Treatment Extension in Longer Oral M/XDR-TB Regimen
The total duration of a longer oral Multidrug/ Extensively drug-resistant TB (M/XDR-TB) regimen is 18–20 months.
Content Status where content has been uploaded and published on LMS.
The total duration of a longer oral Multidrug/ Extensively drug-resistant TB (M/XDR-TB) regimen is 18–20 months.
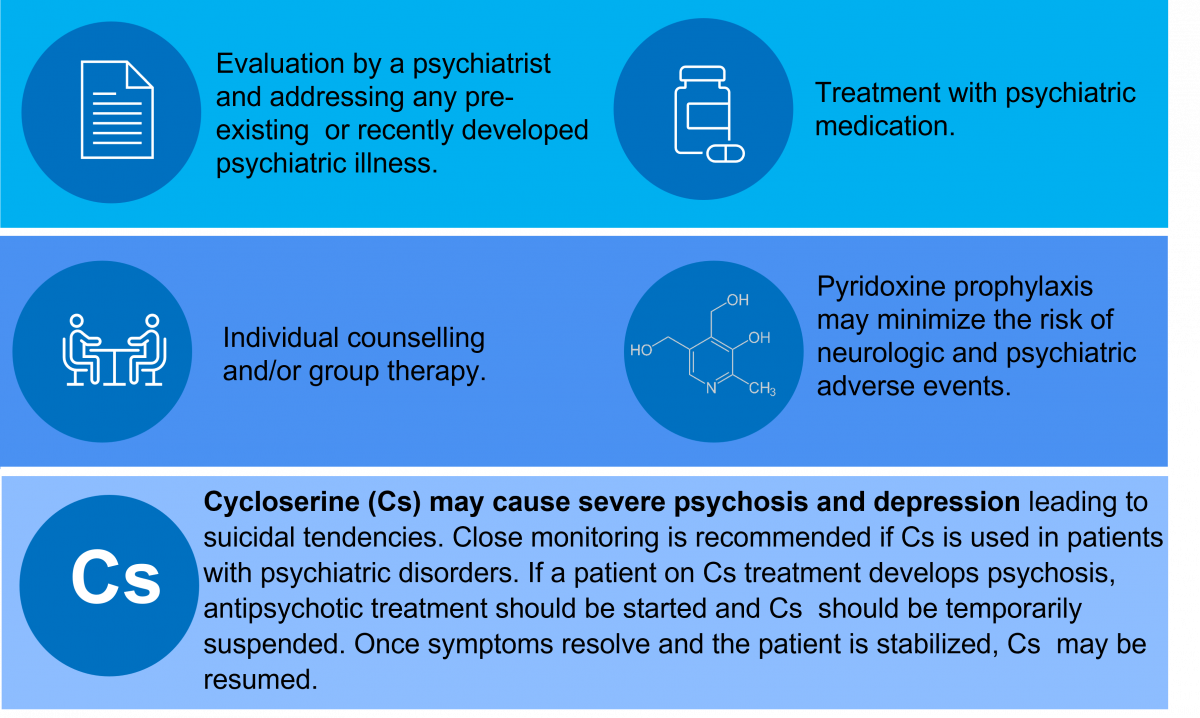
There is a high baseline incidence of depression and anxiety in patients with Multidrug-resistant (MDR)/ Extensively Drug-resistant TB (XDR-TB) patients. Special precautions need to be taken for M/XDR-TB treatment-initiated patients suffering from pre-existing psychiatric illnesses.
Drugs Associated with Psychosis: Cycloserine (Cs), High-dose Isoniazid (Hh), Fluoroquinolone (FQ) and Ethionamide (Eto)
The treating physician should elicit the history of past or present seizure disorders in all Drug-resistant TB (DR-TB) patients.
If the seizures are not under control, initiation or adjustment of anti-seizure medications will be needed before the start of DR-TB treatment. In addition, if other underlying conditions or causes for seizures exist, they should be corrected.
In Drug-resistant TB (DR-TB) patients with localized disease, surgery, as an adjunct to chemotherapy, can improve outcomes; provided that skilled thoracic surgeons and excellent postoperative care is available.
A patient with a unilateral resectable disease may be considered for surgery if there is:
All women of childbearing age who are awaiting results of the Culture and Drug Susceptibility Test (C&DST), as well as those receiving Drug-resistant TB (DR-TB) treatment, should be advised and counselled intensively to use birth control measures because of the potential risk to both the mother and the foetus.
The principles for the treatment and monitoring of Multidrug-resistant (MDR)/ Rifampicin-resistant TB (RR-TB) in children are quite similar to adults and use the same second-line drugs.
Principles
Apart from clinical evaluation, the patients need to be closely assessed by various laboratory parameters to monitor the improvement on treatment, drug-induced adverse events or co-morbidities to enable timely interventions to address these and improve the probability of treatment success, survival and quality of life.
The following points should be covered in follow-up monitoring of Drug-resistant TB (DR-TB) patients:
Because of the risk for potential drug-drug interactions, the following medications are not allowed during the 24-week administration of Bedaquiline (Bdq) and up to one month after the last dose of Bdq:
Drug dose administration for shorter/ longer oral Bedaquiline (Bdq)-containing Multidrug-resistant (MDR)/ Rifampicin-resistant (RR)-TB regimen depends on the factors described below.