Truenat
ContentTruenat is an indigenous rapid molecular test platform that is currently under use in NTEP for diagnosis of TB and Rif Resistance. It is a platform utilising real-time Polymerase Chain Reaction (PCR) technology built into micro-PCR chips.
Testing on Truenat involves three components:
- Workstation (consisting of 2 devices)
- Trueprep AUTO Universal Cartridge-based Sample Prep Device for the automated extraction and purification of DNA
- Truelab Real-time micro PCR Analyzer for performing real-time PCR. It is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports.
- Cartridge and Chip
- Reagent kits (Sample Pre-treatment and Prep kits)

Figure: Truenat Source: MolBio Products.
Test results for MTB detection and Rif Resistance has a turn around time of 1-2 hours. Depending on the micro-PCR chips used various tests can be performed using Truenat. Truenat MTB micro-PCR chips detect Mycobacterium tuberculosis bacteria for TB diagnosis. Truenat MTB RIF micro-PCR chip is used as a reflex test to detect resistance to Rifampicin (RIF), the first-line drug for TB treatment
Truenat has many advantages. Truenat is designed to be mobile and is battery operated (~8 hours on full charge). It can be deployed in peripheral laboratories and microscopy centres with minimal or no added facilities and hence it is more point-of-care. Biosafety requirements are similar to smear microscopy. However, it is multi staged and partially automated, requiring the presence of a Lab Technician through out the test.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat Tests for the Detection of TB and Rifampicin-resistance, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Truenat is used in NTEP for: MTB detection Rif Resistance Detection INH resistance Detection MTB and Rif Resistance Detection 4 Truenat is used for MTB and Rif Resistance detection in NTEP Yes Yes The Truelab Analyzer is available in how many chip ports?
2 (Duo)
1 (Uno), 2 (Duo) and 4 (Quattro)
1 (Uno)
4 (Quattro)
2
The Truelab Analyzer is available as 1 (Uno), 2 (Duo) and 4 (Quattro) chip ports.
Yes Yes - Workstation (consisting of 2 devices)
Truenat Cartridge and Inside Demonstration
ContentTruenat cartridges are components of Trueprep AUTO v2 Universal Cartridge Based Sample Prep Kit. As shown in the figure below, each cartridge is:
- Used with Trueprep Device for DNA extraction and purification
- Disposable
- Single-use
- Preloaded with Internal Positive Control.
Truenat cartridges are used with Trueprep devices for DNA extraction and purification. The components of the cartridge are illustrated in the following figure and include:
- Sample Chamber - Processed samples (pretreated with lysis buffer) are added to the sample chamber of the cartridge for processing on the Trueprep device.
- Matrix Chamber - DNA released by chemical and thermal lysis cells binds to the proprietary matrix here.
- Elute Chamber - Trapped DNA is washed with buffers to remove Polymerase Chain Reaction (PCR) inhibitors and is eluted from the matrix using the elution buffer. The purified DNA is collected from here.
- Waste generated is contained within the dump region inside the cartridge.

Figure: Inside of a TrueNAT Cartridge; Source: Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance
Resources
Consumables Required at a Truenat Lab
ContentConsumables required for Truenat test provided by the manufacturer include:
Trueprep AUTO MTB Sample Pre-treatment Pack for sample processing
- Liquefaction buffer
- Lysis buffer
- Disposable transfer pipette (graduated) - 1ml

Trueprep AUTO v2 Universal Cartridge Based Sample Prep Kit for DNA extraction
- The Reagent Pack contains the following reagents
- Wash Buffer A
- Wash Buffer B
- Elution Buffer
- Priming Waste
- The Cartridge Pack contains the following
- Cartridge
- Elute collection tube
- Elute collection tube label
- Disposable transfer pipette
- Disposable Transfer Pipettes (graduated) - 3 ml
- Reagent Reset Card

Truenat MTB Chip-based Real-Time PCR test for Mycobacterium tuberculosis
- Truenat MTB micro-PCR chip
- Microtube with freeze-dried PCR reagents
- DNase & RNase free pipette tip

Truenat MTB-RIF Dx Chip-based Real-Time PCR Test for Rifampicin Resistant Mycobacterium tuberculosis
- Truenat MTB-RIF Dx micro-PCR chip
- Microtube with freeze-dried PCR reagents
- DNase & RNase free pipette tip

Other Consumables
- Gloves
- Masks
- Sodium hypochlorite
Resources
- MolBio Diagnostics Product Details
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance
Kindly provide your valuable feedback on the page to the link provided HERE
Overview of the Truenat Testing Process
ContentThe Truenat Assay Technology works on Real-time Polymerase Chain Reaction (PCR).
The key steps include:
- Collection of specimens from a presumptive TB/known TB Patient (for UDST).
- Liquefaction and lysis of specimen using the Trueprep AUTO MTB sample pre-treatment pack (20 minutes)
- Extraction and purification of DNA using Trueprep AUTO v2 Universal Cartridge-based sample prep kit and Trueprep AUTO v2 Universal Cartridge-based sample prep device (20 minutes)
- Amplification of extracted DNA by the Truelab Real-time micro-PCR analyzer using freeze dry PCR reagent in microtubes on a Truenat MTB chip (35 minutes)
- DNA from positive test results is tested using Truenat MTB RIF Dx chip as a reflex test (55 minutes)
The process flow from sample to result for Truenat Assay Technology is described in the figure below.


Figure: Process flow from sample to result for Truenat Assay Technology; Source: Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
Resources
- Truenat MTB Kit Insert.
- Trueprep® AUTO Universal Cartridge-based Sample Prep Device.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
Truenat MTB and RIF Assay as a TB Diagnostic Test
ContentThe tests are performed using Trueprep AUTO Sample Pre-treatment and Prep kits and Truenat micro-PCR chips.
To detect M. tuberculosis, the Truenat MTB chip amplifies a portion of the ribonucleoside-diphosphate reductase gene, nrdB with a Limit of Detection (LOD) of about 100 Colony Forming Units (CFU)/ml sputum sample.
DNA extraction and detection of M. tuberculosis takes approximately one hour.
When M. tuberculosis is detected, as a follow-on test, a small volume of the already extracted DNA is used to test for resistance to Rif using Truenat MTB Rif Dx chip.
Mutations associated with Rif-resistance within the RRDR region of rpoB gene are detected using a probe melt assay and read using the Truelab micro-PCR Analyzer.
The detection of Rif-resistance takes an additional one hour.
Truenat Testing Capacity
- The Truelab Analyzer is available as 1 (Uno), 2 (Duo) or 4 (Quattro) chip ports (figure below).
- The different ports can be used independently to test multiple samples.
- In an eight-hour daily work shift, the estimated throughput of Truelab Analyzer Uno, Duo or Quattro can be 7-9, 15-18, 30-36 specimens, respectively.
Results Reporting
A Truelab micro PCR printer (figure below) is used to print test results. It can also be connected via SIM card/ Wi-Fi/ Bluetooth to transmit results.

Figure: Different Equipments used in the Truenat Workstation
Resources
- Truenat MTB Pack Insert.
- Truenat MTB Rif Dx Pack Insert.
- Truelab Manual.
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin-resistance.
Kindly provide your valuable feedback on the page to the link provided HERE
Inbuilt Controls used in TrueNAT
ContentTruenat tests use Internal Quality Control to assess test validity using Internal Positive Control (IPC).
- The cartridge contains pre-loaded IPC.
- IPC is part of the full process control.
- IPC undergoes all processing steps along with specimen from extraction to amplification.
- IPC assesses the validity of the test run from sample processing to result and is illustrated in following figures (Figure 1(A) and Figure 1(B)).

Figure 1(A): Amplification of IPC in Valid Test; Source: MolBio Diagnostics Pvt. Ltd.
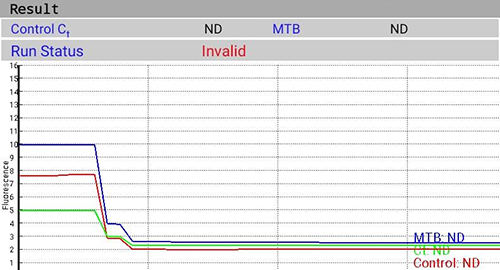
Figure 1(B): Invalid Test with Failure of IPC Amplification; Source: MolBio Diagnostics Pvt. Ltd.
Resources
- Truenat MTB Kit Insert.
- Trueprep AUTO Universal Cartridge Based Sample Prep Device.
- Practical Guide to Implementation of TrueNAT Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen