-
DR-TB HIV Coordinator: General concepts of SCM
FullscreenSupply Chain Management
ContentSupply Chain Management (SCM) is the handing of flow of goods and material from point of origin to point of consumption, with the objective to ensure that the supplies are present for utilization without any interruption. It covers everything from procurement and sourcing of raw materials to delivery of final product to the consumer, along with the related logistics. It will also include the related information systems that enable monitoring and exchange of information.
Effective SCM ensures the following:
- Continuous availability of quality-assured medicines/ products at the right time and at all healthcare levels.
- Minimizes wastage by preventing expiry of drugs at all levels, maintenance of adequate stock levels and accurate records.
- Maximizes patient care by coordination in all departments and by minimizing human errors/ medication errors.
- Economically viable by minimizing monetary loss (e.g., through pilferage) and optimizing cost via bulk purchasing or according to consumption needs.
Robust supply chain management systems have two main components:
- Physical flow: Involved the movement and storage of supplies
- Information flow: Allows the various stakeholders to coordinate and control the flow of supplies
Resources
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
- NTEP Training Modules 5-9 for Programme Managers & Medical Officers, 2020.
- Guidelines for Programmatic Management of Drug-resistant TB, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which of the following statements are correct about supply chain management?
It is useful to ensure a continuous supply of good quality medicines.
It is an essential activity that must be undertaken by health programmes.
It helps reduce the cost burden on the healthcare system.
All of the above
4
Effective SCM is an essential activity that ensures a continuous supply of good quality medicines and cost optimization.
Principles of Supply Chain Management
ContentTo ensure successful implementation, sustainability and quality services under the National TB Elimination Programme (NTEP), some guiding principles in Supply Chain Management (SCM) and drug logistics are to be ensured. These are showcased in the figure below.

Figure: Principles of SCM under NTEP
Important Points
- Timely procurement, uninterrupted supply and maintenance of stock and in-time distribution of anti-TB drugs and other consumables are essential for quality services.
- Monitoring of drug supply from the central to peripheral health institute level through web-based real-time software, Nikshay-Aushadhi, is crucial to avoid under-stocking (and delays in treatment initiation) and over-stocking (resulting in wastages).
- Maintaining appropriate storage and stacking norms i.e., different batches of drugs with different dates of manufacture and expiry are stored separately so as to facilitate First-expiry-first-out (FEFO) principles, viz., drug batches with the most recent expiry are issued first.
Under FEFO, the storekeeper at the drug store is responsible for:
- Installing appropriate tools to periodically monitor controls over the expiry position of drugs.
- Exercising prudence in the case of short-expiry drugs, wherein distribution is on a rational basis that considers the utilization pattern. This includes the following:
- The storekeeper marks ‘Expiry Dates’ in Bold Letters 3” to 4” in size, on the drug cartons with a marker pen, for easy identification and control of drugs immediately on their arrival.
- Routine monitoring of the stock position of all drugs.
- Maintaining proper records.
- Analyzing shelf-life analysis of drug stocks at all levels regularly.
Resources
- NTEP Training Modules 1-4 for Programme Managers & Medical Officers, 2020.
- NTEP Training Modules 5-9 for Programme Managers & Medical Officers, 2020.
- Guidelines for Programmatic Management of Drug-resistant TB, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is FEFO?
FEFO is the division that manages drug receipts under NTEP.
FEFO is a supply chain principle that is used to forecast consumables that are required by the programme.
FEFO means First-expiry-first-out and it is a principle to be followed when issuing drugs/ consumables.
None of the above
3
FEFO means First-expiry-first-out and it is a principle to be followed when issuing drugs/ consumables.
Drug distribution flow
ContentUnder the National TB Elimination Programme (NTEP), the anti-TB drugs are procured at the centre level by the Central TB Division (CTD), Ministry of Health and Family Welfare (MoHFW), and supplied to the central warehouses.
From the central level warehouses, the drugs are supplied to different State Drug Stores (SDS) and further distributed to District Drug Stores (DDS) and sub-district level (TB Unit (TU) Store and Peripheral Health Institute (PHI)).
This movement of drug flow is monitored in real-time through Ni-kshay Aushadhi.

Figure: Flowchart Showing the Overview of Distribution of Drugs
Abbr: CMSS: Central Medical Services Society; GDF: Global Drug Facility; CTD: Central TB Division; GMSD: Government Medical Store Depot; SDS: State Drug Store; DDS: District Drug Store; TU: TB Unit; PHC: Primary Health Centre; PHI: Peripheral Health Institute.
Resources
Logistics
Content'Logistics' is the process of planning and implementing the efficient transportation and storage of supplies (drugs, consumables and other related items) from the point of origin to the point of consumption through a systematic mechanism.
ImageFigure: Flowchart depicting overview of logistics under National TB Elimination Programme (NTEP)
Resources
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, CTD, MoHFW, India, 2018.
- Procurement, Supply Chain Management & Preventive Maintenance, Module 6, CTD, MoHFW, India.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test From where do the PHIs receive supplies? GMSD CMSS SDS TU 4 The PHIs receive the supplies from the TU. Yes Yes Ni-kshay Aushadhi
ContentNi-kshay Aushadhi is a web-based portal that deals with the management of stocks (anti-TB drugs, consumables and other commodities) across all the stocking points across the National TB Elimination Programme (NTEP), i.e., Government Medical Store Depot (GMSD), Central Medical Services Society (CMSS) warehouses, State Drug Stores (SDS), District Drug Stores (DDS), TB Units (TUs) including Peripheral Health Institutes (PHIs).
Ni-kshay Aushadhi also helps in real-time management of stock position, providing expiry details of commodities, routine/ Additional Drug Request (ADR) Requirements, and patient-wise consumption of drugs at all levels.
Table 1: Nikshay Aushadhi Stakeholders
User Services Central TB Division (CTD)
1. Quantification
2. Purchase Order (PO) generation
3. Quality control
4. State warehouse Drug Transfer Advice (DTA)
5. Monitoring of nation-wide stock & expiry
CMSS warehouse
1. Quantification
2. Purchase request generation
3. Advance shipment details
4. Supplier delivery details
5. Release Order (RO) to SDS
6. Acknowledge desk
7. Transfer of drugs to other warehouses
GMSD
1. Quantification
2. Acknowledge desk
3. Advance shipment details
4. Transfer of drugs to other warehouses
Supplier
1. View Purchase Order (PO) and delivery schedule
2. Enter dispatch details
3. View receipt
4. Demand vs issue
SDS/ DDS/ Drug-resistant TB Centre (DR-TBC)/ TU
1. Routine/ ADR & dispatch to sub-stores
2. Acknowledge desk
3. Transfer of drugs to other warehouses
4. Issue voucher DTA
5. Box preparation, box modification, unpacking and box completion
6. Local purchase
PHI
1. Routine/ ADR
2. Acknowledge the receipt of drugs
3. Issue to patient
4. Return from patient
The Ni-kshay Aushadhi can be used for the following purposes:
- Quantification and forecasting
- Monitoring and distribution
- Data management and analysis
- Recording and reporting of the drugs related data
- Training and capacity building
- Quantification of drugs
- Issue/ dispatch
- Return of drugs
- Drug request management - Routine/ ADR
- Stock management (like drug inventory, Physical Stock Verification (PSV))
- Packaging/ repackaging
- Receive from store/ Acknowledge desk
- Quality control management
ImageFigure: Supply chain management in NTEP through Ni-kshay Aushadhi; Source: Ni-kshay Aushadhi Manual
Resources
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct Answer Correct Explanation Page ID Part of Pre-Test Part of Post-Test Ni-kshay Aushadhi can be used for the quantification and forecasting of drug stocks. True False 1 The Ni-kshay Aushadhi can be used for the following purposes:
- Quantification and forecasting
- Monitoring and distribution
- Data management and analysis
- Recording and reporting of the drugs-related data
- Training and capacity building
- Quantification of drugs
- Issue/ dispatch
- Return of drugs
- Drug request management - Routine/ ADR
- Stock management (like drug inventory, PSV)
- Packaging/ Repackaging
- Receive from store/ Acknowledge desk
- Quality control management
- Miscellaneous
Yes Yes Overview : Return and Reconstitution
ContentReturn
- Return is the process of returning the drug to the parent/ issuing store. This is generally followed whenever there are situations like lost to follow-up, transfer out, death, etc.
- Ni-kshay Aushadhi is being used to record and report the process.
- At present, the return of drugs from the patient/ treatment supporters is taking place in Ni-kshay (in Drug Dispensation Module)
- All the unconsumed drugs should be brought back by the treatment supporter to the Peripheral Health Institute (PHI)---->Tuberculosis Units (TU)---->District TB Centre (DTC) within the shortest possible time in order to ensure that they can be re-used in the future.
- Return of Bedaquiline (Bdq): Partially used Bdq bottle should be sent back to State Drug Stores (SDS) wherein it will be accounted for.
- Return of Delamanid (Dlm): Leftover Dlm tablets should be returned back to the District Drug Stores (DDS).
Reconstitution
- Reconstitution is defined as the process of re-packaging the returned anti-TB drugs in the event of loss to follow-up/ death/ discontinuation for any reason, back into a full treatment course for issuing to other patients.
- If the expiry of the remaining drugs is less than six months, the same may be issued at the Nodal Drug-resistant TB Centre (NDR-TBC) for patients while they are admitted and later adjusted from the long expiry bottle that is issued on discharge.
- The reconstitution exercise is carried out only for the Bedaquiline drug and is done at the SDS.
- First Expiry First Out (FEFO) principle should be strictly followed while issuing re-constituted drugs to the patients and also be cautious about the reconstituted drugs belonging to the different expiry batches.
- The reconstituted drugs should be accounted for and reported in Ni-kshay Aushadhi through the Box-preparation module under the Packaging/ Re-packaging service.
Resources
- Standard Operating Procedure Manual Procurement & Supply Chain Management, CTD, MoHFW, India, 2018.
- Return from Patient, Ni-kshay Aushadhi User Manual, CTD, MoHFW, India.
- Packaging and Repackaging, Ni-kshay Aushadhi User Manual, CTD, MoHFW, India.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Where is the reconstitution of Bdq drugs carried out?
District Drug Store
State Drug Store
Tuberculosis Unit
Peripheral Health Institute
2
The reconstitution of Bdq drugs should be carried out only at the State Drug Store.
Yes
Yes
Reconstitution Register
ContentA Reconstitution Register (RR), as the name suggests, is a register used to record the details of drug boxes reconstituted from the leftover/ remaining/ unused drug boxes of patients who have defaulted, died, failed treatment, or transferred out.
This register in the format shown below (Figure) is maintained at all the District Tuberculosis Centres (DTCs).
ImageFigure: Reconstitution Register
Abbr: IP: Intensive Phase; CP: Continuation Phase; PC: Product Code; PP: Prolongation Pouch
The RR consists of 13 columns designated by small alphabets 'a' to 'm'. The information in the relevant columns is filled by the pharmacist or storekeeper of the district under the supervision of the District Tuberculosis Officer (DTO).
Reconstitution of the drugs and hence updation of the RR is done once a quarter. However, this interval can sometimes be shorter based on the need.
The following key information is recorded in the RR:
- Serial no. and date in columns ‘a’ and ‘b’
- Receipt transactions in columns 'c' to 'e'. These include information about the source and type of the Patient-wise Boxes (PWB) returned for reconstitution in
- Constituents of the returned PWBs in columns 'f' to 'j'. Separate columns for IP and CP are provided.
- Withdrawal transactions in columns 'k' to 'm'.
Note: Receipt transactions shall be recorded in the RR in blue ink, whereas withdrawals for reconstitution purposes, shall be in red ink for clear demarcation
At present, reconstitution is carried out for Bedaquiline (BDQ) only. Unlike other second-line drugs, reconstitution of BDQ takes place at the State Drug Store (SDS) under the supervision of the State Tuberculosis Officer (STO). The number of the tablets received back and the newly reconstituted bottles from these tablets are recorded in appropriate columns of the reconstitution register.
Resources
- Standard Operating Procedures Manual for State Drug Stores, Central TB Division, MoHFW, GoI, 2012.
- Standard Operating Procedure Manual, Procurement & Supply Chain Management, CTD, 2017.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Reconstitution registers are maintained at? DMC PHI DTC STC 3 Reconstitution registers are maintained at all the District Tuberculosis Centres (DTCs). YES YES Recording reconstitution in Nikshay Aushadhi
ContentBox reconstitution means to unpack the prepared boxes into loose medicine. The loose medicine is then added back to the inventory of the store where reconstitution is done. Any box whether complete, incomplete, or modified can be reconstituted. Reconstitution can be done at State Drug Store (SDS) and District Drug Store (DDS) level only and once reconstituted, the box will no longer exist in the inventory. The process of reconstitution in Nikshay Aushadhi is known as box unpacking and involves the following steps:

Figure 1: Overview of process of recording of reconstitution in Nikshay Aushadhi
Detailed procedure:
Step 1: Go to the Nikshay Aushadhi website and click on login. Enter your user ID, Password, the captcha text and click login.
Step 2: Reach the ‘Box unpacking’ window following the path Home-menu>services>packing/repacking>Box unpacking and select your store. Select the TB subcategory then drug name and click on the ‘go’ tab.
Step 3: The system will show all the available boxes (complete, incomplete, and modified) with batch number, expiry Date, stock Quantity, and Stock Status. Select the desired box to be unpacked.

Figure 2: Box unpacking window in Nikshay Aushadhi
Source: Nikshay Aushadhi portal
Step 4: Once selected the system will show details of the drugs in that box. Any quantity less than or equal to the stock quantity of that box can be unpacked. For example, if the stock quantity of the selected box is 100 then any number of boxes between 1 and 100 van be unpacked. Specify the number of the boxes to be unpacked and then click the ‘save’ tab to complete the process. Upon completion, the system will generate the ‘box unpacking complete’ alert. After unpacking the box, the drugs from the box will be added loose into the inventory.

Figure 3: Saving details of box reconstitution
Source: Nikshay Aushadhi portal
Resources
Nikshay Aushadhi Manual-Central TB Division, Ministry of Health and Family Welfare, Government of India
Assessment
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Box unpacking can be done at
SDS
DDS
Both 1 and 2
At any store
3
Box unpacking can be done at SDS, and DDS level only
YES
YES
Disposal of expired supplies
ContentExpiry management of supplies is crucial to avoid financial losses and harm to patients.
If any drug expires due to reasons beyond control, the write-off of expired drugs should be as per the guidelines given in NTEP National Strategic Plan. As per NSP, the State is allowed to write off up to 2% of the cost of the annual supply of drugs on implementation of Drug Sensitivity Testing (DST) guided treatment and 2% cost of rapid molecular test cartridges. The expired stock should be disposed-off as per the Bio-medical Waste (Management and Handling) guidelines of Govt. of India.
Disposal of Expired/Discarded Medicines
Colour of the bag to be used: Yellow
Image
Figure 1: Disposal of expired supplies according to Bio-Medical Waste Management Rules 2016
Updating in Ni-kshay Aushadhi
To dispose of or remove the expired/rejected drugs from the online inventory, follow the steps below:
- Go to the ‘Write-Off/Disposal’ process in Stock Management,
- Click on the ‘Request’ button to generate the disposal request,
- Select the ‘Expired or Rejected’ category, and the system will show the respective drugs
- Select the drug with an expired batch and enter the quantity
- Click on the ‘Save’ button.

Figure 2: Write-off/disposal register in Nikshay Aushadhi Source: Nikshay Aushadhi portal

Figure 3: Entering details of expired drugs in the write-off/disposal register in Nikshay Aushadhi Source: Nikshay Aushadhi portal
Steps to follow
- After saving, select the request and click on the ‘Write-off’ button,
- Verify the drug details and select the type of write-off as ‘Burned/Buried’,
- Enter the ‘Remarks’, and click on the ‘Save’ button,
- System will generate the voucher, and the drug quantity will be deducted from the inventory.

Figure 3: Expired drug details in Nikshay Aushadhi Source: Nikshay Aushadhi portal
Condemnation of laboratory supplies

Figure 4: Process of condemnation of laboratory supplies which are non-functional, obsolete, non-reparable equipment in NTEP’s laboratories
Information is required in below mentioned format to condemn the lab equipment:

Figure 5: Form GFR 10 Source: General Financial Rules 2017, Ministry of Finance, Department of Expenditure, GoI
The request for the replacement of the equipment condemned has to be submitted to State TB Officer (STO)/Central TB Division (CTD) in the below-mentioned format:

Figure 6: Annexure 4 for details of equipment for condemnation Source: Guidelines for the condemnation and replacement of Tuberculosis (TB) laboratory equipment under the Revised National Tuberculosis Control Programme (RNTCP) 2019
Resources
1. Guidelines for Management of Healthcare Waste as per Biomedical Waste Management Rules, 2016
Assessment:
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Disposal of expired supplies is done in which colour bag? Yellow Red White Blue 1 Discarded or expired medicine in yellow coloured non-chlorinated plastic bags color coding and type of container
ContentThe National TB Elimination Programme (NTEP) utilizes different coloured bags for the segregation of waste generated in TB laboratory settings as shown in the table below.
Table: Waste Segregated and Collected According to Colour-coded Bags
Yellow bag:
- Broomsticks
- Parafilm tape & plastic bag – contaminated with samples
- Löwenstein-Jensen (LJ) media without the bottle
- Microbial sample/ blood/ body fluids contaminated paper/ cotton/ swab
- Blood bags
- Microbiological cultures Truenat chips (MTB/ Rif)
- Used mask/ gowns
- Expired medicines/ drugs/ antibiotics

Red bag:
- Specimen collection tubes
- Sputum cups
- Cartridge Based Nucleic Acid Amplification Test (NAAT)/ Truenat cartridges
- Infected plastic
- Contaminated tips
- Pasteur pipettes
- Polymerase Chain Reaction (PCR) tubes
- Mycobacteria Growth Indicator Tube (MGIT) tubes
- Disposable LJ tubes
- Contaminated falcon tubes
- Used gloves
- Contaminated droppers
- Empty Cartridge-based Nucleic Acid Amplification Test (CBNAAT) reagent bottles

Blue bag:
Glass slide in Truenat machine and used microscopy slides. Slides should not be broken.

All these bags are to be labelled with the Biohazard logo (figure below) on them.

Figure: Biohazard Logo
- Waste generated in the Culture Drug Susceptibility Testing (CDST) laboratories is autoclaved prior to segregation in colour-coded bags.
- The biohazard materials are collected and handed over to handlers authorized by the pollution control board.
- Personnel handling/segregating biomedical wastes must use appropriate Personal Protective Equipment (PPE) and should be trained in spill management.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, 2021.
- Guidelines for Management of Healthcare Waste as per Biomedical Waste Management Rules, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
-
DR-TB HIV Coordinator: Stocking Norms
FullscreenStorage norms
ContentTo preserve the quality of medicines, good storage practices should be in place at all levels, which requires that staff are appropriately trained and storage conditions adequate.
General Guidelines on Storage of Drugs
Mechanisms to improve TB stock management in central or peripheral drug stores are:
- Stock rotation follows a First-expiry, First-out (FEFO) approach.
- The cleanliness of the area is ensured.
- The environment of the warehouse protects medicines from factors that could inhibit their effectiveness or use, such as sunlight, heat, cold, moisture, pests and theft.
- The stock area is divided into zones for easy location of different products.
- There is a designated area for second-line TB medicines.
Storage Guidelines under the National TB Elimination Programme (NTEP)
Under NTEP, space requirements change at the state, district and peripheral levels according to the NTEP stocking norms, but all other requirements remain the same as shown in the table below. The State TB Officer (STO)/ District TB Officer (DTO) must ensure that the pharmacist/ storekeeper adheres to the following guidelines on the proper storage of drugs.
Table: Guidelines for the Storage of Anti-TB Drugs in NTEP Drug Stores
Space Requirements at the State Drog Store (SDS)
- For every ten lakh population, provision should be made for the storage of about 45 cartons (of 20 boxes each), of Patient-wise Boxes (PWBs) for new cases (PC-1) and PWBs for retreatment cases (PC-2) taken together.
- This is approximately equivalent to 6 months requirement of drugs. For this volume of drugs, the minimum space requirement may be approximated as 50 cubic feet.
- For loose drugs, space provision would be 10% of space allocated to PC-1 and PC-2 PWB cartons. These could alternatively be stored in cupboards/ almirahs where volumes are low and should be kept under lock and key. Do not stack drug cartons on the floor or on top of one another.
Room Requirements
- The store should preferably comprise one large room. Where multiple rooms exist, these should be contiguous or proximate to each other.
- The ceiling must have a height of at least 5 metres.
- There should be a lockable door and at least one window with a grill and wire meshing.
- Properly lit with extra light points for plugging in the required office equipment.
- An even-level, ‘pukka’ floor.
Stacking Requirements
- Ensure that different drug/ consumable items are clearly segregated and stacked on separate racks.
- Different batches of drugs with different dates of manufacture and expiry are stored separately to facilitate FEFO principles (drug batches with the most recent expiry are issued first)
- Mark ‘Expiry Dates’ in Bold Letters 3” to 4” in size, on the drug cartons with a marker pen, for easy identification and control of drugs immediately on their arrival.
- Separate and dispose of damaged or expired products without delay as soon as approval of the same has been received, according to the biomedical waste guidelines.
Temperature and Humidity Control
- To keep humidity levels below the maximum 60% recommended for storage of drugs ensure appropriate ventilation and air circulation, and do not open cartons/ drug boxes unless necessary.
- Hydro thermometers are to be installed up to TB Unit (TU) drug store levels to monitor humidity and temperature regularly.
- Overhead exhaust fan required.
- Plastered walls and ceiling with whitewash without any kind of seepage in the room.
- The store should be clean, dry and well-ventilated.
- Ceiling and sidewalls should preferably be insulated, ensuring that the ambient temperature during peak summer does not result in damage to anti-TB drugs. The ambient temperature may be taken as 15-25°C or depending on climatic conditions, up to 30°C.
- PWBs/ cartons should be placed on shelves ensuring that there is sufficient space between shelves and walls of the storeroom.
- A regular power supply should be available for air conditioning.
Protection from Sunlight
- Shade the windows or use curtains if they are in direct sunlight.
- Keep products in cartons/ drug boxes.
- Do not store or pack products in sunlight.
- Maintain trees around the premises of the drug store to help provide shade and cooling. Check their condition regularly to prevent any untoward incidents.
Fire Safety
Ensure that the fire safety equipment is available and accessible, and that personnel are trained to use it.
Others
- Store medical supplies separately, away from rodents, insecticides, chemicals, old files, office supplies and other materials.
- Stores should not have any odour or indications of contamination and should be sanitised periodically including pest control measures.
Resources
- NTEP Training Modules (5-9) for Programme Managers & Medical Officers, 2020.
- Standard Operating Procedures Manual for State Drug Stores, NTEP, 2012.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Concerning storage of anti-TB drugs, which of the following is correct?
Stock rotation follows a First-expiry, First-out (FEFO) approach.
Store drugs away from direct sunlight.
Hydro thermometers are to be installed up to TU drug store levels to monitor humidity and temperature regularly.
All of the above
4
Appropriate anti-TB drug storage conditions include keeping drugs away from direct sunlight, maintaining appropriate temperature and humidity and following a FEFO approach.
Yes Yes Paper-based Format: Stock Register
ContentThe Stock Register is a paper-based recording register kept in drug stores of the National Tuberculosis Elimination Programme (NTEP). It is useful to ensure uninterrupted supply of anti-TB drugs to the patient. The stock register is maintained at the state, district, and tuberculosis unit (TU) drug stores, by the storekeeper.
Uses of the Stock Register
- It is used for recording receipt, issue and balance of stocks of drugs and consumables.
- It provides drug stock visibility as well details such as batch numbers and expiry dates (thus, facilitating the issue of drugs according to First Expiry, First Out (FEFO) principles).
- It provides a receipt for reconstituted Patient-wise Boxes (PWBs) since these are recorded in the District TB Centre (DTC) stock register.
Important Points to Note
- All receipts should be entered neatly in red colour including transfers from other districts.
- The issue of drugs should be entered in blue colour including all transfers out to other districts/state drug stores.
- The medical officer at the peripheral health institution (MO-PHI) should monitor the monthly replenishment of drugs stock to treatment supporters if drugs are not already given and update the drug stock register accordingly.
The figure below shows the NTEP stock register format.

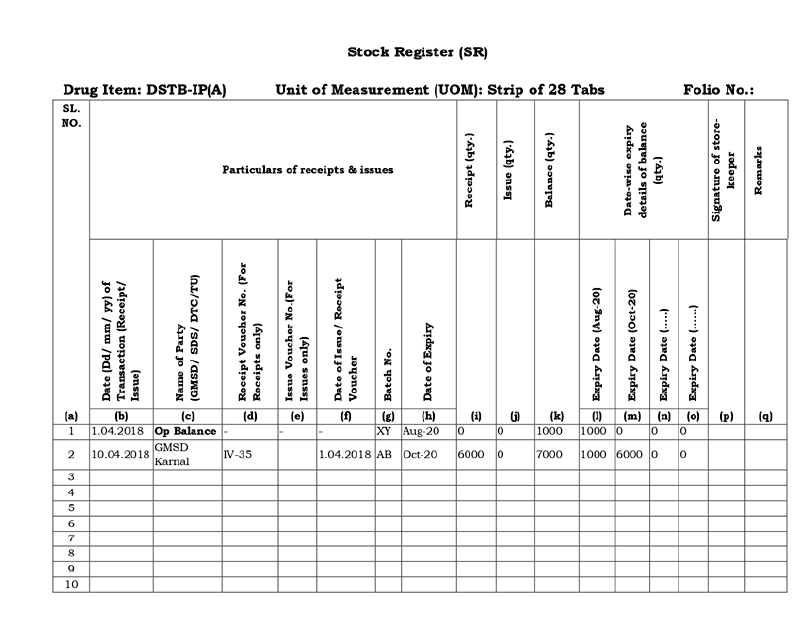
Figure: The NTEP Stock Register; Source: Standard Operating Procedures Manual for State Drug Stores, 2012, p.79
Resources
- NTEP Training Modules 5-9 for Programme Managers & Medical Officers, 2020.
- Standard Operating Procedures Manual for State Drug Stores, 2012.
Kindly provide your valuable feedback on the page to the link provided HERE
-
DR-TB HIV Coordinator: Supply Chain Processes
FullscreenPreparation of Patient-wise Boxes [PWB] for DR-TB Patients
ContentUnder the National TB Elimination Program (NTEP), Drug-resistant TB (DR-TB) patients receive standardized DR-TB treatment boxes (called Patient-wise Boxes - PWB) for each month. These boxes are either with the health facility nearest to DR-TB patients (DOT centre) or with the treatment supporter who is involved in the care of DR-TB patients.
Steps involved in the preparation of second-line drug boxes for DR-TB patients:
- NTEP State Drug Store (SDS) receives quality-assured second-line drugs from the centre through the General Medical Stores Depot (GMSD) and/or Central Medical Services Society (CMSS).
- The loose medicines are utilized for individual patient-wise box creation at the SDS/DDS by the store staff.
- Each patient-wise second-line drug box is prepared according to the NTEP standard box preparation guidelines utilizing the logistics, labels and drugs, and the same details are entered in Ni-kshay Aushadi for maintaining accountability of all drug boxes.
- Each patient-wise box needs to clearly mention the batch number of different drugs it contains, the expiry of the drug box and individual drugs, and any specific instructions that need to be followed, e.g., temperature, humidity or local storage.
- Second-line drug regimen box for INH mono/ poly regimen can be prepared at the district level considering its 6-month duration with the selected 4 drugs while Bdq/ Dlm regimen-based boxes are usually prepared at SDS and provided to District Drug Stores (DDS).
The exercise of preparing the standard patient-wise boxes is done at the NTEP DDS under the guidance and supervision of the District TB Officer (DTO). In such cases, drugs to the district will be supplied in loose form through Nikshay Aushadhi.
Resources
Constituents of Monthly Patient-wise Boxes [PWB] for Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
ContentThe patient on shorter oral Bedaquiline-containing multi-drug resistant (MDR)/rifampicin resistant TB (RR-TB) regimen shall be put on Type A and Type B box when initiated on treatment on a monthly patient wise box (PWB). Bedaquiline (Bdq) needs to be issued separately and stopped after 6 months. The patient should be provided with only Type A boxes when started on continuation phase (CP).
Table 1: Constituents of monthly-type A and B PWB of shorter oral Bedaquiline-containing MDR/RR-TB regimen; Source : Guidelines for PMDT, India 2021, p.132-133 TYPE A BOX
(USE IN IP AS WELL AS CP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin#
250/500 mg
250 mg *30tab
250 mg *30tab + 500 mg *30tab
500 mg *60tab
500 mg *60tab
Tab. Clofazimine
50/100 mg
50 mg *30tab
100 mg *30tab
100mg *30tab
100 mg *60tab
Tab. Ethambutol
400/800 mg
400 mg *30tab
800 mg *30tab
400 mg *30tab + 800 mg *30tab
800 mg *60tab
Tab. Pyrazinamide
500/750 mg
750 mg *30tab
500 mg *30tab + 750 mg *30tab
500 mg *60tab + 750 mg *30tab
500 mg *30tab + 750 mg *60tab
Tab. Pyridoxine
50/100 mg
50 mg*30tab
100 mg*30tab
100 mg*30tab
100 mg*30tab
Type B Box
(Use in IP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Isoniazid
100/300 mg
300 mg *30tab
300 mg *60tab
300 mg *90tab
300 mg *90tab
Tab. Ethionamide
125/250 mg
125 mg *30tab + 250 mg *30tab
250 mg *60tab
250 mg *90tab
250 mg *120tab
Bedaquiline bottle
100 mg
1 Jar (Jar of 188 tablets for full course)
# When moxifloxacin prescribed under exceptional condition instead of levofloxacin, the modified box with moxifloxacin (normal dose) can be prepared from standard box at district drug store (DDS)
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Constituents of Patient-wise Boxes [PWB] for Isoniazid [H] Mono/Poly DR-TB Regimen
ContentStandard Patient-wise Boxes (PWBs) are constituted for TB patients initiated on Isoniazid (H) Mono/ Poly Drug-resistant TB (DR-TB) regimen.
This regimen has no segregation in terms of Intensive Phase (IP) or Continuation Phase (CP), hence drugs are provided in a single type of PWB.
Table: Constituents of standard PWB (6 months) of H mono/ poly DR-TB regimen; Source: Guidelines for PMDT, India 2021, p134
STANDARD PWB
CONTINUE FOR COMPLETE TREATMENT Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin
250/ 500 mg
250 mg *180 tab
250 mg *180 tab + 500 mg *180 tab
500 mg *360 tab
500 mg *360 tab
Tab. Rifampicin
150/ 300/ 450 mg
300 mg *180 tab
450 mg *180 tab
300 mg *360 tab
300 mg *360 tab + 150 mg *180 tab
Tab. Ethambutol
400/ 800 mg
400 mg *180 tab
800 mg *180 tab
400 mg *180 tab + 800 mg *180 tab
800 mg *360 tab
Tab. Pyrazinamide
500/ 750 mg
750 mg *180 tab
750 mg *180 tab + 500 mg *180 tab
750 mg *180 tab + 500 mg *360 tab
750 mg *360 tab + 500 mg *180 tab
Tab. Pyridoxine
50/100 mg
50 mg *30 tab
100 mg *30 tab
100 mg *30 tab
100 mg *30 tab
*No separate box for IP and CP.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Central TB Division, MoHFW. Training Modules (5-9) for Programme Managers & Medical Officers, 2020.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, RNTCP.
- Technical and Operational guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Guidelines for Issuing Bedaquiline and Delamanid to the Patient
ContentLoose drugs will be supplied to the Nodal (N)/ District DR-TB Centre (DDR-TBC) from the State Drug Stores (SDS)/ District Drug Stores (DDS) as per the consumption pattern of the previous month and requested via Nikshay Aushadhi.
- On discharge, the patient will be handed over the drugs for one month of consumption for transit.
- The senior DR-TB TB-HIV supervisor under the guidance of the N/DDR-TBC, shall guide the senior treatment supervisor (STS) for arranging the treatment supporter.
- The entire course available with the patient shall be handed over to the treatment supporter.
- For the provision of newer drugs, the table below shows the protocol to be followed for different regimens.
Table: Protocol for New Drug Provision for DR-TB Patients
PATIENT CATEGORY PROTOCOL Patient put on Bedaquiline (Bdq)-containing regimen The entire bottle of Bdq (188 tablets) shall be earmarked for each enrolled patient and handed over to the treatment supporter under the supervision of the senior DR-TB TB-HIV supervisor in every district. Patients put on the Delamanid (Dlm)-containing regimen The entire course of Dlm treatment will be earmarked at the DDS level. However, drugs will be supplied on a monthly basis to the TB Unit (TU) and further to the Health Facility (HF). Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Management of Patient-wise Boxes in Different Scenarios
ContentThere are different scenarios regarding patient-wise box (PWB) management. These are described below.
Scenario 1: Modification in regimen
In this scenario, the nodal and district drug-resistant tuberculosis centre (N/DDR-TBC) committee decides on a regimen modification.
- The district drug store (DDS) prepares modified PWBs from standard available boxes and loose drugs and arranges the supply of these PWBs to the treatment supporter (TS).
- The district TB officer (DTO) ensures that the drugs are supplied as per the modified regimen for all subsequent months.
Scenario 2: Extension of intensive phase
If intensive phase (IP) is extended:
- N/DDR-TBC informs the DTO
- DTO informs the health facility (HF) doctor and respective TB unit (TU)
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
When the patient is switched to continuation phase (CP) in case of a shorter regimen:
- DTO, again, informs HF doctor and respective TU
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
In the case of a longer regimen, after completion of 6-8 months of treatment, Linezolid (Lzd) should be reduced to 300 mg following N/DDR-TBC directives.
All patients must complete their monthly boxes before switching to the subsequent box provided.
Scenario 3: Change in DR-TB regimen
If N/DDR-TBC decides to change the DR-TB regimen of a patient, the DDS supplies a new treatment regimen box from PWB/ loose drugs. Unused drugs including Bdq containers should be sent back to the DDS. In this scenario, the patient should be immediately switched to the new regimen designed by N/DDR-TBC.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the State Drug Store [SDS] in the Constitution of Patient-wise Boxes
ContentThe State Drug Store (SDS) shall constitute drug boxes for:
- Shorter oral Bedaquiline (Bdq)-containing Multi-drug resistant (MDR)/ Rifampicin-resistant Tuberculosis (RR-TB) regimen (Type A and B)
- Longer oral Multi (M)/ Extensively Drug-resistant Tuberculosis (XDR-TB) regimen
- Isoniazid (H) mono/ poly DR-TB regimen
Boxes will be prepared manually only and not through Nikshay Aushadhi software.
From Nikshay Aushadhi drugs will be supplied in loose to districts.
Based on the requirement of districts SDS will supply boxes to respective districts by a hired transport agency or other mechanisms.
Loose drugs will also be supplied from SDS to the District Drug Store (DDS) for modification and preparation of new boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the District Drug Store [DDS] in the Constitution of Patient-wise Boxes
ContentWhen there is a modification in the Drug-resistant TB (DR-TB) regimen as suggested by the Drug-Resistant Tuberculosis Centre (DR-TBC), the district drug storekeeper/ pharmacist prepares the modified boxes from loose Second-line anti-TB Drugs (SLD) supplied from the State Drug Store (SDS).
- The state shall provide the necessary support for capacity building of the District Drug Store (DDS) for carrying out the entire exercise of preparing standardized/ modified patient-wise drug boxes at the DDS level.
- Whenever oral regimens are modified during the course of treatment, the DDS needs to ensure that the change in the regimen should be incorporated into the supply of subsequent boxes.
- A full-time DDS storekeeper/ pharmacist must be mandatorily recruited/ placed for a successful decentralized system of preparation of drug boxes at the DDS level.
- District TB Officer (DTO) and Medical officer-DTC usually perform a random check to understand if there is any challenge in the constitution of patient-wise boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Patient-wise Drug Boxes: Packing Instructions
ContentPackaging of loose drugs into monthly patient-wise boxes should be done under the guidance of those in charge of drug logistics at the state and district levels.
Patient-wise drug box preparation should be done for the following Drug-resistant TB (DR-TB) regimens:
- Shorter oral Bedaquiline-containing Multidrug-resistant/ Rifampicin-resistant TB (MDR/RR-TB) regimen
- Longer oral Multi (M)/ Extensively Drug-resistant TB (XDR-TB) regimen
- Isoniazid (H) mono/poly DR-TB regimen
Packaging Instructions
Patient-wise drug boxes are prepared as per the type of DR-TB regimen and weight bands. One monthly pouch of the capsule Cycloserine (Cs) and tablet Ethambutol (E) each, should be made from a plastic bag with a zip lock facility in which 1 gm pouch of silica gel desiccant should be kept.

Figure 1: Isoniazid (H) Mono/ Poly Regimen DR-TB Treatment Box
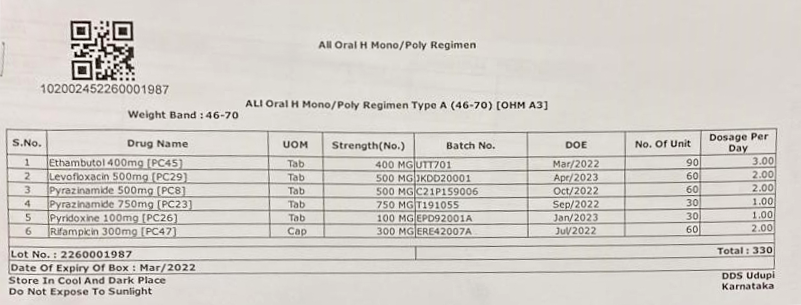
Figure 2: Isoniazid (H) Mono/ Poly Regimen DR-TB Label
In each Type A box, one pouch of silica gel desiccant of 4 gm weight should also be kept.
Labels for the boxes should be developed with the following information (Figures 2 and 3):
- Item-wise name of drugs with a quantity of each drug in the box
- Batch number and Date of Expiry (DOE) of the individual drugs
- DOE of boxes, with expiry date of the drug having the shortest expiry date
- Date of issue of the box from the State Drug Store (SDS)
- Serial number of the box
- Storage instructions on the box for ensuring adequate precautions in storage of the drugs, especially at the treatment supporter level. Some suggested messages are:
- “Store in a cool and dark place, preferably in a clean cupboard”
- “Do not expose to direct sunlight”
- “Keep away from children/unauthorized persons”
- “Box to be closed properly every time after the withdrawal of drugs”.
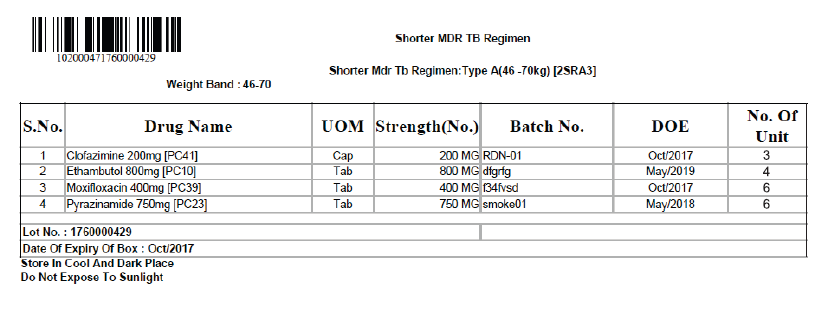
Figure 3: Prototype of a Label (Type A box for shorter DR-TB regimen for the weight band - 46 to 70 kg)
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, MOHFW, GOI.
Kindly provide your valuable feedback on the page to the link provided HERE
Recording box preparation in Nikshay Aushadhi
ContentRecording Box Preparation in Ni-kshay Aushadhi
As per the National TB Elimination Programme (NTEP) guidelines, CTD procures loose medicines for the treatment of DR-TB patients. These loose medicines are packed into Monthly Patient-Wise Boxes (PWB). PWB are prepared only at the State Drug Store (SDS) and District Drug Store (DDS) levels. Depending upon the availability of drugs, two types of boxes can be prepared in Ni-kshay Aushadhi.
Complete box – contains all the required drugs as per the regimen
Incomplete box – has less than the required number of drugs as per the regimen
ImageFigure 1: Overview of the process of recording box preparation in Ni-kshay Aushadhi
Detailed stepwise procedure:
Box Preparation
Step 1: Go to the Ni-kshay Aushadhi website and click on login. Enter your User ID, Password, and then the captcha text and click login.
Step 2: Reach the ‘Box Preparation Window’ following the path Home-menu > Services > Packing/Repacking > Box preparation and select the appropriate options as per the regimen from the drop-down menus of different sections given in the window and then press the ‘Go’ tab.
Image
Figure 2: Box completion window in Ni-kshay Aushadhi
Source: Ni-kshay Aushadhi portal
Step 3: Select the drug from the checkbox. The system will auto-fill the quantity required as per the box preparation quantity. Batch of near expiry date will be highlighted in pink colour. This is to be noted that the expiry date of the nearest expiry drug in the box will be the expiry date of the box itself. You can select any batch as per your requirement.
Image
Figure 3: Entering details of the box
Source: Ni-kshay Aushadhi portal
Step 4: After selecting the batch, click on ‘Add’ button to add the drug to the box. The system will highlight the added drug into the table with green colour. Repeat the process for all the drugs in the regimen. When all the drugs are added into the box as per the required quantity, the system will highlight them in green.

Figure 4: Details of drugs added to the box
Source: Ni-kshay Aushadhi portal
Step 5: Enter the ‘Remarks’ if required and click on the ‘Save’ button. The system will generate the voucher. You can save or print the voucher.

Figure 5: Voucher generated
Source: Ni-kshay Aushadhi portal
Note: If the available number of drugs is less than required, the selection could still be saved, and a voucher generated. Such boxes will be shown in blue in the view boxes section and are called as incomplete boxes.
Box Completion
This process involves adding the remaining drugs to the prepared incomplete boxes and can also be done at the receiving store. The process can be repeated multiple times depending upon the availability of the remaining drugs.
Steps to be followed in Ni-kshay Aushadhi:
Step 1: Reach the ‘Box Completion’ window following the path Menu > Services > Packing/Repacking > Box completion and enter your store name.
Step 2: On selecting the regimen from Tuberculosis (TB) subcategory, those batches with incomplete boxes will be displayed within the ‘Batch’ section. On selecting the batch from the Drug Details section will get displayed.
Step 3: Select a drug from the Drug Details table. Once selected, the batch details table of that drug will appear. Select the batch and add the drug quantity into the ‘To Be Added Qty' column.

Figure 6: Box completion window
Source: Ni-kshay Aushadhi portal
Step 4: Click on the 'Add' button. The selected row in the drug details gets coloured in green, which means that the given drug has been successfully added.
Step 5: Repeat steps 3 and 4 to fill more drugs and complete the box. Click on 'Save' to complete the process and generate the voucher.
Resource
Ni-kshay Aushadhi Manual-Central TB Division, Ministry of Health and Family Welfare, Government of India.
Assessment
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Where can box preparation be done? SDS only SDS and DDS only
TU
DDS only
2
This activity can be done at the SDS and DDS levels only.
Yes
Yes
Reconstitution of Bedaquiline [Bdq] Bottle
ContentThe reconstitution of Bedaquiline (Bdq) bottle should be done at the State Drug Store (SDS).
In case of lost to follow-up/ death/ transferred-out/ treatment stopped patients, Bdq bottle shall be brought back from the treatment centre to the Health Facility (HF) to the TB Unit (TU) to the District TB Centre (DTC) within the shortest possible time.
- A partially used Bdq bottle shall be sent back to the SDS for repackaging.
- The remaining tablets in the bottle received back shall be accounted for in the stock register and Nikshay-Aushadhi at the SDS.
- Upon reconstitution, the bottle shall be accounted for in the stock register (loose tablets to be mentioned in the remarks column) to be issued as per First-expire-first-out (FEFO) principles.
- When reconstitution is done, tablets of the same expiry can be considered using the same container to a maximum of 188 tablets.
- These reconstituted containers shall be used for the treatment of subsequent patients found eligible for Bdq.
- All such drugs that are taken from the new containers shall be collected as a group of 188 tablets of the same expiry and put in a light-resistant container as per the advice from the manufacturer.
- The actual expiry of tablets should be mentioned over the container.
- In the event of the SDS falling short of 188 tablets from an expiry batch, reconstitution can still be done using a number of tablets to complete 188 tablets with another expiry batch.
- In such a case, tablets of the respective expiry should be retained in their same respective containers and issued to patients and providers with counselling to consume the tablets with the nearest expiry first.
- If the expiry of the remaining tablets is less than six months, the same shall be consumed at the Nodal/ District Drug-resistant TB Centre (N/DDR-TBC) for admitted patients. It will be adjusted from the new long expiry bottle on discharge.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, MOHFW, GOI.
Kindly provide your valuable feedback on the page to the link provided HERE
Storage of Drugs
ContentGood storage conditions and safe custody of drugs is important to ensure quality of drugs. Temperature and humidity control systems should be appropriately designed, installed, qualified and maintained, to ensure that the required storage conditions are maintained.
The State TB Officer (STO)/ District TB Officer (DTO) must ensure that the pharmacist/ store-keeper adheres to the following guidelines on proper storage of drugs.
Temperature
- The storage temperature should be 250C and maintained with an air conditioner wherever applicable.
Humidity
- Humidity levels should be below 60% and maintained with a dehumidifier, wherever required
Power Supply
- Regular power supply should be available for cooling devices (AC, ceiling fans etc) and dehumidifiers.
Drug Safety
Storage Room
- Should be cleaned and disinfected regularly, should be dry, well-lit and well-ventilated
- Should be free from any water penetration, rodent and pests
- Should have fire safety equipment in place
- The drugs should not be exposed to direct sunlight.
Stacking
- The shelves should be placed in such a way that there is sufficient space around for air circulation and free movement of personnel.
- Similar boxes should be stored adjacent to each other and stacked as per their expiry dates.
- The drugs expiring early should be placed closer to the ground and as those expiring late should be place at higher levels.
- Expired drugs should be segregated, sealed and stored in a separate part of the store so as to avoid issue to patients
Labelling
- The identification label, expiry date & manufacturing date of the anti-TB drugs should be marked with a bold marker pen on the visible side of the carton.
Resources
- WHO Technical Report Series, No. 908, 2003; Annex.9: Guide to Good Storage Practices for Pharmaceuticals.
- Procurement, Supply Chain Management & Preventive Maintenance, Module 6, CTD, MoHFW, India.
- Standard Operating Procedure Manual Procurement & Supply Chain Management, CTD, MoHFW, India, 2018.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the maximum acceptable humidity levels in a drug store to maintain drug efficacy?
10%
25%
45%
60%
4
Humidity levels should be below 60% and maintained with a dehumidifier wherever required in order to ensure shelf life and efficacy of the drugs.
Yes
Yes
Fullscreen