Biosafety in TB Laboratory: Factors to be Considered in TB Laboratory Risk Assessment
Staff working with patient specimens and live mycobacterial cultures must operate under appropriate biosafety conditions with adequate infection control measures in place, including staff health checks.
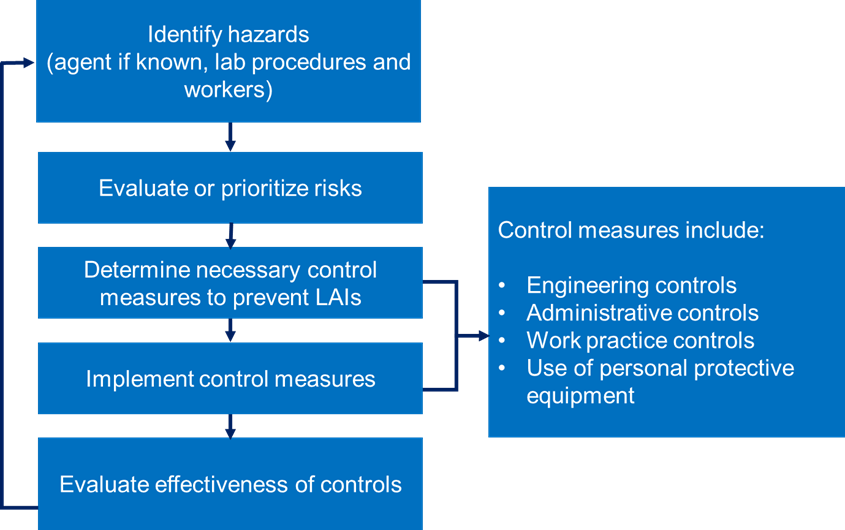
For the safety of personnel working in TB labs, the following factors must be considered to avoid the risk associated with pathogenicity and transmission of TB bacilli.
The 4 key factors that must be considered during risk assessment of TB labs include: