Specimen Processing for TB Cultures
ContentSputum specimens are viscous materials contaminated with normal flora. Therefore, processing involves pre-treatment of the sputum specimens via:
- Digestion: To free the TB bacilli from the mucus in which they may be embedded.
- Decontamination: To eradicate normal flora that grows more rapidly than TB bacilli, and would interfere with the ability to recover TB bacilli.
- Homogenization: Of the digested materials.
- Concentration: Of the TB bacilli by centrifugation before smear preparation and media inoculation.
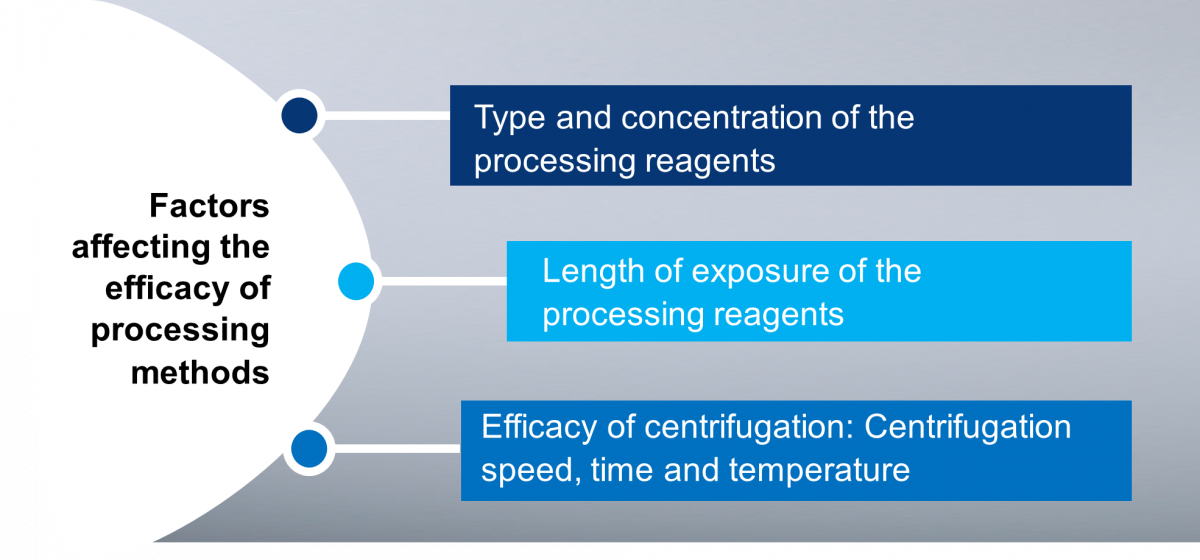
Figure: Factors Affecting the Efficacy of Processing Methods
Methods of Culture Specimen Processing
Various processing methods are used for TB specimens; amongst them, the most common methods are:
- N-acetyl-L-cysteine - sodium hydroxide (NALC-NaOH) method: It is the mildest decontamination method which can kill about 33% of mycobacteria in a clinical specimen. It can be used with both liquid and solid media.
- Petroff’s sodium hydroxide method: It is a harsher method – it can kill up to 70% of mycobacteria in specimens. Although useful with highly contaminated specimens, it is not recommended for use with liquid MGIT media.
Resources
- MGIT Procedure Manual, Mycobacteria Growth Indicator Tube (MGIT) Culture and Drug Susceptibility Demonstration Projects, FIND Training Manual.
- GLI Training Module on Specimen Processing, STOP TB Partnership.
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Pulmonary specimens
ContentThese are the steps to be followed when processing pulmonary specimens in TB culture laboratories:
Beginning the Specimen Processing Procedure:
- Process only one specimen at a time, and do not leave open containers or open centrifuge tubes in the Bio Safety Cabinet (BSC).
- Process the available specimen in a 50 ml sterile, plastic, screw-capped centrifuge tube (Figure).
Figure: Capped 50 ml sterile, plastic, screw-capped centrifuge tubes
NALC - NaOH Procedure:
- Always open the cap of the specimen container slowly to minimize aerosol production.
- Aliquot reagent in a separate tube for each specimen to avoid contamination of reagent stocks. A freshly prepared single-use aliquot is preferred.
- Note the volume of the specimen. Add an equal volume of N-acetyl-L-cysteine-sodium hydroxide (NALC-NaOH) solution and tighten the cap.
- In order to avoid cross-contamination, do not allow the NALC-NAOH solution container to touch the specimen tubes.
- After the addition of the decontaminant and or digestant tighten the caps and vortex for not more than 20 seconds at a moderate speed.
- Invert each tube 5 times, making the figure 8 with your wrist, to ensure that the NALC-NaOH solution contacts the entire inner surface of the tube and cap.
- Avoid extreme agitation or shaking which can cause inactivation of the NALC.
- Let the tubes stand at room temperature for 20-25°C, for 15 minutes and mix by gently inverting the tube.
- NaOH exposure time must be strictly limited to 15 minutes to prevent the over-killing of the TB bacilli.
- If stronger decontamination is needed, the starting concentration of NaOH may be increased to 5-6%, but the time of exposure should not be extended.
- Neutralize the specimen with a phosphate buffer of pH 6.8 to the 45 ml mark. Do not exceed the 45 ml mark.
- To avoid cross-contamination do not allow the diluent (phosphate buffer) container to touch the mouth of the specimen tubes.
- Single-use aliquots of phosphate buffer or a dispenser are preferred to avoid cross-contamination during the procedure.
- After centrifugation, open the safety bucket in the BSC and carefully pour off the supernatant into a splash-proof discard container with a suitable disinfectant (5% phenol).
- If required, swab the tube with a disinfectant-soaked gauze (use individual pieces) and recap carefully.
- While wiping, do not allow the disinfectant to flow into the tube.
- Re-suspend the sediment in 1–2 ml of sterile phosphate buffer (pH 6.8) using a new transfer pipette.
Please click here to see a full video on sputum specimen processing for culture.
Resources
- MGIT Procedure Manual, Mycobacteria Growth Indicator Tube (MGIT) Culture and Drug Susceptibility Demonstration Projects, FIND Training Manual.
- GLI Training Module on Specimen Processing, STOP TB Partnership.
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Extrapulmonary specimens
ContentExtrapulmonary specimens are divided into 2 groups based on the site and mode of collection and the extent of contamination:
- Aseptically collected specimens, usually free from other microorganisms (sterile), which include fluids like spinal, pleural, pericardial, synovial, ascitic, blood, bone marrow, tissues (lymph node, tissue biopsies) and fine needle aspirates (FNAs)
- Specimens contaminated by normal flora or specimens not collected aseptically (not sterile), such as gastric lavage, bronchial washings, urine, pus, other muco-purulent specimens and stool (in case of disseminated TB in HIV infected patients and infants)
All extrapulmonary specimens have to be appropriately collected, transported, registered, decontaminated, cultured via solid culture methods or processed for MGIT960.
All extrapulmonary specimens have different processing procedures that need to be used for the respective specimen.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Pus and other muco-purulent specimens
ContentThe procedure for processing pus and other muco-purulent specimens in TB cultures is as follows:
- If the specimen is thick or mucoid and less than 10 ml in volume, digest and decontaminate with N-acetyl-L-cysteine-sodium hydroxide (NALC-NaOH) method similar to the procedure used for sputum specimens.
- If the specimen is not thick, it may be treated with 2-4% NaOH.
- The concentration of NaOH depends upon the contaminating bacteria expected to be present in the specimen.
- If the volume is over 10-12 ml, process only 10 ml or the first concentrate by centrifugation at 3000x g for 15-20 minutes.
- In such a situation, if the specimen is thick, liquefy the specimen by adding a small quantity of NALC only (50-100 mg powder) and mix well.
- After the concentration step, resuspend the sediment in 5 ml sterile water, decontaminate with NaOH and concentrate again by centrifugation.
- Always resuspend the sediment (pellet) in buffer to reduce the pH.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Gastric Aspirate
ContentThe procedure for processing gastric aspirates in TB cultures is as follows:
- Concentrate by centrifugation before decontaminating.
- Resuspend the sediment in about 5 ml of sterile water and decontaminate with N-acetyl-L-cysteine-sodium hydroxide (NALC-NaOH) method.
- After decontamination, concentrate again prior to inoculation of the sediment into the culture media.
- Due to the low pH, gastric aspirates should be processed as soon as possible (within 4 hours of collection).
- If the specimen cannot be processed quickly, it should be neutralized with NaOH before transportation or storage.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Bronchial Lavage
ContentThe procedure for processing bronchial lavage specimens in TB cultures is the same as that for sputum samples. The following aspects need to be considered:
- If the specimen is larger than 10 ml in volume, it needs to be concentrated by centrifugation (3000x g, 15-20 minutes) and the sediment resuspended in 5 ml sterile water.
- Thick/ mucoid samples need to be liquefied by mixing the specimen with a pinch (50-100 mg) of N-acetyl-L-cysteine (NALC) powder.
The processed specimen is used to inoculate the MGIT tubes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Laryngeal Swab
ContentThe procedure for processing laryngeal swab specimens in TB cultures is as follows:
- Transfer the swab (without a stick) into a sterile centrifuge tube and add 2 ml sterile water.
- Add 2 ml N-acetyl L-cysteine - sodium hydroxide (NALC-NaOH) solution, replace the cap, mix well (vortex mix), keep for 15 minutes.
- Remove the swab with forceps, squeezing the liquid out of the swab, and discard the swab.
- Fill the tube with phosphate buffer, mix and centrifuge at 3000x - 3500x g (15-20 minutes).
- Discard the supernatant fluid and resuspend the sediment in 1-2 ml sterile buffer.
The processed specimen is used to inoculate the MGIT tubes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: Tissue
ContentThe procedure for processing tissue specimens in TB cultures is as follows:
- Add saline or water (2 - 4 ml) and homogenize the tissue in a Biosafety Cabinet (BSC) using sterile equipment:
- Tissue grinder or homogenizer
- If these are not available, use a mortar and pestle
- Small tissue specimens may be placed in a petri dish with 2-4 ml sterile water and torn apart with the help of two sterile needles
- Decontaminate the homogenized specimen with N-acetyl L-cysteine - sodium hydroxide (NALC-NaOH) procedure as in processing sputum. Tissue biopsies collected aseptically do not require decontamination procedures.
- Resuspend the sediment in phosphate buffer.
The processed specimen is used to inoculate the MGIT tubes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Add saline or water (2 - 4 ml) and homogenize the tissue in a Biosafety Cabinet (BSC) using sterile equipment:
Procedure for Culture Specimen Processing: Urine
ContentThe procedure for processing urine in TB cultures is as follows:
- Aliquot the entire volume in several centrifuge tubes.
- Concentrate the specimen by centrifugation in 50 ml centrifuge tubes for 20-25 minutes.
- Resuspend the pellet in each tube with 1-2 ml of sterile water and pool together with a total volume of 5-10 ml.
- Decontaminate sediment with 4% sodium hydroxide (NaOH) for 15-20 minutes and proceed as similar to processing sputum.
The processed specimen is used to inoculate the MGIT tubes.
NOTE: Isolation of mycobacteria from urine specimens using MGIT has not been validated by Becton, Dickinson & Company (MGIT 960 manufacturer) but other investigators have reported successful isolation of mycobacteria from urine specimens. When performed, the sample to be used is:
- Totally voided, early morning urine specimen
- Pooled or mid-stream urine is not recommended.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Procedure for Culture Specimen Processing: CSF and other body fluids
ContentBody fluids collected aseptically (cerebrospinal fluid, synovial fluid, pleural fluid) can be inoculated into MGIT medium without decontamination (with the addition of PANTA). However, since it is difficult to maintain sterile conditions throughout the collection of specimens, it is recommended that all specimens be decontaminated.
Aseptically collected specimens need only light decontamination (NALC without NaOH). Steps for processing such specimens are:
- Large volume samples (>10 ml) are concentrated by centrifugation at 3000x - 3500x g (15-20 minutes).
- N-acetyl-L-cysteine (NALC) powder (50-100 mg) is added to thick/ mucoid specimens prior to centrifugation.
- Resuspend the sediment in 5 ml saline.
- Decontaminate as per the procedure for sputum samples.
The processed specimen is used to inoculate the MGIT tubes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Steps to be done after Culture Specimen Processing
ContentAfter specimen processing, the resuspended pellet may be used for various tests as per the laboratory policy or national guidelines recommended for TB diagnosis such as:
- Inoculation of sediments in Mycobacterium Growth Indicator Tube (MGIT) broth (Liquid media) and or solid media for primary culture isolation
- Subculture a loopful on a Brain Heart Infusion (BHI) Agar plate for contamination check
- Concentrated smear of processed specimen can be prepared if not prepared earlier, for a microscopy smear
- DNA extraction for Line Probe Assay
Transfer the remaining sediment into 2 mL labelled cryovials and store at 2-8⁰C for repeat or additional testing.
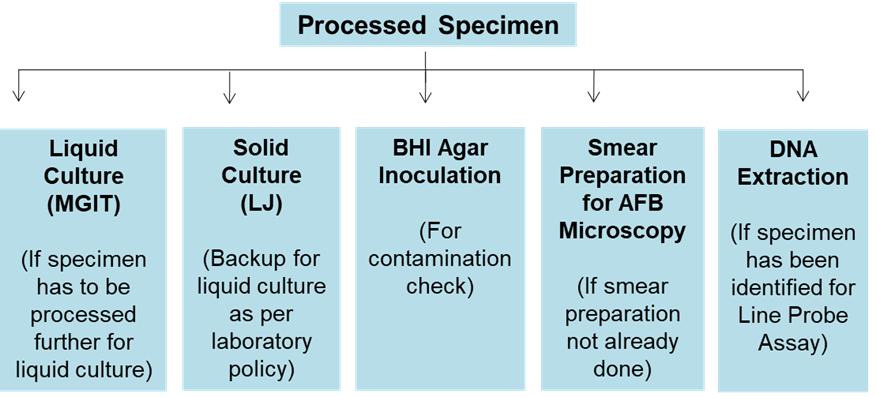
Figure: Steps After Sample Processing
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Preparation of PANTA for MGIT TB Cultures
ContentPreparation of PANTA is the first step when inoculating and incubating MGIT 960 tubes for TB cultures. The preparation steps are shown below (Figure 1).

Figure 1: Steps for Preparation of PANTA

Figure 2: Reconstitution of PANTA with growth supplement using a micropipette
NOTE:
- Reconstituted PANTA can be used for up to 5 days if stored at 2 to 8ºC, but do not freeze.
- Each PANTA vial is used for 15-18 MGIT tubes (BACTEC MGIT 960).
Please click the video below to see the full procedure for inoculating and incubating MGIT tubes for TB cultures:
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Preparation of MGIT Tubes for MGIT TB Cultures
ContentPreparation of MGIT tubes occurs after preparation of PANTA when inoculating and incubating MGIT 960 tubes for TB cultures. The preparation steps are shown below.
- Prepare the MGIT tubes in a clean Biosafety Cabinet (BSC) preferably prior to specimen processing.
- Put an absorbent sheet on the work surface of the BSC and soak it with disinfectant (bactericidal for mycobacteria).
- Before preparation of MGIT tubes:
- Know the number of samples for batch or per day
- Label each 7 ml MGIT tube with a specimen number
- Decontaminate all items to be used like micropipettes, racks, tip boxes and vortex
- Aseptically, add 0.8 ml of PANTA mixture to each MGIT tube.
- Inoculate tubes with specimen within 2 hours of adding PANTA.

Figure: Addition of 0.8 ml of PANTA/ Growth supplement mixture to the MGIT tube using a micropipette
Please click the video below to see the full procedure for inoculating and incubating MGIT tubes for TB cultures:
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Inoculation of MGIT Tubes
ContentMycobacteria Growth Indicator Tube (MGIT) contains a modified Middlebrook 7H9 broth base. When supplemented with MGIT Growth Supplement and PANTA, it provides an optimum medium for the growth of a majority of mycobacterial species. All types of specimens, pulmonary as well as extra-pulmonary (except blood), can be inoculated into MGIT for primary isolation of mycobacteria.
The steps for inoculating MGIT tubes are elaborated below:
- Using a sterile transfer pipette, add 0.5 ml of the processed sample to a 7 ml MGIT tube (0.8 ml of PANTA-growth supplement mixture already added).
- Tightly recap the tube and invert gently several times to mix well.
- Recap and swab the exterior of each tube with disinfectant-soaked gauze (use individual swabs); ensure the disinfectant does not flow into the tube.
- Leave the inoculated tubes at room temperature for 30 minutes.

Figure: Micropipette that will be used to add processed sputum specimen to the prepared MGIT tube
Precautions
- Do not add more than 0.5 ml of processed specimen.
- Volumes greater than 0.5 ml may alter the pH of the medium and result in false-positive fluorescence.
- Use a separate pipette or pipette tip for each specimen.
- Always recap the tube tightly, as loose caps may affect the detection of fluorescence.
Please click the video below to see the full procedure for inoculating and incubating MGIT tubes for TB cultures:
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Incubation of MGIT Tubes
ContentAfter inoculating the tubes, they have to be incubated in the BACTEC MGIT 960 instrument to check if there is mycobacterial growth after the stipulated time.
The steps for incubating MGIT tubes for TB cultures are as follows:
1. Enter the inoculated MGIT tubes into BACTEC MGIT 960 instrument.
2. Scan the tube bar code and specimen bar code (if available) (Figure 1).

Figure 1: Scanning the tube bar code
3. Insert tubes into the instrument in the slots assigned (indicated by a green light) (Figure 2).

Figure 2: Slots with green light indicated for inserting the tubes
4. The tubes are incubated in the MGIT 960 instrument at 37±1°C.
a. The temperature readout of each drawer must be checked daily.
b. Since the optimum temperature for growth of M. tuberculosis is 37ºC, make sure the temperature is close to 37ºC.
5. Fluorescence in each tube is measured every 60 minutes by the photodetector assembly.
6. Tubes are detected Positive or Negative, based on the fluorescence.
7. MGIT tubes should be incubated until the instrument flags them positive. After a maximum of six weeks, the instrument flags the tubes negative if there is no growth.
8. Some species such as M. ulcerans and M. genavense may require extended incubation time; if such species are expected to be present, incubate further for 2-3 weeks.
Please click the video below to see the full procedure for inoculating and incubating MGIT tubes for TB culture:
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen