Requirements from Institutions for the Establishment of Nodal DR-TB Centre
The requirements from institutions for establishing Nodal Drug-resistant Tuberculosis Centres (NDR-TBCs) are as follows:
Default status of page at the time it is added for creation.
The requirements from institutions for establishing Nodal Drug-resistant Tuberculosis Centres (NDR-TBCs) are as follows:
Shorter Oral Bedaquiline-containing regimen is recommended for Multidrug-resistant/ Rifampicin-resistant (MDR/RR-TB) patients in whom:
Inclusion Criteria
Drug Susceptibility Testing (DST) based inclusion criteria:
There are five principal ways to prevent Drug-resistant Tuberculosis (DR-TB) as given in the figure below.

Figure: Five Principal Ways to Prevent DR-TB; Source: Guideline for PMDT in India, 2021.
Baseline Second-line Drug-susceptibility Test (SL-DST) means to offer (DST) for Fluoroquinolones (FQ) once Rifampicin (R) and/or Isoniazid (Inh) resistance is diagnosed.
Pre-treatment counselling must serve as an informed decision-making process that enables patients to make a duly informed decision regarding the use of all anti-TB drugs and regimen, including newer drugs.
Written consent is not needed for any treatment regimen under National TB Elimination Programme (NTEP).
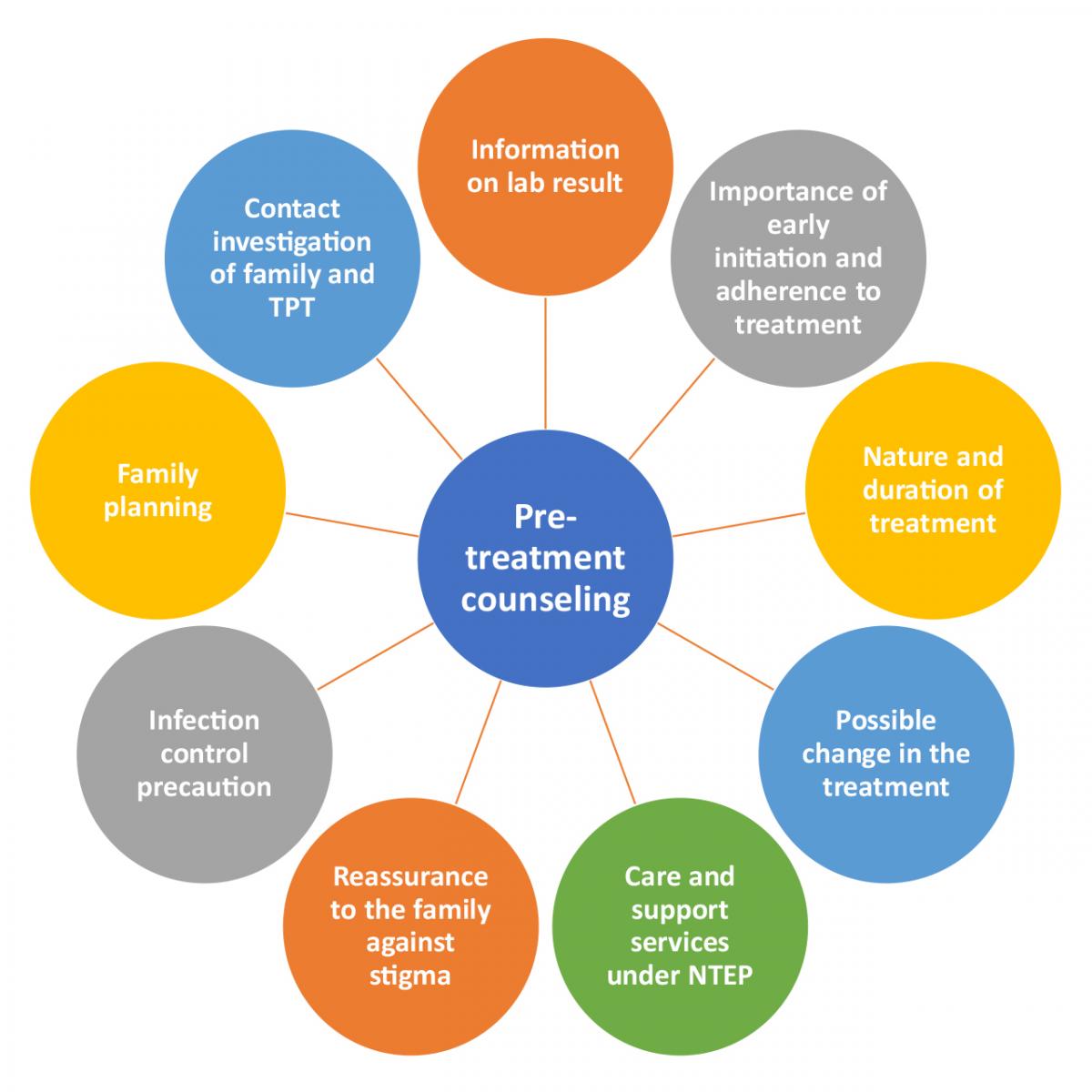 Figure: Key points to be covered during pre-treatment counselling; Source: PMDT Guidelines India, 2021, p45.
|
There are two main evaluations that must be conducted during pre-treatment evaluation to initiate patients on a shorter oral bedaquiline-containing MDR/RR-TB regimen. These evaluations are:
Clinical Evaluation
Laboratory-based Evaluation
Delamanid (Dlm) is the first approved drug in the class of nitro-dihydro-imidazo-oxazoles for the treatment of Multi-drug Resistant TB (MDR-TB).

Figure: Delamanid (DLM) Tablets
Mechanism of Action
It is a bactericidal drug with 36 hours of half-life and acts with two different mechanisms of action.
 Figure: Sirturo 100 mg Bedaquiline Tablets
|
Adherence to the Tuberculosis Preventive Treatment (TPT) course and treatment completion are important determinants of clinical benefit, both at individual and population levels.
Once Tuberculosis Preventive Treatment (TPT) is initiated, the individuals will be monitored by the doctor for clinical and laboratory parameters as shown below: