-
CDST_LT: Truenat as a rapid molecular diagnostic test
FullscreenTruenat
ContentTruenat is an indigenous rapid molecular test platform that is currently under use in NTEP for diagnosis of TB and Rif Resistance. It is a platform utilising real-time Polymerase Chain Reaction (PCR) technology built into micro-PCR chips.
Testing on Truenat involves three components:
- Workstation (consisting of 2 devices)
- Trueprep AUTO Universal Cartridge-based Sample Prep Device for the automated extraction and purification of DNA
- Truelab Real-time micro PCR Analyzer for performing real-time PCR. It is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports.
- Cartridge and Chip
- Reagent kits (Sample Pre-treatment and Prep kits)

Figure: Truenat Source: MolBio Products.
Test results for MTB detection and Rif Resistance has a turn around time of 1-2 hours. Depending on the micro-PCR chips used various tests can be performed using Truenat. Truenat MTB micro-PCR chips detect Mycobacterium tuberculosis bacteria for TB diagnosis. Truenat MTB RIF micro-PCR chip is used as a reflex test to detect resistance to Rifampicin (RIF), the first-line drug for TB treatment
Truenat has many advantages. Truenat is designed to be mobile and is battery operated (~8 hours on full charge). It can be deployed in peripheral laboratories and microscopy centres with minimal or no added facilities and hence it is more point-of-care. Biosafety requirements are similar to smear microscopy. However, it is multi staged and partially automated, requiring the presence of a Lab Technician through out the test.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin-resistance, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Truenat is used in NTEP for: MTB detection Rif Resistance Detection INH resistance Detection MTB and Rif Resistance Detection 4 Truenat is used for MTB and Rif Resistance detection in NTEP Yes Yes The Truelab Analyzer is available in how many chip ports?
2 (Duo)
1 (Uno), 2 (Duo) and 4 (Quattro)
1 (Uno)
4 (Quattro)
2
The Truelab Analyzer is available as 1 (Uno), 2 (Duo) and 4 (Quattro) chip ports.
Yes Yes - Workstation (consisting of 2 devices)
Truenat Cartridge and Inside Demonstration
ContentTruenat cartridges are components of Trueprep AUTO v2 Universal Cartridge Based Sample Prep Kit. As shown in the figure below, each cartridge is:
- Used with Trueprep Device for DNA extraction and purification
- Disposable
- Single-use
- Preloaded with Internal Positive Control.
Truenat cartridges are used with Trueprep devices for DNA extraction and purification. The components of the cartridge are illustrated in the following figure and include:
- Sample Chamber - Processed samples (pretreated with lysis buffer) are added to the sample chamber of the cartridge for processing on the Trueprep device.
- Matrix Chamber - DNA released by chemical and thermal lysis cells binds to the proprietary matrix here.
- Elute Chamber - Trapped DNA is washed with buffers to remove Polymerase Chain Reaction (PCR) inhibitors and is eluted from the matrix using the elution buffer. The purified DNA is collected from here.
- Waste generated is contained within the dump region inside the cartridge.

Figure: Inside of a TrueNAT Cartridge; Source: Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance
Resources
Consumables Required at a Truenat Lab
ContentConsumables required for Truenat test provided by the manufacturer include:
Trueprep AUTO MTB Sample Pre-treatment Pack for sample processing
- Liquefaction buffer
- Lysis buffer
- Disposable transfer pipette (graduated) - 1ml

Trueprep AUTO v2 Universal Cartridge Based Sample Prep Kit for DNA extraction
- The Reagent Pack contains the following reagents
- Wash Buffer A
- Wash Buffer B
- Elution Buffer
- Priming Waste
- The Cartridge Pack contains the following
- Cartridge
- Elute collection tube
- Elute collection tube label
- Disposable transfer pipette
- Disposable Transfer Pipettes (graduated) - 3 ml
- Reagent Reset Card

Truenat MTB Chip-based Real-Time PCR test for Mycobacterium tuberculosis
- Truenat MTB micro-PCR chip
- Microtube with freeze-dried PCR reagents
- DNase & RNase free pipette tip

Truenat MTB-RIF Dx Chip-based Real-Time PCR Test for Rifampicin Resistant Mycobacterium tuberculosis
- Truenat MTB-RIF Dx micro-PCR chip
- Microtube with freeze-dried PCR reagents
- DNase & RNase free pipette tip

Other Consumables
- Gloves
- Masks
- Sodium hypochlorite
Resources
- MolBio Diagnostics Product Details
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance
Kindly provide your valuable feedback on the page to the link provided HERE
Overview of the Truenat Testing Process
ContentThe Truenat Assay Technology works on Real-time Polymerase Chain Reaction (PCR).
The key steps include:
- Collection of specimens from a presumptive TB/known TB Patient (for UDST).
- Liquefaction and lysis of specimen using the Trueprep AUTO MTB sample pre-treatment pack (20 minutes)
- Extraction and purification of DNA using Trueprep AUTO v2 Universal Cartridge-based sample prep kit and Trueprep AUTO v2 Universal Cartridge-based sample prep device (20 minutes)
- Amplification of extracted DNA by the Truelab Real-time micro-PCR analyzer using freeze dry PCR reagent in microtubes on a Truenat MTB chip (35 minutes)
- DNA from positive test results is tested using Truenat MTB RIF Dx chip as a reflex test (55 minutes)
The process flow from sample to result for Truenat Assay Technology is described in the figure below.


Figure: Process flow from sample to result for Truenat Assay Technology; Source: Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
Resources
- Truenat MTB Kit Insert.
- Trueprep® AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
Truenat MTB and RIF Assay as a TB Diagnostic Test
ContentThe tests are performed using Trueprep AUTO Sample Pre-treatment and Prep kits and Truenat micro-PCR chips.
To detect M. tuberculosis, the Truenat MTB chip amplifies a portion of the ribonucleoside-diphosphate reductase gene, nrdB with a Limit of Detection (LOD) of about 100 Colony Forming Units (CFU)/ml sputum sample.
DNA extraction and detection of M. tuberculosis takes approximately one hour.
When M. tuberculosis is detected, as a follow-on test, a small volume of the already extracted DNA is used to test for resistance to Rif using Truenat MTB Rif Dx chip.
Mutations associated with Rif-resistance within the RRDR region of rpoB gene are detected using a probe melt assay and read using the Truelab micro-PCR Analyzer.
The detection of Rif-resistance takes an additional one hour.
Truenat Testing Capacity
- The Truelab Analyzer is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports (figure below).
- The different ports can be used independently to test multiple samples.
- In an eight-hour daily work shift, the estimated throughput of Truelab Analyzer Uno, Duo or Quattro can be 7-9, 15-18, 30-36 specimens, respectively.
Results Reporting
A Truelab micro PCR printer (figure below) is used to print test results. It can also be connected via SIM card/ Wi-Fi/ Bluetooth to transmit results.

Figure: Different Equipments used in the Truenat Workstation
Resources
- Truenat MTB Pack Insert.
- Truenat MTB Rif Dx Pack Insert.
- Truelab Manual.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin-resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
Inbuilt Controls used in TrueNAT
ContentTruenat tests use Internal Quality Control to assess test validity using Internal Positive Control (IPC).
- The cartridge contains pre-loaded IPC.
- IPC is part of the full process control.
- IPC undergoes all processing steps along with specimen from extraction to amplification.
- IPC assesses the validity of the test run from sample processing to result and is illustrated in following figures (Figure 1(A) and Figure 1(B)).

Figure 1(A): Amplification of IPC in Valid Test; Source: MolBio Diagnostics Pvt. Ltd.
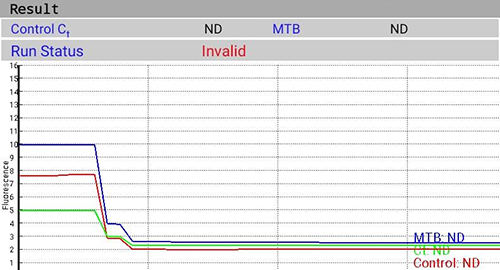
Figure 1(B): Invalid Test with Failure of IPC Amplification; Source: MolBio Diagnostics Pvt. Ltd.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge Based Sample Prep Device.
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Specimen processing for truenat
FullscreenProcessing of Sputum Sample before Truenat Testing
ContentThe sputum sample needs to be processed to liquefy and lyse the specimen before Truenat testing. This is because of the following reasons:
- Sputum samples contain Polymerase Chain Reaction (PCR) inhibitors but PCR amplification requires pure DNA from sputum samples.
- The sputum sample should be homogenized and pipettable before DNA extraction can begin.
- Pre-treatment also decontaminates specimen for storage/ transportation/ extraction.
The Trueprep AUTO MTB Sample pre-treatment pack is used for processing the specimen. It digests sputum, releases bacteria, and removes PCR inhibitors.
Steps for specimen pre-treatment include:
- Open a Trueprep AUTO MTB sample pre-treatment kit which contains a graduated 1 ml transfer pipette, lysis buffer bottle and liquefaction buffer bottle.
- The sample and all reagents including the liquefaction buffer, lysis buffer should be at room temperature before starting the steps of pre-treatment.
- Switch on the Trueprep AUTO v2 sample prep device.
- Label lysis buffer bottle with corresponding sample number and date of extraction.
- Add 2 drops of liquefaction buffer to the sputum sample, swirl mix, incubate for 10 minutes at room temperature. If the sample is not pipettable after 10 minutes, incubate for another 5 minutes and swirl at 2 minutes intervals.
- Transfer 0.5 ml of the liquefied sputum sample from the sample container to the corresponding lysis buffer bottle using the 1 ml graduated transfer pipette provided.
- Dispose of the used transfer pipette into the container filled with freshly prepared 1% sodium hypochlorite.
- Add 2 drops of liquefaction buffer into the lysis buffer bottle; swirl gently to mix and incubate the lysis buffer bottle at room temperature for 3-5 minutes.
- Ensure that the sample has completely liquefied.
- Sputum may be stored in lysis buffer for up to 1 week at 30°C with no degradation of DNA.
Video fileResources
- Trueprep AUTO MTB Sample Pre-treatment Pack.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
TrueNAT Sample Processing: Other Body Fluids
ContentThe extra-pulmonary samples (Non-sputum) can also be processed and used for MTB and Rif diagnosis using Truenat. The processing of various extra-pulmonary samples is described below.
Reagents required:
Trueprep AUTO MTB Sample Pre-treatment Pack for sample processing
- Reagents stable for two years (2-40°C) or one month (temperatures up to 45°C)
- Avoid exposure to light or elevated temperatures
- Do not freeze
Steps in processing of non-sputum samples
Non-sputum samples like tissue biopsy should not be processed at the Designated Microscopy Centre (DMC), but should be transferred to a higher centre.
General Instructions
- Disinfect work surfaces with freshly prepared 1% bleach, followed by 70% alcohol.
- Process fresh specimens immediately or store frozen at -20oC. Avoid more than three freeze thaws.
- Bring frozen samples and refrigerated reagents to room temperature (20-30°C).
- Open the Trueprep AUTO MTB sample pre-treatment pack that contains:
- Liquefaction buffer bottle
- Lysis buffer bottle
- Graduated 1 ml transfer pipette.
- Processing is to be done as instructed for each sample type.
- Non-sputum sample is stable for 3 days at up to 40°C and 1 week at 30°C in the lysis buffer.
A. Processing for Bronchoalveolar Lavage (BAL), Pleural fluid, Peritoneal fluid
Equipment: Centrifuge and centrifuge tubes (10-15 ml volume)
- Add 5-10 ml specimen in a centrifuge tube.
- Centrifuge at 4000x for 5 minutes to concentrate the sample.
- Discard the supernatant until 500 μl remains.
- Add two drops of the liquefaction buffer to the concentrated sample (500 μl).
- Transfer the contents to a labelled lysis buffer tube.
- Incubate for five minutes and proceed for DNA extraction.
B. Processing for Pus, Abscess, Lymph node aspirate, Cerebrospinal Fluid (CSF)
Equipment: Centrifuge tubes (1.5 ml volume)
- Add 0.5 ml specimen in a 1.5 ml tube.
- Add two drops of liquefaction buffer.
- Transfer the contents to a labelled lysis buffer tube.
- Incubate for 5 minutes and proceed for DNA extraction.
C. Processing for Tissue/ Biopsy Samples
Equipment: Mortar and pestle (for tissue homogenization)
- Homogenize sample by using 100 μl lysis buffer in mortar and pestle.
- Add two drops of liquefaction buffer.
- Transfer the contents to a labelled lysis buffer tube.
- Incubate for five minutes.
- Use only clear fluid for DNA extraction.
The processed samples will be used for DNA extraction and PCR amplification for diagnosis of MTB and Rif resistance.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
DNA Extraction in TrueNAT MTB and TrueNAT MTB/RIF DX Assay
ContentEquipment and Consumables
- Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device
- Trueprep AUTO v2 Universal Cartridge Based Sample Prep Kit
Steps in DNA extraction (Molbio Trueprep Auto Universal Cartridge Based Sample Prep Device)
1. Wear gloves, mask.
2. Insert the plug-in connector to attach the reagent pack to the Trueprep device (Figure 1).

Figure 1: Trueprep Cartridge Based Sample Prep Kit attached to the Trueprep Auto Device using the Plug-in Connector; Source: Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin Resistance
3. Disinfect the work surfaces with the freshly prepared 1% bleach, followed by 70% alcohol.
4. Open the cartridge pouch, take out the cartridge.
5. Label the cartridge (Patient ID, Date), place it on the cartridge stand.
6. Open the black cap of the cartridge sample chamber.
7. Using a pipette transfer 3 ml contents of the lysis buffer bottle (processing step) in the sample chamber.
8. Discard the pipette and lysis buffer bottle (1% sodium hypochlorite).
9. Re-cap the sample chamber.
10. Press the “Power” button to switch on the Trueprep device.
11. Press “Eject” to eject the cartridge holder (Figure 2), pull out gently.

Figure 2: Top view of the Trueprep AUTO Universal Cartridge-based Sample Prep Device; Source: Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin Resistance
12. Insert the cartridge in the cartridge holder (sample chamber to the right when seen from the front).
13. Push to close the cartridge holder (the click sound confirms the correct cartridge loading).
14. Press the “Start” button (Figure 2).
15. DNA extraction begins, reagents from bottles are automatically added to the cartridge (pre-programmed) (Figure 2).
16. On completion (18-20 min), the cartridge holder automatically ejects.
17. Remove the cartridge, place it on the cartridge stand.
18. Label the ECT tube (Elute Collection Tube) with Patient ID & Date.
19. Pierce the covering of elute compartment in the cartridge with a filter barrier pipette tip, aspirate elute (DNA) into the ECT tube.
20. Discard the used pipette tips, used cartridge (1% sodium hypochlorite).
21. DNA can be used immediately for amplification/ stored in fridge (4°C for up to 24 hours) or -20°C (for up to 1 year).
Video fileResources
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
- Operation of Molbio Trueprep AUTO Universal Sample Prep Device.
- Molbio Trueprep Auto Universal Cartridge-based Sample Prep Device.
Kindly provide your valuable feedback on the page to the link provided HERE
PCR Amplification of the Target genes in Truenat
ContentTruenat Mycobacterium tuberculosis (MTB) and Truenat MTB Rifampicin (Rif) Dx Assays amplify target genes through Polymerase Chain Reaction (PCR).
The presence of mutant gene is detected using probe melt assay.
The target genes include:
- Truenat MTB - ribonucleoside-diphosphate reductase gene nrdZ (the precursor for DNA synthesis)
- Truenat MTB Rif Dx assay – Rifampicin Resistance Determining Region (RRDR) region of the RNA polymerase Beta (rpoB) gene for resistance to Rif (between codon positions 509 and 533)

Figure: Image showing RRDR of the rpoB gene of M. tuberculosis; the target gene of Truenat MTB Rif Dx assay includes codons 509-533
Resources
- Truenat MTB Pack Insert.
- Truenat MTB Rif Pack Insert.
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Testing using Truenat
FullscreenUnloading the Truenat Cartridge
ContentVideo fileUsing Truenat autoanalyzer for MTB testing
ContentVideo fileLoading the elute onto the Truenat MTB Chip
ContentVideo fileAmplification and Detection of MTB and MTB-Rif
ContentProcedure
Equipment and reagents
- Truelab Uno/ Duo/ Quattro Real Time Quantitative micro–PCR analyzer
- Truenat MTB micro-PCR kit
- Truenat MTB Rif micro-PCR kit
- Truepet fixed volume (6 μl) precision micropipette
Amplification and detection of Mycobacterium tuberculosis (MTB)
- Wear gloves, laboratory coats, mask.
- Clean working surfaces with 1% Sodium hypochlorite, followed by 70% alcohol.
- Clean instruments with a paper towel dipped in 70% alcohol.
- Switch on the Truelab device by pressing the red button for two seconds.
- Select test bay.
- Select test profile: “MTB”, enter Details (Referred By, Patient ID, Gender, Patient Name, Age).
- Select the sample type.
- Press “Start Reaction". For Truelab Uno Dx, Press the eject button to open the chip tray. For Truelab Duo/Quattro, the chip tray opens automatically on tapping the “Start Reaction” .
- Take out “Truenat MTB chip” from the chip sleeve (check desiccant colour. If blue -chip can be used; discard chip if desiccant is pink/ white which indicates moisture is absorbed).
- Place the chip on the tray by aligning the registration holes with tray pins without touching the white reaction well. (the white reaction well should face upward).
- Open the microtube containing the freeze-dried PCR reagent (white colour) and place it on the microtube stand.
- Pipette 6 μl DNA from the Extraction Chamber (After DNA extraction and purification step) into the microtube.
- Wait 30-60 seconds for the DNA elute and PCR reagent to mix (clear solution obtained).
- Use the same tip to pipette 6 μl treated DNA from the microtube and load on the white reaction well of the chip.
- Discard the microtube and microtip (1% sodium hypochlorite).
- Start the test run.
- The test completes in 35 minutes, press ‘’RESULT’’ to view the result screen.
- Possible results:
- MTB Detected/ Not Detected/ Errors/ Invalid
- If MTB detected, test the same DNA eluate for Rif resistance using MTB-Rif chip. Select the MTB-Rif assay.
- If the result is Invalid/ Error, repeat amplification using the same extracted DNA and a new “Truenat MTB chip”. If Invalid again, repeat the test with a fresh sample.
- Tap “Open/ Close Tray” button to eject the chip tray.
- Lift the chip and discard (1% sodium hypochlorite).
Amplification and Detection of MTB Rif
- DNA from the MTB positive elutes is tested for Rif-resistance using the MTB-Rif chip.
- Select the test profile “MTB Rif”.
- Select the sample type.
- Press “Start Reaction". For Truelab Uno Dx, Press the eject button to open the chip tray. For Truelab Duo/Quattro, the chip tray opens automatically on tapping the “Start Reaction”.
- Take out “Truenat MTB Rif Dx chip” from the chip sleeve (check desiccant colour. If blue -chip can be used; discard chip if desiccant is pink/ white which indicates moisture is absorbed).
- Follow steps 10-16 as described in Amplification and detection of MTB (above).
- The test completes in 55 minutes, press ‘’RESULT’’ to view the result screen.
- Possible results:
- MTB Rif resistance detected/ MTB Rif resistance not detected/ Error/ Indeterminate.
- If result is Indeterminate/ Error, repeat the amplification using the same extracted DNA and new “Truenat MTB Rif Dx chip”. If Indeterminate/ Error again, repeat the test with a fresh sample.
- Tap “Open/ Close Tray” button to eject the chip tray.
- Lift the chip and discard (1% sodium hypochlorite).
The algorithm for Truenat MTB Assay (Figure) describes interpretation of test results obtained for Truenat MTB and Truenat MTB Rif Dx assays.
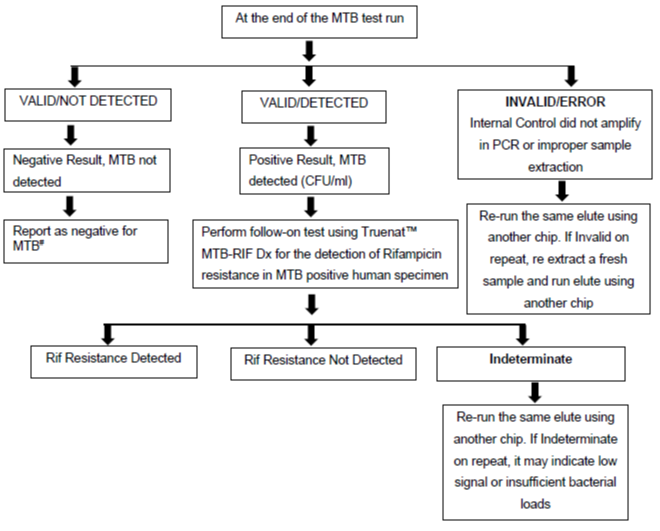
Figure: Truenat MTB Assay Results Interpretation: Algorithm; Source: Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin Resistance
# Test is Valid as Internal Positive Control amplified
Video fileResources
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
- Truenat MTB Pack Insert.
- Truenat MTB Rif Pack Insert.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Truenat result interpretation and recording
FullscreenTruenat MTB Assay Results Interpretation: Visualization
ContentTruenat MTB Assay results are visualized:
- During testing
- Completion of test
Test Status Screen (Figure 1)
Results visualization during testing include:
- “Green” Test Progress Bar helps to monitor the test progress and the current cycle number.
- “Yellow” Cycle Progress Bar indicates the progress of the current cycle.
- Test Details Bar indicates patient details, sample type, system health, battery level, current cycle temperature.
- “Red” Test Completion Indicator turns green when the test completes.
- PLOT button displays optical (fluorescence measure) and thermal graphs (temperature during PCR) on real time basis.

Figure 1: Test Status Screen; Source: TrueLab Manual
Test Result Screen (Figure 2(A)-(E))
On tapping the "Result" button, results visualization on the completion of test includes:
- Input details display; Patient details - Name, ID, Age, Sex, Referred by
- Chip details display; Type of Chip - Truenat MTB/ Truenat MTB Rif
- Test results of Truenat MTB:
- DETECTED
- NOT DETECTED
- Error
- Test results Truenat MTB-Rif Dx:
- Rif Resistance Detected
- Rif Resistance Not Detected
- Indeterminate
- Error
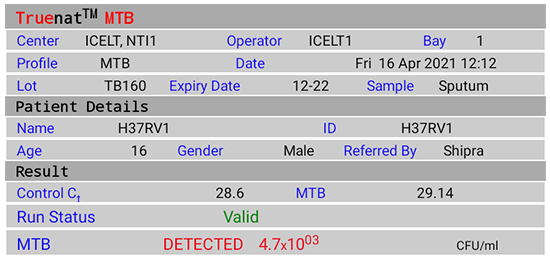
Figure 2(A): Truenat MTB Test Result Screen; DETECTED; Source: MolBio Diagnostics Pvt. Ltd.
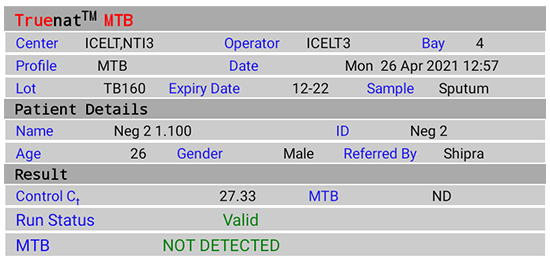
Figure 2(B): Truenat MTB Test Result Screen; NOT DETECTED; Source: MolBio Diagnostics Pvt. Ltd.

Figure 2(C): Truenat MTB Rif Test Result Screen; Rif Resistance Detected; Source: MolBio Diagnostics Pvt. Ltd.
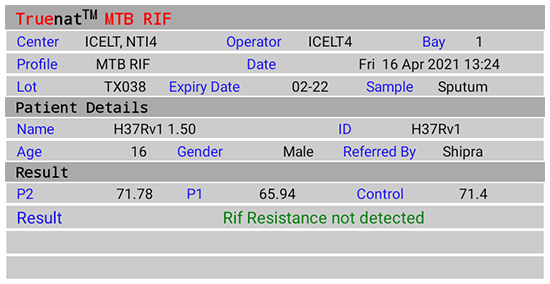
Figure 2(D): Truenat MTB Rif Test Result Screen; Rif Resistance Not Detected; Source: MolBio Diagnostics Pvt. Ltd.

Figure 2(E): Truenat MTB Rif Test Result Screen; Indeterminate; Source: MolBio Diagnostics Pvt. Ltd.
Video fileResources
Kindly provide your valuable feedback on the page to the link provided HERE
TrueNAT MTB Assay Results Interpretation: Detection of Mycobacterium tuberculosis and RIF Resistance
ContentOn completion of test run, the Test Result Screen displays the results for:
- Detection of Mycobacterium tuberculosis (Truenat MTB)
- “DETECTED” for Positive result, Cycle Threshold (Ct) value, Colony Forming Units per milliliter (CFU/ ml)
- “NOT DETECTED” for Negative result
- “Valid”/ “Invalid” for test validity based on amplification of Internal Positive Control (IPC)
- “Error” for test failure
- Detection of Rif Resistance (Truenat MTB-Rif Dx)
- “Rif Resistance Detected” if mutations to Rif are detected
- “Rif Resistance Not Detected” if mutations to Rif are not detected
- “Indeterminate” when test did not determine resistance to Rif
- “Error” for test failure
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Detection of Mycobacterium tuberculosis (Truenat MTB)
Retrieving test results from the Truenat machine
ContentVideo fileRecording Truenat Results
ContentVideo fileResults Entry in Lab Register for NAAT
ContentThe results for Nucleic Acid Amplification Test (NAAT) assays are entered in Culture and Drug Susceptibility Testing (C&DST) register. The key variables entered are shown in the table below.
Table: Key Variables entered in C&DST Register; Source: Guidelines for PMDT in India, 2021. VARIABLES SET 1 VARIABLES SET 2 VARIABLES SET 3 VARIABLES SET 4 Test ID Health Facility (HF) name Residential district Current facility HF type Date of test updated in Nikshay Lab type Type of test Predominant symptom Date tested Patient ID Reason for testing Predominant symptom duration Date reported Episode ID Treatment status History of Anti-TB Treatment (ATT) Test status Name Diagnosis date No. of Health Care Provider (HCP) visited before the diagnosis of the current episode Type of specimen Gender TB treatment start date The visual appearance of sputum Date of specimen collection Age Current facility state State name Primary phone Current facility district District name Address Current facility TB Unit (TU) TB unit Residential state Current facility HF NAAT results are reported in the results section of “Request Form for examination of biological specimen for TB” including:
- Select Type of test: Cartridge-based Nucleic Acid Amplification Test (CBNAAT)/ TrueNAT
- Select Sample: A/B
- Select M. tuberculosis: Detected/ Not Detected/ Not Available (NA)
- Select Rif Resistance: Detected/ Not Detected/ Indeterminate/ NA
- Select Test: No result/ Invalid/ Error; Error code
- Date tested
- Date Reported
- Reported by (name and signature)
- Laboratory name
Video fileVideo : CBNAAT/Truenat Results Entry in Lab Register
Resources
Reset Procedure for Truenat Testing
ContentAfter completion of the set number of nucleic acid extractions (i.e., buffer count 05/ 25/ 50/ 100 completed) the Trueprep AUTO v2 device will prompt the alert on its 2- line LCD screen to “Change the reagent pack” and “Insert Reagent Reset Card & Start” on the screen alternatively. When this happens, the new reagent pack needs to be inserted into the device and the buffer count needs to be reset.
ImageFlowchart: Procedure for Reset in Truenat Testing
Image
Figure: 1) The Trueprep AUTO v2 Back Panel, with tubing and caps for plugging in reagent bottles 2) The Trueprep AUTO v2 device Back Panel with Reagent Pack 3) Reagent Reset Card; Source:Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device User Manual.
Resources
- Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device User Manual.
- Trueprep AUTO v2 Pack Insert Version 04.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test The buffer count in the Trueprep Auto V2 device will be set to what after the reset procedure is completed? 0 1 10 100 1 The buffer count will be reset to ‘0’ once the reset procedure is completed. Yes Yes
-
CDST_LT: Troubleshooting in truenat
FullscreenInterfering Substances causing errors in NAAT Assay
ContentIt is common in laboratories to see specimens with the following particles which may potentially alter nucleic acid amplification test (NAAT) results:
- Food particles
- Blood
- Tobacco
- Pan
- Debris or tissue pieces
These interfering substances cause failures in Truenat assays and may have inhibitory effects on CBNAAT assays. This interferes with the accuracy of results, leading to false positive or negative tests and delayed cycle threshold values.
Hence, it is important to follow certain precautions when collecting the specimen to avoid contaminating the specimen with these substances.
In such situations, the following protocol needs to be followed:
- Reject frank blood/bloody samples; however, the presence of blood up until 30% does not interfere in Truenat assays
- For samples containing food/tobacco/tissue particles that cause interference in processing:
- Use a fresh sample, if available
- Use a sample after food particles or tissue pieces settle/sediment; otherwise, reject the sample
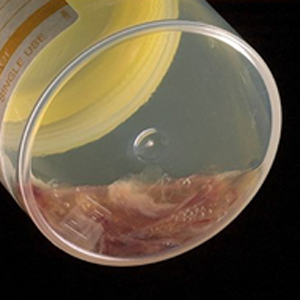
Figure: Blood in Sputum Sample Interferes with PCR
Resources
Monitoring Different Types of Errors with Truenat
ContentMonitoring of Truenat errors
There are various types of errors that occur with the Truenat machines, which include:
- Coded errors related to the Trueprep AUTO v2 sample prep device
- Coded errors related to the Truelab micro-PCR Analyzer
- Non-coded system errors/failures
Monitoring the Trueprep AUTO v2 errors
-
The Truenat system is a closed amplification system (i.e., the amplified product is sealed in the chip), and an enzyme system is incorporated in the reaction mix to prevent previously amplified material from getting re-amplified.
- It is recommended that testing sites perform negative control tests using Trueprep AUTO lysis buffer reagent and sterile Phosphate Buffered Saline (PBS) monthly or when contamination is suspected (e.g., an unusually high proportion of specimens with ‘M.tb detected’).
- Swab testing of work surfaces and both the Truelab and Trueprep machines should be conducted monthly.
- Used and expired cartridges should be discarded as per the protocols, so they are not re-used.
Monitoring the Truelab errors
-
To ensure that the Truelab micro Polymerase Chain Reaction (PCR) Analyzer is working accurately, the manufacturer recommends running positive and negative controls (which can be purchased as part of the Truenat™ Positive Control Kit- Panel I) periodically.
- The positive and negative controls can also be used for lot-to-lot verification and assessment of reagents if the temperature of storage areas falls outside the recommended ranges.
- An optional “Plot” view on the digital interface is also available while the process is being performed that allows for monitoring test progress in real-time.
- Truelab Analyzers can be configured to send data on device performance to the manufacturer’s (default configuration) or local servers to allow the manufacturer (without sharing any patient data) and/or the National TB Elimination Programme to monitor instrument performance on a real-time basis.
- This helps in the identification and possible prevention of instrument malfunctions or breakdowns, detection of user errors and retraining needs, and monitoring of instrument and test utilization across fleets of instruments.
Resources
- Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India;CTD, MoHFW, India,2021.
- Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device; User Manual V4.
- TRUELAB UNO Dx Real Time Quantitative micro PCR Analyzer; manual VER 04.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Identify if the statement is True or False
Running positive and negative controls periodically is recommended to ensure that the Truelab micro PCR Analyzer is working accurately.
True False 1 Running positive and negative controls periodically is recommended by the manufacturer to ensure that the Truelab micro PCR Analyzer is working accurately Yes Yes Truelab Analyzers can be configured to send data on device performance to the manufacturer’s (default configuration) or local servers to allow the manufacturer (without sharing any patient data) and/or the National TB Elimination Programme to monitor instrument performance on a real-time basis.
True False 1 This helps in the identification and possible prevention of instrument malfunctions or breakdowns, detection of user errors and retraining needs, and monitoring of instrument and test utilization across fleets of instruments. Yes
Yes
Trueprep Error Classes and actions
ContentTrueprep Error Classes
The Trueprep errors are classified as:
-
Cartridge Errors
-
Reagent Pack Errors
-
Device Errors and Warnings
ImageFigure 1: Classification of Trueprep errors
Trueprep Error Actions
I Cartridge Error Actions
ImageFigure 2: Actions for Trueprep cartridge errors
II Reagent Error Actions
ImageFigure 3: Actions for Trueprep reagent errors
III Device Error and Warning Actions
ImageFigure 4: Actions for Trueprep device errors and warnings
Resource
-
Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, 2021, CTD, MoHFW, India.
-
Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device; User Manual V4.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
In ‘E11 – RTD L Error’, what does L stand for?
Liquid
Lower
Light
Lysis
4
In E11 - RTD L Error (The code L is for error in the Lysis Heater)
Yes
Yes
-
Truelab Error Classes and actions
ContentHere we discuss the error classes and actions in Truelab.
Truelab Errors - Classes and Actions
Error Class
Error Name
Reason for the error
Error Actions
Error 1
Thermal Cycling Error
Occurs when the test chip is faulty and the thermal cycling does not happen.
Re-run the same elute using another chip. If the error repeats, process the sample again and re-run the elute using another chip. If the problem still persists, contact the Molbio support team.
Error 2
Test Stopped Manually
Occurs when the user has manually stopped the ongoing test and the analyser does not have sufficient run time to compute data.
Error 3
Incorrect Optical Profile
Occurs when there is a deviation in the expected optical profile due to a reduction in reaction volume in the chip during the course of the reaction.
Error 4
Runtime Error
Occurs when run data capture/ analysis is incomplete.
Error 5
Probe Check Error
Occurs in the event of a low initial signal due to insufficient mastermix dispensed onto the chip.
Invalid
Whenever Truelab displays the result as 'INVALID', it means that the internal control did not amplify in Polymerase Chain Reaction (PCR) or sample extraction was not proper.
Resources
-
Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device, User Manual Version 4.
-
Truelab UNO Dx Real-time Quantitative Micro PCR Analyzer Manual Version 4.
-
Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, 2021, CTD, MoHFW, India.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
When does thermal cycling error occur in Truelab's real-time micro PCR analyzer?
Occurs when the test chip is faulty and the thermal cycling does not happen.
Occurs when run data capture/ analysis is incomplete.
Occurs in the event of low initial signal.
Occurs when the user has manually stopped the ongoing test.
1
Thermal cycling error occurs when the test chip is faulty and the thermal cycling does not happen.
Yes
Yes
When does the Runtime Error occur in Truelab's real-time micro PCR analyzer? Occurs when the test chip is faulty and the thermal cycling does not happen.
Occurs when run data capture/ analysis is incomplete.
Occurs in the event of low initial signal.
Occurs when the user has manually stopped the ongoing test.
2 Runtime error occurs when run data capture/ analysis is incomplete. Yes
Yes
-
Troubleshooting Failures Without Error Codes
ContentThe Truenat is a rapid molecular diagnostic method based on the Nucleic Acid Amplification Testing (NAAT) principle. Truenat uses portable, battery-operated devices to rapidly detect Mycobacterium tuberculosis Complex Bacteria (MTBC) and Rifampicin resistance.
Truenat system involves two main devices:
- Trueprep® AUTO v2 Universal Cartridge based Sample Prep Device: Used for the automated extraction and purification of DeoxyriboNucleic Acid (DNA).
- Truelab® Real Time micro-PCR Analyzer: Used for performing Real-Time Polymerase Chain Reaction (RT-PCR), resulting in the semi-quantitative detection of MTBC.
The errors in Truenat are broadly categorised as:
- Errors with codes (Trueprep and Truelab errors)
- Errors without codes (System failures)
The errors without codes in Truenat and the ways to resolve them are listed in the table below:
Sl. No. System failures Reason Troubleshoot 1 ‘Unable to read chip information’ Analyzer was unable to read chip memory Check if the chip was loaded properly into the tray. Remove the chip and re-select the profile from Status Screen and repeat the steps. If the message reappears, load a new chip and re-load the elute again 2 ‘Could not initialize. Please try again’ The system was unable to establish an internal connection Attempt the test again by using a new chip and re-loading the elute again 3 “Chip is already used” OR “Chip loaded is expired” User loaded a used chip/expired chip in the tray Use a fresh chip and re-load the elute 4 Login attempt failure Login failed due to incorrect password entered Re-enter the correct password 5 Invalid patient name in sample details screen The patient’s name in the sample details section is blank Fill patient’s name in the sample details section Resource
-
Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India; CTD, MoHFW, India. 2021.
-
Trueprep AUTO v2 Universal Cartridge Based Sample Prep Device; User Manual V4.
-
TRUELAB UNO Dx Real Time Quantitative micro PCR Analyzer; manual VER 04.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What should you do when you get a system failure alert as:
“ Chip is already used” ?
Re-insert the same chip
Insert a fresh chip
Do not insert any chip
None of the above
2
When you get a system failure alert as: “ Chip is already used” , Use a fresh chip and re-load the elute.
Yes
Yes
What should you do when you get a system failure alert as:
"Unable to read chip information"
Re-insert the same chip
Insert a fresh chip
Do not insert any chip
Check if chip is loaded properly, remove the chip and reselect the profile from status screen and repeat the steps.
4 When you get a system failure alert as "Unable to read chip information" Check if the chip was loaded properly into the tray. Remove the chip and re-select the profile from Status Screen and repeat the steps. If the message reappears, load a new chip and re-load the elute again Yes
Yes
-
CDST_LT: Truenat instrument maintenance and record keeping
FullscreenMaintenance of the Truenat Instrument - General Principles
ContentLike any other laboratory instrument, the TrueNAT machine needs a periodic maintenance.
Maintenance of Truelab and Trueprep instruments includes:
- Cleaning the instrument
- Disinfecting the surfaces
- Cleaning the spillages
- Discarding the used consumables
- Calibrations
- Replacement of parts
- Flush protocol
- Data backup
- Troubleshooting errors, alerts, warning messages
Frequency of maintenance:
The following schedule is required to maintain TrueNAT instrument(s):
- Daily, monthly maintenance by lab personnel
- As and when required (need based) by lab personnel/ manufacturer
- Annual maintenance by the manufacturer.
Posters on maintenance provided by manufacturer are shown in Figure 1(A)- (B)

Figure 1(A): Considerations (Do’s and Don’ts) for use of Trueprep Device; Source: MolBio Diagnostics Pvt. Ltd.

Figure 1(B): Considerations (Do’s and Don’ts) for use of Truelab Device; Source: MolBio Diagnostics Pvt. Ltd.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- Trueprep Manual.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Ensuring Compliance of Truenat Instrument Installation Criteria
ContentCompliance to equipment installation criteria is necessary to/for:
- Establish accurate and reliable results as claimed by the manufacturer
- To assure standard/specific requirements are met
- Provide proof that the process will consistently produce predetermined outcome
- Equipment safety
- Improve overall performance of the equipment
- Decrease work interruptions due to equipment failure
- Lower repair costs
- Cover equipment warranty and free maintenance
Instrument Qualification Documents include:
- Installation Qualification- IQ
- Operational Qualification- OQ
- Performance Qualification- PQ
Installation Qualification
IQ provides evidence for delivery, installation and configuration of the instrument as per manufacturer’s standards using an installation checklist.
Operational Qualification
OQ is a collection of test cases used to verify the proper functioning of a system before the instrument is released for use.
Performance Qualification
PQ is a collection of test cases used to verify that the system performs as expected under simulated real-world conditions.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Daily, Weekly and Monthly Maintenance of Truenat Instrument
ContentDaily maintenance of Truelab and Trueprep instruments performed by the laboratory personnel includes:
- Maintaining a dust-free environment by wiping the exterior of instruments with a dry, lint-free cloth
- Cleaning the surface of the Trueprep device once with 70% ethanol
- Cleaning the spill or leak with a cloth or tissue-paper dipped in disinfectant (1% sodium hypochlorite solution, followed by 70% ethanol)
- Not spilling water or any other solution on the surface of instruments
- Discarding used cartridges, chips, reagent bottles and other consumables in 1% sodium hypochlorite solution, soaking for minimum 30 minutes, and disposing as per the biomedical waste management guidelines.
Monthly maintenance of the Truelab Analyzer performed by laboratory personnel includes:
- Cleaning the surface of the Truelab Analyzer with 70% ethanol
- Cleaning the Truelab bays
- Calibrating temperature.
Annual/ need-based maintenance of the Truenat instrument performed by the laboratory personnel/ manufacturer includes:
- Flush protocol for the Trueprep device when the device is left idle (not used) for 10 days and/ or errors relating to extraction process occur
- Replacement of spillage tray or linear motion guide tray for the Trueprep device in case of sample spillage on trays during extraction or when cross-contamination is suspected
- Replacement of slider glass for Truelab Analyzer after 200 tests and/ or when related errors occur
- Temperature calibration for Truelab Analyzer when error related to temperature occur and/ or when the temperature curve is abnormal (shows blips)
- Calibration of micropipettes (every 6 months/ biannual).
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- Trueprep Manual.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
NAAT Instrument Monthly Data Archive and Data Back-up
ContentSecurity of laboratory data and confidentiality of TB patient data are important. Hence, it is recommended to archive or take periodic data backup.
The Truelab Analyzer has an internal memory to store 20000 test results. The digital data can be transmitted through connectivity via email, SIM card, Wi-Fi or Bluetooth.
Recommended ways of documenting the results and archiving data are:
- Records should be maintained in the National TB Elimination Program (NTEP) recording and reporting formats.
- Entries should be made in Nikshay online platform of NTEP.
- Monthly/ periodically data should be exported in CSV format and saved on external devices-desktop/ laptop/ hard drive.
- Data records from Nucleic Acid Amplification Testing (NAAT) instruments should be kept in a secure area.
- Data access should be given to authorized personnel only.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Annual Maintenance of the Truenat Instrument
ContentNeed-based maintenance for Truenat instrument is done by lab personnel/ manufacturer while the manufacturer does annual maintenance.
The tasks included under annual maintenance are:
- Flush protocol for Trueprep device when the device is left idle (not used) for 10 days/ annually and/or errors relating to extraction process occur.
- Spillage tray or linear motion guide tray replacement for Trueprep device in case of sample spillage on trays during extraction or when cross-contamination is suspected/ done annually.
- Slider glass replacement for Truelab Analyzer after 200 tests/ annually and/or when related errors occur.
- Temperature calibration for Truelab Analyzer when error related to temperature occur and/or when temperature curve is abnormal (shows blips)/ annually.
- Annual calibration of micropipettes.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- Trueprep Manual.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Monitoring Truenat quality and lab performance
FullscreenExternal Quality Assurance of TrueNAT
ContentExternal Quality Assurance (EQA) ensures quality of Truenat laboratory is maintained by comparing their results through retesting and panel testing by a higher level laboratory (Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL)).
EQA for Truenat laboratory is done by:
- Onsite supervision: Visits conducted at regular intervals by the IRL/ NRL/ Central TB Division (CTD)
- Proficiency Testing or Panel Testing (PT): Inter-laboratory comparisons conducted by IRL/ NRL to assess the performance of the laboratory supervised by them; re-checking of the standard panel provided by the IRL/ NRL.
- Random Blinded Re-checking (RBRC): Re-examination of randomly selected samples by the IRL/ NRL.
Plan for Truenat EQA
- Truenat EQA visits need to be conducted annually.
- Panel is provided by the National Tuberculosis Institute (NTI), Bangalore, to the participating sites.
- Participating site to conduct panel testing and report results to NTI as per timelines.
- Feedback/ corrective action provided by NTI to participating sites.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Monitoring Quality Indicators of Truenat
ContentMonitoring of key quality indicators or performance indicators of Truenat is important to assess the functioning of the instrument, calibration/ service requirements of the instrument, competency of the technician performing the assays and inventory management. Overall quality indicators should assess:
- Total tests done
- Type of samples tested
- Test failure
- Availability of consumables
- Turnaround time
- Specimen rejected
- External Quality Assurance (EQA) results
- Instrument downtime
Quality performance indicators should be reviewed weekly by personnel in charge of Truenat laboratory, and monthly by the Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL)/ Central TB Division (CTD).
Under the National Tb Elimination Programme (NTEP) the following quality indicators are to be reported monthly:
- Total number of tests performed using Truenat (including EP-TB and private sector)
- Total presumptive TB tested
- Total number of presumptive DR TB tested
- Total number of Rif indeterminate, invalid, error results
- Total number of re-tests done and results
- Total number of samples sent for First Line-Line Probe Assay (FL-LPA) and Second Line-Line Probe Assay (SL-LPA)
- Total number of chips (MTB, MTB Rif) in stock
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
-
CDST_LT: Biomedical waste management in Truenat
FullscreenBiosafety measures required for Truenat
ContentGeneral biosafety requirements for Truenat laboratory include:
- Using gloves, laboratory coat, mask when handling specimens
- Minimizing aerosol generation during sample processing and handling specimens carefully
- Working in a well-ventilated room
- Using laboratory work instructions, job aids, Standard Operating Procedures (SOPs) and checklists, from sample collection to reporting results, to avoid mishaps
- Maintaining a spill management kit
- Following manufacturer instructions to operate the instruments
Precautions during processing the specimen
- Handle all specimens as potentially infectious.
- Avoid cross-contamination.
- Nozzle of the liquefaction buffer bottle should not touch the sample container.
- Avoid aerosol formation during the transfer of the liquefied sample to the lysis buffer bottle.
- Use the transfer pipettes provided with the pre-treatment pack.
- Dispose the used transfer pipettes (1% sodium hypochlorite).
- Clean the spills with 1% sodium hypochlorite, followed by 70% alcohol.
Precautions during cartridge loading and transportation
- Keep the cartridges on the provided holder while loading the specimen.
- Load the cartridges onto the machine with care.
- Avoid turning the cartridge during transportation to prevent specimen spill.
- If a spill occurs, soak tray in 1% sodium hypochlorite solution for 30 minutes and dispose. Spray the cartridge holder with 70% alcohol and load a new tray in the cartridge holder after 5 minutes.
- Pipette carefully to avoid cross-contamination between reagents and/ or samples.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- Tuberculosis Laboratory Biosafety Manual.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Disposal of Infectious Samples and Used Cartridges in TrueNAT Laboratories
ContentDisposal of infectious samples and used cartridges in Truenat laboratories is an essential activity which needs to be performed to keep everyone safe and prevent TB infection in healthcare workers.
- Biomedical Waste (BMW) management guidelines need to be followed-up to dispose infectious samples and used Truenat consumables.
- Solutions and/ or solid waste containing biological samples should be disinfected before discarding.
- Lab personnel should soak material to be disposed in freshly prepared 1% sodium hypochlorite for 30 minutes and discard.
- Used Truenat chips, microtube, microtube cap, transfer pipette, pipette tips, reagent bottles need to be submerged in freshly prepared 1% sodium hypochlorite solution for at least 30 minutes before disposal as per biomedical waste disposal guidelines.
- Consumables submerged in sodium hypochlorite should NOT be autoclaved,
- Cartridge pouches, chip pouches, transfer pipette wrappers, desiccant pouches, sleeves needs to be discarded as general waste.
Resources
- Guidelines for PMDT in India, 2021.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- Tuberculosis Laboratory Biosafety Manual.
- MolBio Diagnostics Pvt. Ltd.
- Central Pollution Control Board: Guidelnes for Hazardous and Other Wastes.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen