Methods for Pharmacovigilance Activity Reporting [aDSM]
ContentThere are three methods for reporting pharmacovigilance activities (see figure below).
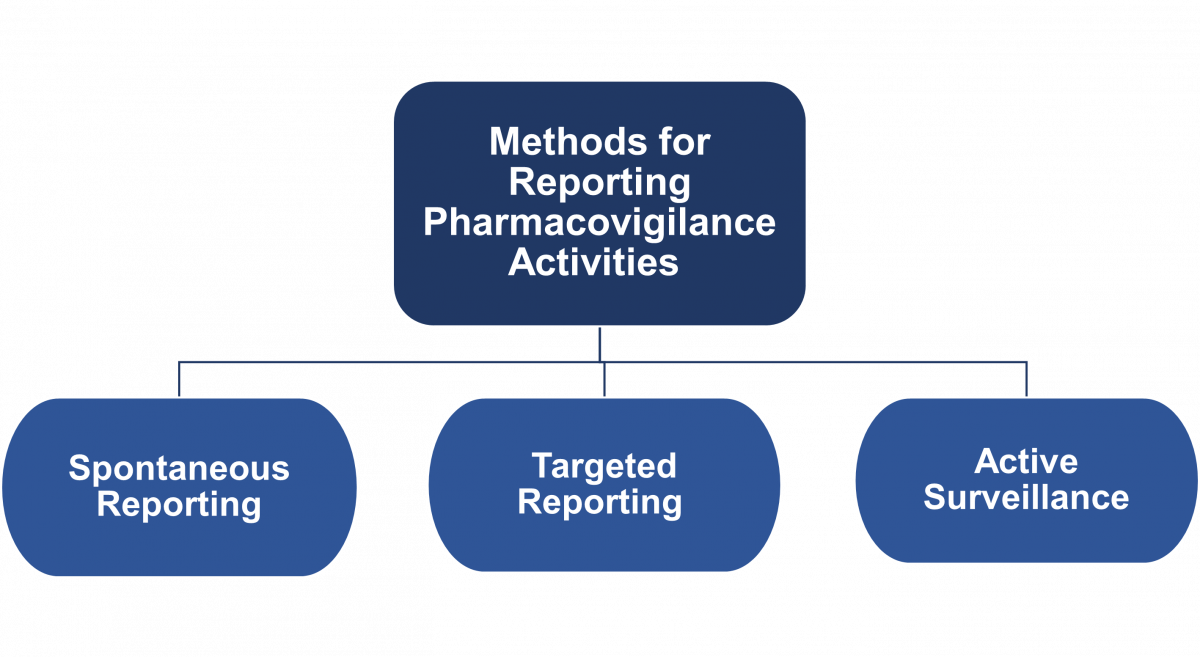
Figure: Three Methods for Reporting Pharmacovigilance Activities
- Spontaneous Reporting
- Spontaneous (or voluntary) reporting means that no active measures are taken to look for adverse effects other than the encouragement of health professionals and others to report safety concerns.
- Reporting is entirely dependent on the initiative and motivation of the potential reporters.
- This is the most common form of pharmacovigilance, sometimes termed passive reporting.
- In some countries this form of reporting is mandatory.
- Clinicians, pharmacists and community members should be trained on how, when, what and where to report.
- Targeted Reporting
- It focuses on capturing Adverse Drug Reactions (ADRs) in a well-defined group of patients on treatment.
- Health professionals in charge of the patients are sensitized to report specific safety concerns.
- Active Surveillance
- It is a proactive effort made to elicit adverse events.
- This is achieved by active follow-up after treatment and the events may be detected by asking patients directly or screening patient records.
- It is best done prospectively.
- The most comprehensive method of active surveillance is Cohort Event Monitoring (CEM), which is an adaptable and powerful method of getting good comprehensive data.
- Other methods of active monitoring include the use of registers, record linkage and screening of laboratory results in medical laboratories.
- This is an important method of reporting under active Drug Safety Monitoring (aDSM) for Drug-resistant TB (DR-TB) patients.
Resources
- Training Modules (1-4) for Programme Managers & Medical Officers (NTEP), 2020.
- A Practical Handbook on the Pharmacovigilance of Medicines Used in the Treatment of Tuberculosis, 2012.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
- Spontaneous Reporting
Causality Assessment for ADR Reporting
ContentAttribution definitions for causality assessment are divided into five categories and are as follows:
- Not related
Adverse Event (AE) that is not related to the use of the drug.
- Doubtful
AE for which an alternative explanation is more likely, e.g., concomitant drug(s), concomitant disease(s) or the relationship in time suggests that a causal relationship is unlikely.
- Possible
AE that might be due to the use of the drug. An alternative explanation, e.g., concomitant drug(s) or concomitant disease(s) is inconclusive. The relationship in time is reasonable, therefore, the causal relationship cannot be excluded.
- Probable
AE that might be due to the use of the drug. The relationship in time is suggestive, e.g., confirmed by de-challenge. An alternative explanation is less likely, e.g., concomitant drug(s), concomitant disease(s).
- Certain (very likely)
AE that is listed as a possible adverse reaction and cannot be reasonably explained by an alternative explanation, e.g., concomitant drug(s), concomitant disease(s). The relationship in time is suggestive, e.g., confirmed by de-challenge and rechallenge.
The Drug-resistant TB Centre (DR-TBC) committee, in coordination with the Adverse Drug Reaction (ADR) Monitoring Centre (AMC) will review and confirm the causality of all serious events/ reactions in relation to therapy.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
Causality Assessment: WHO-UMC [UPSALA Monitoring Centre] Causality Categories for ADR Reporting
ContentThe World Health Organisation-Uppsala Monitoring Centre (WHO-UMC) system has been developed as a practical tool for the assessment of case reports.
The table below lists the various causality categories and their assessment criteria that have been developed under this system.
Table: WHO-UMC Causality Categories; Source: WHO-UMC System for Standardised Case Causality Assessment, p2. CAUSALITY TERM/ CATEGORY ASSESSMENT CRITERIA Certain/ Very Likely - Event or laboratory test abnormality, with plausible time relationship to drug intake
- Cannot be explained by disease or other drugs
- Response to withdrawal plausible (pharmacologically, pathologically)
- Event definitive pharmacologically or phenomenologically (i.e., an objective and specific medical disorder or a recognized pharmacological phenomenon)
- Rechallenge satisfactory, if necessary
Probable/ Likely - Event or laboratory test abnormality, with reasonable time relationship to drug intake
- Unlikely to be attributed to disease or other drugs
- Response to withdrawal clinically reasonable
- Rechallenge not required
Possible - Event or laboratory test abnormality, with reasonable time relationship to drug intake
- Could also be explained by disease or other drugs
- Information on drug withdrawal may be lacking or unclear
Unlikely - Event or laboratory test abnormality, with a time to drug intake that makes a relationship improbable (but not impossible)
- Disease or other drugs provide plausible explanations
Conditional/ Unclassified - Event or laboratory test abnormality
- More data for proper assessment is needed, or additional data under examination
Unassessable/ Unclassifiable - Report suggesting an adverse reaction
- Cannot be judged because the information is insufficient or contradictory
- Data cannot be supplemented or verified
Resources
- The Use of the WHO-UMC System for Standardised Case Causality Assessment.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
Severity Criteria for Reporting ADRs
ContentAdverse Drug Reactions (ADRs) have been graded based on their severity. The figure below provides criteria for the assessment of the severity grade of ADRs.

Figure: Criteria for Severity Grade Assessment of ADRs
- The investigator should use their clinical judgment in assessing the severity of events not directly experienced by the subject, e.g., laboratory abnormalities.
- Safety assessment measure is the proportion of patients experiencing a grade 3 or greater adverse event, as defined by Division of AIDS (DAIDS) criteria during treatment and follow-up.
- Please click here for more information on the DAIDS criteria.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Division of AIDS (DAIDS) Table for Grading the Severity of Adult and Paediatric Adverse Events, 2017.
Kindly provide your valuable feedback on the page to the link provided HERE
Reporting of Adverse Events and Serious Adverse Events
ContentAll serious and non-serious adverse events which are possibly, probably or very likely related to any anti-TB drug need to be reported by the physician to the National TB Elimination Programme (NTEP).Serious Adverse Events (SAE) needs to be reported to the nearest ADR monitoring centre (AMC) and Central TB Division (CTD) within 24 hours. Any death of a patient occurring during treatment, regardless of causality, must be reported as an SAE.
aDSM-treatment review form to be filled whenever the DR-TB patient develops any SAE
- The primary responsibility of filling up the aDSM forms will be with the nodal officer of the DR-TB centre with the help of a senior medical officer (SMO) or medical officer (MO) designated.
- It is essential that Nikshay data entry are being done on regular basis by statistical assistant at the NDR-TBC and senior DR-TB TB-HIV supervisor at the DDR-TBC centre.
- Forms should be maintained in hard copies until the ADR module is active in Nikshay.
Once relevant forms of aDSM are filled in Nikshay, information is directly communicated to the pharmaco-vigilance programme of India (PvPI) through the Vigiflow=connecting bridge for signal generation.
Resources
aDSM treatment initiation form
ContentUnder the National TB Elimination Programme (NTEP), as an integral part of the Programmatic Management of Drug-resistant TB (PMDT), the aDSM treatment initiation form is to be filled by the staff at the health facility during the treatment initiation of all DR-TB patients.
This form is available as annexure 32 in the PMDT guidelines 2021 and covers the following information:
- Patient’s name, age, sex, Ni-kshay id, PMDT number, and date of interview
- TB-related details - Type of TB, type of drug resistance, and previous history of TB treatment
- Pregnancy and lactation-related details (if applicable)
- History of addiction/ substance use
- Current and past medical conditions/ events – with the date of onset and recovery (if applicable)
- Details about the medication consumed in the past 30 days (both TB and traditional)
- Details about medicines (other than anti-TB medicines) prescribed during the interview
- Name of the treating facility, name of treating clinician and signature of the person reporting
ImageFigure: aDSM Treatment Initiation Form; Source: Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2021.
Importance of aDSM Treatment Initiation Form
- This form records the pre-treatment medical (including past TB history) history of the patients and hence helps the treating physician during the treatment initiation regarding the drugs being prescribed, additional care required, etc.
- The form also helps as a document to refer back for making important medical decisions in the event of any adverse reactions (both serious and otherwise) reported by the patient during the course of the treatment.
Resources
- Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2021.
- Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2017.
- Active Tuberculosis Drug-safety Monitoring and Management (aDSM) - Framework for Implementation, WHO End TB Strategy, 2015.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test When should the aDSM treatment initiation form be filled? When a DR-TB patient is initiated treatment When a DR-TB patient is lost to follow-up When ADR is reported by a DR-TB patient When a DR-TB patient completes treatment 1 The aDSM treatment initiation form is to be filled out by the staff at the health facility during the treatment initiation of all DR-TB patients. Yes Yes aDSM review form
ContentThe aDSM treatment review form is a schedule to be filled out:
i) When any adverse event is reported by a patient on a newer drug containing a DR-TB regimen
ii) When a Serious Adverse Event (SAE) of the Division of Allergy and Infectious Diseases (DAIDS) grade 3 or 4 is reported by patients on other DR-TB treatment
This form is to be filled out by the health facility managing the SAE and the following information needs to be covered:
- Patient details (name, age, sex, weight, height, Ni-kshay id, PMDT no.)
- TB-related details - Type of TB, type of drug resistance, and previous history of treatment
- Pregnancy and lactation-related details (if applicable)
- Adverse Drug Reaction (ADR) details – Course of events, timing and suspected cause of ADR, DAIDS grading of the Adverse Event (AE), date of onset and resolution of ADR
- Seriousness of the AE (life-threatening, requires hospitalisation, permanent disability, congenital anomaly, conditions where the intervention is required to prevent permanent impairment/ damage, death)
- Outcome of the adverse event
- Causality with the anti-TB medicines the patient was consuming and actions taken by the clinician in case of suspected adverse event linked to a drug
- Any other relevant clinical information
ImageImageFigure: aDSM Treatment Review Form; Source: Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2017.
Importance of aDSM Review Form
- Although all AEs are to be clinically managed, aDSM treatment review form focuses mainly on the serious AEs and is an integral component of the Programmatic Management of Drug-resistant TB (PMDT).
- This form helps to carefully monitor and document the drug-related harms attributed to the recent developments in DR-TB treatment, particularly the approval for use of new medicines ahead of the completion of phase III trials, increased use of repurposed drugs for Extensively Drug-resistant TB (XDR-TB) treatment and the development of novel second-line anti-TB regimens, some of which may not have been described yet.
- This also helps clinicians to take corrective actions and help improve the health and safety of patients.
Resources
- Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2021.
- Guidelines on Programmatic Management of Drug-resistant TB (PMDT) in India, CTD, MoHFW, India, 2017.
- Active Tuberculosis Drug-safety Monitoring and Management (aDSM) - Framework for Implementation. WHO End TB Strategy, 2015.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Who should fill out the aDSM treatment review form?
Patient
Health facility managing the Serious Adverse Event (SAE)
Both of the above
None of the above
2
aDSM treatment review form should be filled out by the health facility managing the Serious Adverse Event (SAE).
Yes
Yes
Fullscreen