Delivery of TB Care in Private Sector
- Read more about Delivery of TB Care in Private Sector
- Log in to post comments
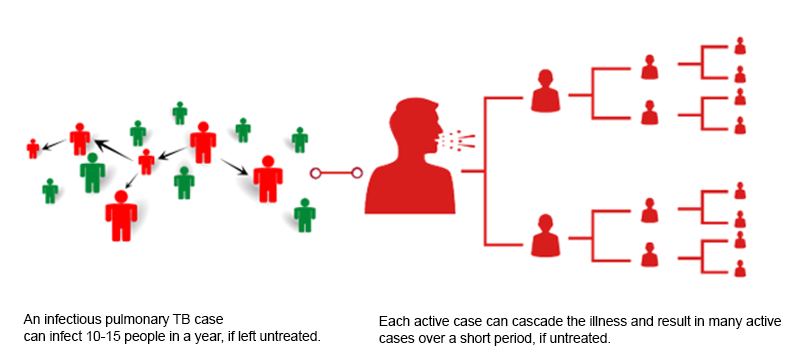
The private sector for tuberculosis (TB) care is everything outside the ambit of government-run public health initiatives. It consists of a wide range of providers from individual medical practitioners of many different systems of medicine, including: