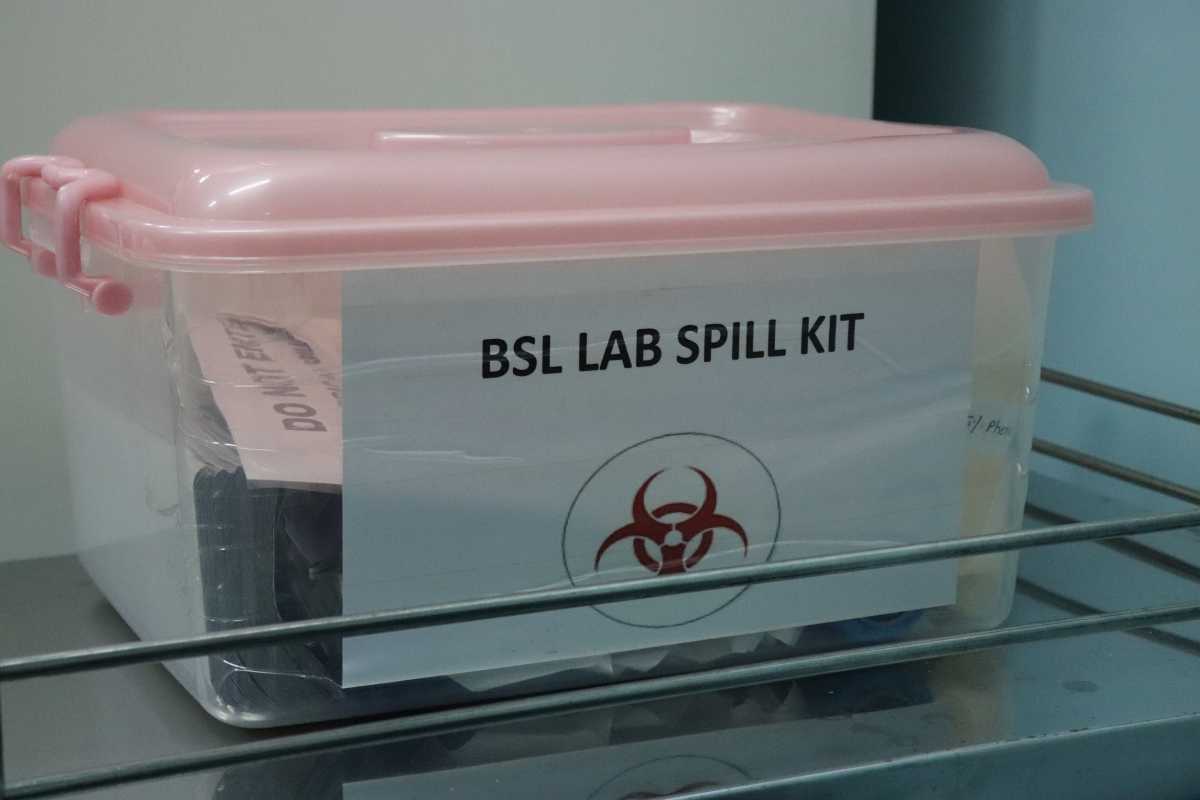
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Chemical Safety in TB Lab Settings
ContentWorkers in microbiological laboratories are not only exposed to pathogenic microorganisms, but also to chemical hazards. Therefore, knowledge of the toxic effects of these chemicals, exposure routes and hazards, that may be associated with handling and storage, is important.
A few important general safety measures against chemical exposure in laboratories are:
- Wear eyeglasses when using a chemical that can cause fumes or vapours.
- Wear gloves at all times.
- Wear lab coat all the time.
- Do not eat in laboratories.
- Tightly secure lids of all chemical bottles when not in use.
- Read chemical cautions on the labels carefully for all chemicals before use.
- Label the chemicals with appropriate icons to mark the potential risks (Figure).
Safety data sheets or other chemical hazard information are available from chemical manufacturers and/or suppliers. These should be part of the safety or operations manual and easily accessible.
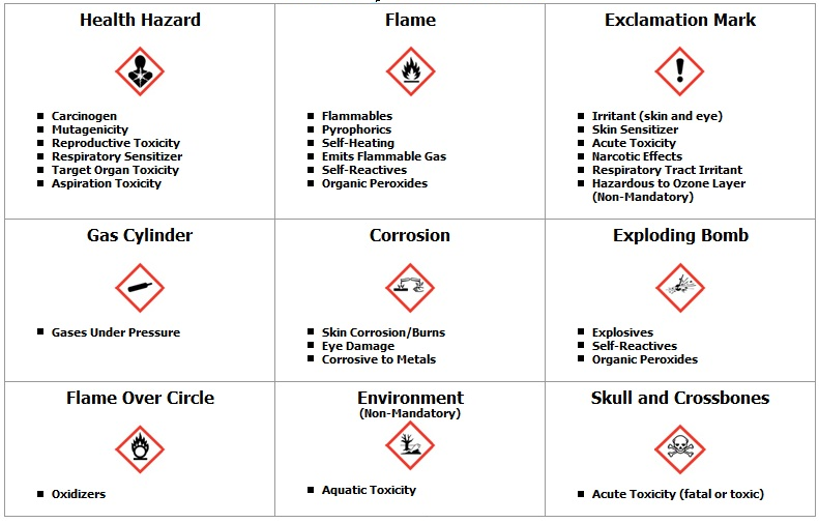
Figure: Different symbols on chemical or reagent bottles and their meaning
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Fire Safety in TB Lab Settings
ContentFor a fire to start, three components of the triangle namely: Fuel, Oxygen and Ignition sources need to come together (Figure).
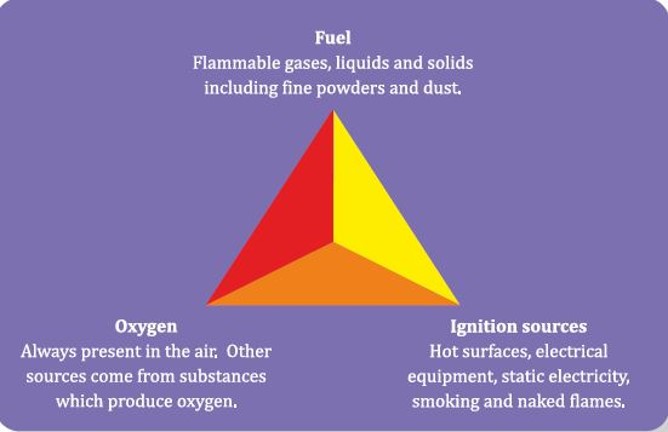
Figure: Components that cause a fire on coming together

- Fire will not start if any one of these components is missing.
- The laboratory personnel should ensure these three components will not come together, to reduce the chance of fire in the laboratory.
- Engineering controls and a variety of fire prevention and suppression strategies should be part of laboratory safety protocols.
Common Causes of Fires in TB Laboratories
- Electrical circuit overloading
- Poor electrical maintenance, e.g., poor and perished cable insulation
- Excessively long gas tubing or long electrical leads
- Equipment unnecessarily left switched on
- Equipment that was not designed for the laboratory environment
- Open flames
- Deteriorated gas tubing
- Improper handling and storage of flammable or explosive materials
- Improper segregation of incompatible chemicals
- Sparking equipment near flammable substances and vapours
- Improper or inadequate ventilation
Considerations for Fire Safety
- Close cooperation between safety officers and local fire prevention officers is essential.
- The effect of fire on the possible dissemination of infectious material must be considered as this may determine whether it is best to extinguish or contain the fire.
- Fire warnings, instructions and escape routes should be displayed prominently in rooms, corridors and hallways.
- Fire-fighting equipment should be placed near room doors and at strategic points in corridors and hallways.
- Fire extinguishers should be regularly inspected and maintained, and their shelf-life kept up to date.
General Measures to Handle Fire in the Laboratory
- All lab personnel must learn how to operate a fire extinguisher.
- Fire extinguisher must be inspected annually and replaced as needed.
- Laboratory should have appropriate class of fire extinguisher.
- In general Class BC or class ABC extinguisher is appropriate.
- The contact information for reaching out to the fire department, should be posted at various locations in the lab.

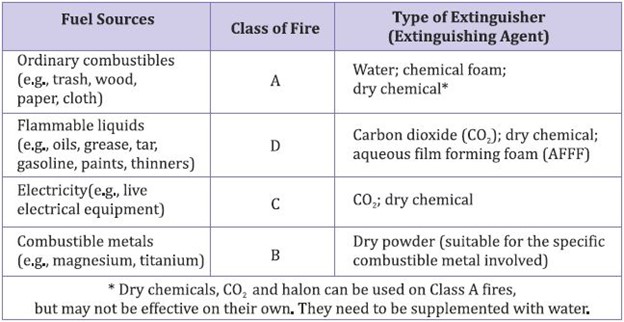
Table: Types and Uses of Fire Extinguishers
Prevention of Fire due to Electrical Hazards
- Electrical installations and equipment should be inspected and tested regularly, including earthing/ grounding systems.
- Circuit-breakers and earth-fault-interrupters should be installed in appropriate laboratory electrical circuits.
- Circuit-breakers do not protect people; they are intended to protect wiring from being overloaded with electrical current, and hence to prevent fires.
- Earth-fault-interrupters are intended to protect people from electric shock.
- All laboratory electrical equipment should be earthed/ grounded, preferably through three-prong plugs.
- All laboratory electrical equipment and wiring should conform to national electrical safety standards and codes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen