CBNAAT Cartridge Loading
- Read more about CBNAAT Cartridge Loading
- Log in to post comments
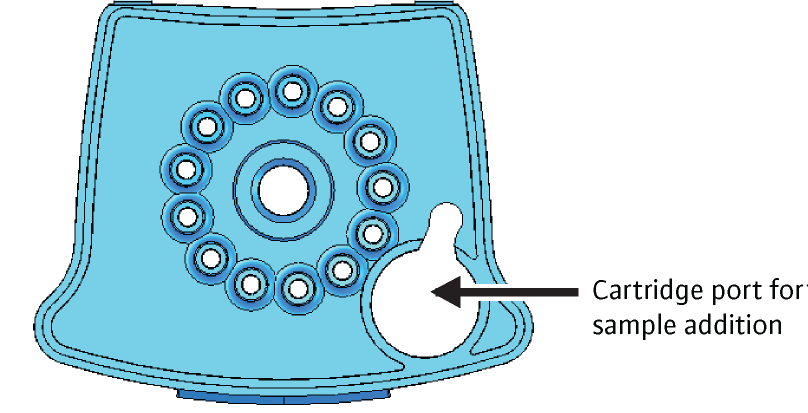
- Use the sterile transfer pipette provided in the CBNAAT kit to draw liquefied sample into the transfer pipette
- The minimum amount to be loaded into the cartridge is 2 ml
- Do not process the sample if there is insufficient volume
- Open the cartridge lid
- Transfer the sample into the open port (Figure 1) of the CBNAAT cartridge and dispense slowly to minimize the risk of aerosol formation