LPA Troubleshooting for No Signal
ContentThere could be overall weak or no signals on the strip including or except for the conjugate control zone.
Weak or no signal including conjugate control (CC) zone could be due to:
- Too low room temperature
- Reagents not equilibrated to room temperature (20-22°C)
- Hybridization solutions not pre-warmed
- Too high or too low incubation temperature
- No or too little amount of CON-C and/or SUB-C used
Solution: Repeat reverse hybridization.

Figure: No Signal including CC Zone
Points to Note
- A higher incubation temperature may prevent the binding of the amplicons to the probes.
- A higher temperature during the stringent wash step may result in weak intensities of the banding patterns.
- The temperature of the hybridizer should be monitored if such problems are seen.
- If the problem persists and the thermostat on the hybridizer gives an appropriate reading of 45ºC, and a digital thermometer reads a different temperature that is above or below 45ºC; the problem is likely with the machine’s internal temperature probe and should be reported to the manufacturer and corrected.
Do’s and Don'ts to Avoid Weak or No Signals
- Perform LPA at the appropriate room temperature.
- Bring reagents to room temperature before use (20-22°C).
- The green and red hybridization solutions must be pre-warmed to 45ºC.
- Follow instructions to add the exact volume of reagents.
- The central heating/cooling thermostat must be appropriately adjusted.
- Do not use pipettes that are not calibrated.
- Do not miss any step in the hybridization process.
Resources
- GenoType MTBDR plus ver 2.0 Kit, Instructions for Use.
- GenoType MTBDRsl VER 2.0 Kit, Instructions for Use.
- GLI Training Package on LPA.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Troubleshooting for No Signal Except Conjugate Control [CC]
ContentThere could be overall weak or no signals on the strip, including or except, the Conjugate Control (CC) zone.

Figure: LPA showing No Signal Except CC
Reasons and Troubleshooting for No Signal Except CC are given in the table below:
Table: Reasons and Troubleshooting for No Signal Except CC
REASON FOR ERROR
SOLUTION
The quality of extracted DNA does not allow efficient amplification
Repeat the extraction step
Amplification Mixes (AM-A and AM-B) were either not mixed properly or were added in the wrong amounts
Prepare a new master mix and repeat the test
The incubation temperature was too high
Repeat reverse hybridization
The extracted bacterial species cannot be detected by the Universal Control
Use an alternative identification method Resources
- GenoType MTBDR plus ver 2.0 kit, Instructions for Use.
- GenoType MTBDRsl VER 2.0 kit, Instructions for Use.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Troubleshooting for Uneven Staining
ContentDuring hybridization, single-stranded amplicons bind to probes and highly specific binding is ensured to produce even staining in the presence of stringent buffer and temperature conditions. However, uneven staining can occur due to the following reasons:
- Strips not completely immersed in reagents during incubation steps.
- Tray not shaken properly.
- Denatured amplicons were not properly mixed with the hybridization buffer before adding the strip to the well.
- The reusable tray had not been properly cleaned so residual amplicons remained.
Solution: Repeat reverse hybridization step.

Figure: Uneven Staining of the Strip
Points to Note
- The strips must be checked to ensure that they are fully immersed after the addition of each reagent. If necessary, a pipette tip can be placed on top of a strip to keep it submerged. Care must be taken so that buffer from a well with a problematic strip does not spill into any of the other wells.
- Before adding the strip to the well, the purple solution containing the denatured amplicons should be completely diffused into the green hybridization buffer.
- The instruments should be checked to ensure that the Twincubator is shaking at 300 rpm and the GT-Blot 48 is oscillating properly.
- The trays must be thoroughly washed and rinsed after each use.
Do’s and Don’ts
- Gently shake the tray until the solution has homogenous colour.
- Do not spill solution into the neighboring wells.
- Tray must be dipped into the water to at least 1/3rd of its height.
- Use washed and clean trays.
Resources
- GenoType MTBDR plus ver 2.0 Kit, Instructions for Use.
- GenoType MTBDRsl VER 2.0 Kit, Instructions for Use.
- GLI Training Package on LPA.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Troubleshooting for High Background Colour
ContentDuring hybridization, single-stranded amplicons bind to probes and highly specific binding is ensured to produce even staining in the presence of stringent buffer and temperature conditions. However, high/ dark backgrounds in strips may be due to the following reasons:
- CON-C and/or SUB-C used too concentrated
- Washing steps not performed with necessary care
- Wash solutions were too cold
Solution: Repeat reverse hybridization step.

Figure: Dark background in the LPA Strip
Points to Note
- The rinse step should be done carefully to remove excess conjugate buffer followed by a wash with deionized water to remove salts in the rinse buffer.
- Post-substrate development, wash with deionized water is essential to stop colorimetric reaction and to remove excess substrate buffer.
- Reagents should always be warmed before use.
Resources
- GenoType MTBDR plus ver 2.0 Kit, Instructions for Use.
- GenoType MTBDRsl VER 2.0 Kit, Instructions for Use.
- GLI Training Package on LPA.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Troubleshooting for Other Possible Reasons
ContentIssue #1:
Absence of Conjugate Control (CC) and Amplification Control (AC) in a negative control (Figure 1).

Figure 1: Absence of CC and AC in a Negative Control
Table 1 below lists the reasons and troubleshooting for the absence of CC and AC in a negative control.
Table 1: Reasons and troubleshooting for the absence of CC and AC in a Negative Control
REASONS
TROUBLESHOOTING
Mistakes during setup
The test is invalid – Repeat test
Performance of amplification reaction
Presence of amplification inhibitors
Issue 2:
Absence of locus control for a specific drug, with presence of CC and M. tuberculosis complex (TUB) band (Figure 2).

Figure 2: Absence of locus control for a specific drug, with presence of CC and TUB band
Table 2 below lists the reasons and troubleshooting for the absence of locus control for a specific drug, with the presence of CC and TUB band.
Table 2: Reasons and troubleshooting for the absence of locus control for a specific drug, with the presence of CC and TUB band
REASONS
TROUBLESHOOTING
Mutation or deletions in locus control region
- The test is indeterminant - Repeat test
- Sequencing may be requested to identify the specific mutation
Complete or partial deletion of a target gene
Resources
- GenoType MTBDR plus ver 2.0 kit, Instructions for Use.
- GenoType MTBDRsl VER 2.0 kit, Instructions for Use.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Quality Assurance [QA] Including Quality Control
ContentThe quality concepts of the Line Probe Assay (LPA) laboratory include:
- Infrastructure
- Dedicated areas for different steps of the procedure.
- Colour code/ label for different rooms:
- Red for pre-amplification area
- Green for DNA extraction area
- Blue for amplification area
- Yellow for post-amplification area.
- Unidirectional workflow for reduced risk of contamination. Under no circumstances should:
- Anything from the specimen preparation area be taken into the reagent preparation area
- Anything from amplification and hybridization/detection areas be taken into specimen or reagent preparation areas.
- Safety in the laboratory.
- Proper functioning instruments.
- Manpower
- Trained and competent staff.
- Maintained laboratory turn-around-time (sample receipt to reporting results).
- Documentation and Records: There is a need to have documents/records on the following:
- Standard Operating Procedures (SOPs)
- Laboratory Information Management System (LIMS): Worksheet, recording and reporting results
- Reagent manuals: Manufacturers’ instructions and specifications manuals
- Daily usage logs, corrective action logs, calibration, repair and maintenance records for instruments
- Safety aspects.
Quality Assurance including Quality Control (QC) includes:
Reagent QC: The following should be done:
- Testing of all lots of reagents
- Integrity testing upon receipt (e.g., Tag polymerase).
- Stock rotation
- Storage at the appropriate temperature
- Supply chain and inventory management.
- Expiry prevention
Equipment QC: This includes:
- Calibration
- Maintenance
- Validation
- Corrective actions for QC failures
- Knowledge to operate
- Service and repairs
- Manuals.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Infrastructure
LPA Procedural Quality Control
ContentA batch of samples is defined as a group of samples that are processed, amplified and hybridized:
- At the same time
- Under the same conditions
- Using the same PCR master mix, thermal cycler and hybridization solutions
- On the same hybridization platform.
QC during the DNA extraction procedure
- Use two sets of 1.5 ml tubes are foreach specimen.
- Mark and label the tubes according to the organization of the batch on the worksheet.
- The first tube in every batch should be labelled with a coloured sticker.
- Check the functionality of the instruments prior to processing.
- Use calibrated pipettes.
QC during the preparation of the master mix
Use homogenous reagents and thaw them properly to ensure even distribution of contents.
- Use calibrated pipettes.
- Set functional refrigerator/freezer at -20ºC.
- Verify addition of DNA polymerase visually.
- Document lot numbers and expiration dates of reagents.
- Test quality control specimens with a new lot of DNA polymerase.
QC during DNA template addition
- Select thermal cycler program based on the type of specimen to be amplified.
- Check that the caps of PCR tubes do not contain any liquid when loaded into the thermal cycler (to prevent dilution of the reaction mixture).
- Check the PCR reaction tubes for any bubbles (prevent uneven temperature distribution).
QC during the hybridization and detection procedure
- Maintain consistency in the numbering process. For example, strip marked number 1 must correspond with the first specimen extracted, amplified and added to denaturing buffer in hybridization well.
- Use dedicated pair of forceps to handle unhybridized strips.
- Avoid strips sticking together into the same well.
- If an automated hybridiser is used for less than 48 specimens, the rest of the wells should be filled with distilled water to ensure an even temperature across the heating platform.
- TwinCubator (12 strip capacity) should be used with one or two hybridization runs depending on the number of specimens.
- Use a clean tray in the TwinCubator.
Use of positive and negative controls
To demonstrate competency for the line probe assay (LPA), positive and negative quality control (QC) samples must be performed on a routine basis, for each batch of specimens as shown in Figure 1. The overview of the use of positive and negative quality control is shown in Figure 2.
Each batch of specimens tested with the LPA must have an “extraction positive control” (ATCC strain H37Rv), an “extraction negative control”, and a “PCR master mix negative control” (Figure 1).

Figure 1: The H37Rv extraction positive control shown in strip number 9 (circled in blue) and the extraction negative control is shown in strip number 10 (circled in red); Source: LPA Laboratory Manual, FIND
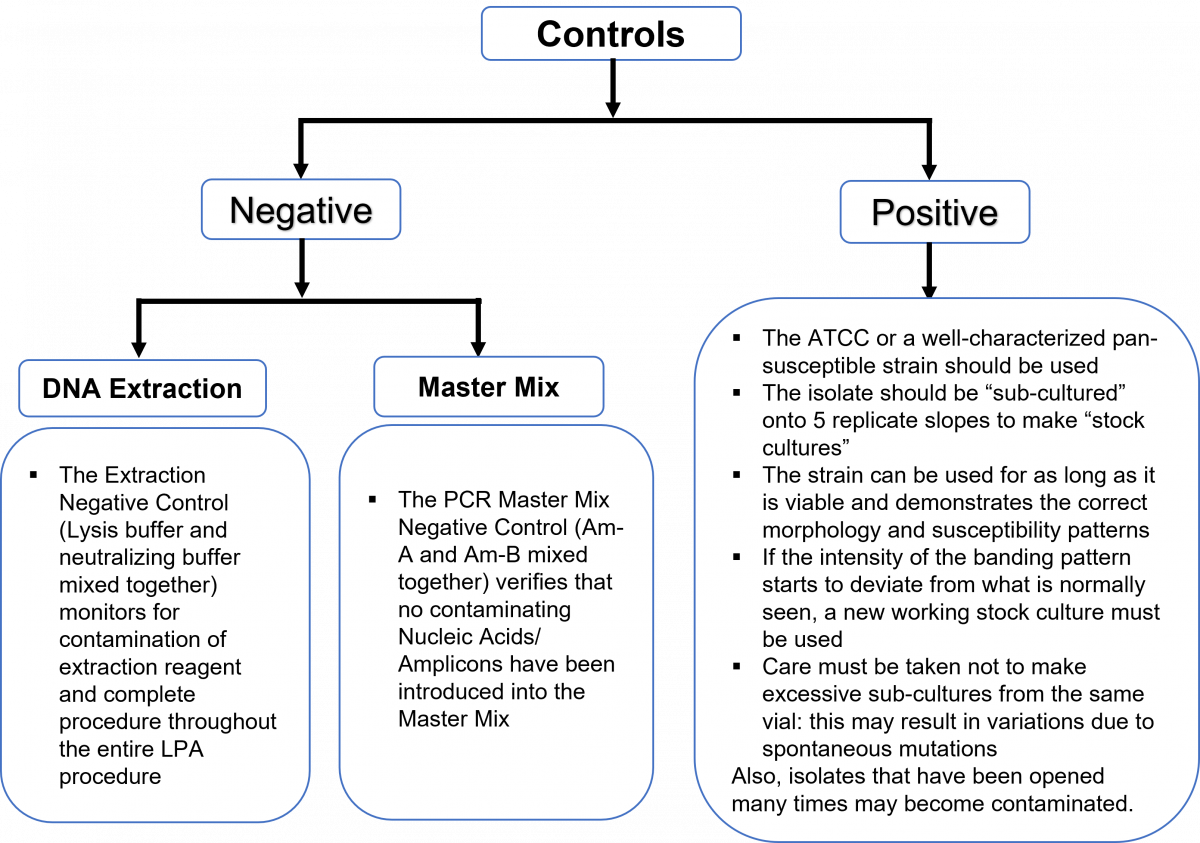
Figure 2: Overview of positive and negative controls
Amplification Control (AC) on the LPA strip functions as both:
The internal “PCR Positive Control”
Amplification control (AC) band indicates that the DNA extraction and PCR procedures were carried out successfully (Figure 3).
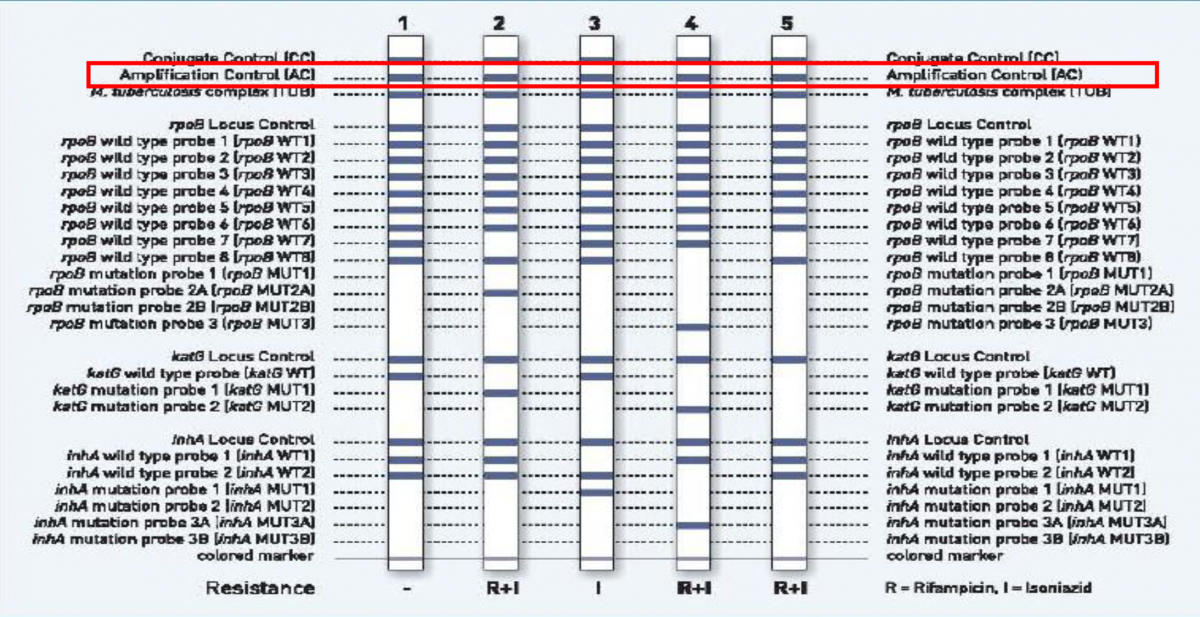
Figure 3: Amplification Control (highlighted in red) as seen on the Genotype MTBDRplus test strip; Source: LPA Laboratory Manual, FIND.
The “Inhibition Positive Control”
- The AC band will not appear if there are PCR inhibitors in the extracted material.
- The strips for the two negative controls must be positive only at the Conjugate Control (CC) and AC bands.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Quality Assurance: Proficiency Testing
ContentSteps Involved in Proficiency Testing (PT):
- Each new laboratory undertakes line probe assay (LPA) testing for 50 smear-positive TB affected patients.
- The specimen is anonymized (stripped of name and any personal identifiers), assigned a number, and processed by N-acetyl L-cysteine- Sodium Hydroxide (NALC-NaOH) method.
- Processed sputum deposit, DNA extracts and PCR products are stored at -20oC.
- High resolution scanned images of the ‘line probe result form’ and the line probe assay result strips (scotch-taped onto separate LPA – run form) are sent to the Central TB Division/National Reference Laboratory (NRL).
- 20 DNA extracts are randomly selected by the NRL for testing concordance.
- The extracts are sent by express courier to the NRL.
- Blinded LPA testing on the 20 DNA extracts is done at NRL.
- Once the pilot and proficiency phase has been satisfactorily completed, the LPA laboratory is assessed for proficiency, based on the following indicators:
- The proportion of invalid LPA results; PT benchmark: less than 10%
- Contamination of negative controls; PT benchmark: Clean in all runs
- Internal concordance: concordance of results between 1st and 2nd tested parts for each specimen; PT benchmark: should be >95%
- External concordance: concordance of results of randomly selected specimens with the reference site; PT benchmark: should be >95%.
Steps involved in Annual Proficiency Testing:
- Every NRL sends 30 cultures to the LPA laboratory.
- Cultures are taken for LPA procedures. DNA extraction is followed by:
- Master mix preparation, amplification and hybridisation.
- The results are then shared with the respective NRL, who asses the results based on the following indicators:
- The proportion of invalid LPA results; PT benchmark: less than 10%
- Contamination of negative controls; PT benchmark: Clean in all runs
- External concordance; PT benchmark: should be >95%.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Each new laboratory undertakes line probe assay (LPA) testing for 50 smear-positive TB affected patients.
LPA Quality Assurance: Cleaning of Lab
ContentGeneral Instructions
- Dedicated cleaning supplies, brooms required for different areas.
- Cleaning should be done by trained Polymerase Chain Reaction (PCR) laboratory staff, not by general cleaning personnel.
- Meticulous cleaning of the work area, surfaces, racks, pipettes and instruments should be done before and after performing each procedure with disinfectants (freshly prepared 1% sodium hypochlorite, followed by 70% alcohol).
- Use dedicated spray flasks/ containers for disinfectants.
- Clean paper towels (not cloth) should be used to decontaminate surfaces and equipment.
- Walls should have high-gloss enamel/ gloss-based paint for easy cleaning.
- Floors must be covered with vinyl, linoleum or ceramic tiles.
- Bench-tops must be made of impermeable melamine to eliminate crevices.
- Instruments should be cleaned and maintained daily/ weekly as described in the equipment training content.
- Cleaning and maintenance log sheets of all areas should be maintained.
Area-wise Cleaning
Weekly meticulous cleaning once; clean top-to-bottom, starting with bench tops and finishing with mopping the floor with disinfectants.
Area 1: DNA extraction room
Clean and maintain instruments (pipettes, biosafety cabinet, centrifuge and water bath) daily/ weekly.
Area 2: Pre-amplification room
Clean and maintain instruments (pipettes, PCR equipment) daily/ weekly.
Area 3: Amplification room
Clean and maintain instruments (thermal cycler, pipettes, PCR equipment) daily/ weekly.
Area 4: Hybridization room
Clean and maintain instruments (GT-Blot, Twincubator, pipettes, GT-Blot hybridization machine) daily/ weekly.
Resources
- Line Probe Assays for Drug-resistant Tuberculosis Detection, GLI.
- Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay, FIND.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen