Visualization of CBNAAT Test Results
ContentThe test results of the Cartridge-based Nucleic Acid Amplification Test (CBNAAT) assay are displayed in the ‘View Results’ window, of the CBNAAT software.
For visualizing the results after the test is completed:
- In the CBNAAT Dx System window, click View Results on the menu bar.
- The View Results window appears.
- To select a test result, click View Test.
- ‘Select Test to Be Viewed’ - dialog box appears.
- Select the test of interest.
- Click OK.
Note: The selected test result appears in the ‘View Results’ window. A result will be displayed in PDF format.
To generate a PDF report
- To generate a report in PDF format, click on “Report” and then select the result you want.
- The PDF report will be generated.
Interpretation of Test Results
- The ‘View Results’ window displays information about the test, such as sample ID and run-time on the left-hand side panel.
- The interpretation of the result is in the center, and the real-time PCR amplification curves are displayed at the bottom.
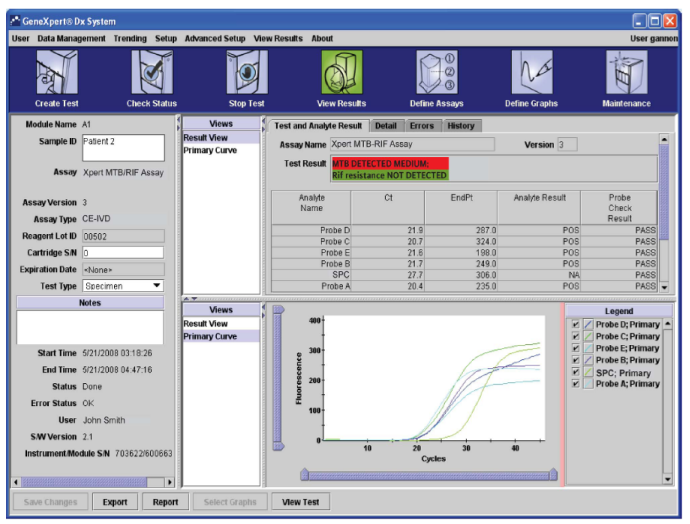
Figure: User 'View Results' Window showing the information about test, interpretation of results and real-time PCR curves
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
CBNAAT Results Interpretation
ContentOn completion of a test run, the Cartridge-based Nucleic Acid Amplification Test (CBNAAT) gives the following results:
- MTB DETECTED; Rif Resistance DETECTED
- MTB DETECTED; Rif Resistance NOT DETECTED
- MTB DETECTED; Rif Resistance INDETERMINATE
- MTB NOT DETECTED
- Error
- Invalid
- No result
Conclusive results include: MTB NOT DETECTED, MTB DETECTED with Rif Resistance/ without Rif Resistance.
Non-conclusive results include: MTB Detected, Rif Resistance Indeterminate, Errors, Invalid and No Result - the test has to be repeated in these cases.
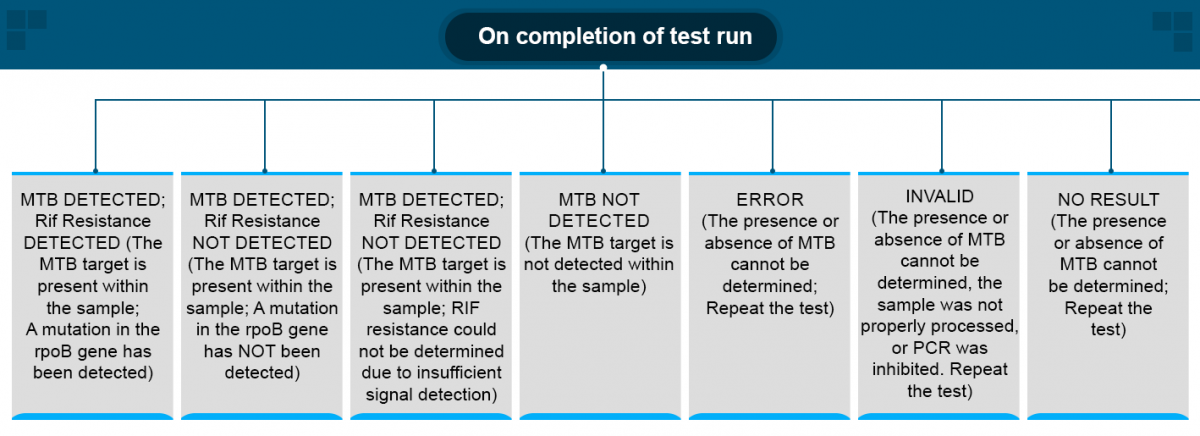
Figure: CBNAAT Result Algorithm
Video fileVideo : CBNAAT Results Interpretation
Resources
CBNAAT Assay Limitations
ContentThe Cartridge-based Nucleic Acid Amplification Test (CBNAAT) test has some limitations such as:
- Bacterial load below the Limit of Detection (LOD ~ 130 CFU/ml) may result in a false-negative result.
- Patients on an anti-TB regimen can still have positive results due to killed bacilli in the specimen and hence cannot be used for follow-up.
- A positive test result does not necessarily indicate the presence of viable organisms. It is, however, presumptive for the presence of Mycobacterium tuberculosis (MTB) and Rifampicin (Rif) resistance.
- MTB detection is dependent on the number of organisms present in the sample. Quality specimen collection and timely processing of the sample will minimize the errors.
- Test results might be affected by anti-TB medication. Therefore, therapeutic success or failure cannot be assessed using this test, because DNA might persist following antimicrobial therapy.
- Mutations or polymorphisms in primer or probe binding regions may affect the detection of new or unknown MDR or Rif-resistant strains, resulting in a false-negative result.
- Any modification in sample processing may alter the performance of the test.
- Results should be interpreted in conjunction with clinical data available to the clinician.
Video fileVideo:
CBNAAT Assay Limitations
Resources
Results Entry in Lab Register for NAAT
ContentThe results for Nucleic Acid Amplification Test (NAAT) assays are entered in Culture and Drug Susceptibility Testing (C&DST) register. The key variables entered are shown in the table below.
Table: Key Variables entered in C&DST Register; Source: Guidelines for PMDT in India, 2021. VARIABLES SET 1 VARIABLES SET 2 VARIABLES SET 3 VARIABLES SET 4 Test ID Health Facility (HF) name Residential district Current facility HF type Date of test updated in Nikshay Lab type Type of test Predominant symptom Date tested Patient ID Reason for testing Predominant symptom duration Date reported Episode ID Treatment status History of Anti-TB Treatment (ATT) Test status Name Diagnosis date No. of Health Care Provider (HCP) visited before the diagnosis of the current episode Type of specimen Gender TB treatment start date The visual appearance of sputum Date of specimen collection Age Current facility state State name Primary phone Current facility district District name Address Current facility TB Unit (TU) TB unit Residential state Current facility HF NAAT results are reported in the results section of “Request Form for examination of biological specimen for TB” including:
- Select Type of test: Cartridge-based Nucleic Acid Amplification Test (CBNAAT)/ TrueNAT
- Select Sample: A/B
- Select M. tuberculosis: Detected/ Not Detected/ Not Available (NA)
- Select Rif Resistance: Detected/ Not Detected/ Indeterminate/ NA
- Select Test: No result/ Invalid/ Error; Error code
- Date tested
- Date Reported
- Reported by (name and signature)
- Laboratory name
Video fileVideo : CBNAAT/Truenat Results Entry in Lab Register
Resources
Retest Procedure for CBNAAT
ContentThe assay needs to be repeated by using a new cartridge if one of the following test results occur:
INVALID: An INVALID result indicates that Sample Processing Control (SPC) failed. The sample was not properly processed, or Polymerase Chain Reaction (PCR) was inhibited.
ERROR: An ERROR indicates that Probe Check Control (PCC) failed, and the assay was aborted possibly due to the reaction chamber being filled improperly, a reagent probe integrity, syringe pressure issues, or failure of the CBNAAT module.
NO RESULT: A NO RESULT indicates that insufficient data were collected. For example, the operator stopped a test that was in progress.
RIF Indeterminate: RIF indeterminate result indicates that the sample has a less bacterial load.
Program guidelines recommend obtaining a second specimen to confirm rifampicin resistance in these scenarios.
How to Perform the Retest?
- Leftover sputum or fresh sputum or reconstituted sediment:
Treat it with a new Sample Reagent (SR) and load it into the new CBNAAT cartridge.
- Sufficient leftover SR-treated sample:
Use within 4 hours of initial addition of SR to the sample - Load into the new CBNAAT cartridge.
Do not use SR-treated sample if it is more than 4 hours old - Over-treatment may lead to false-positive test results.
Always use a new cartridge!
Video fileVideo : Retest Protocol for CBNAAT
Resources
Fullscreen