External Quality Assurance for CBNAAT
- Read more about External Quality Assurance for CBNAAT
- Log in to post comments
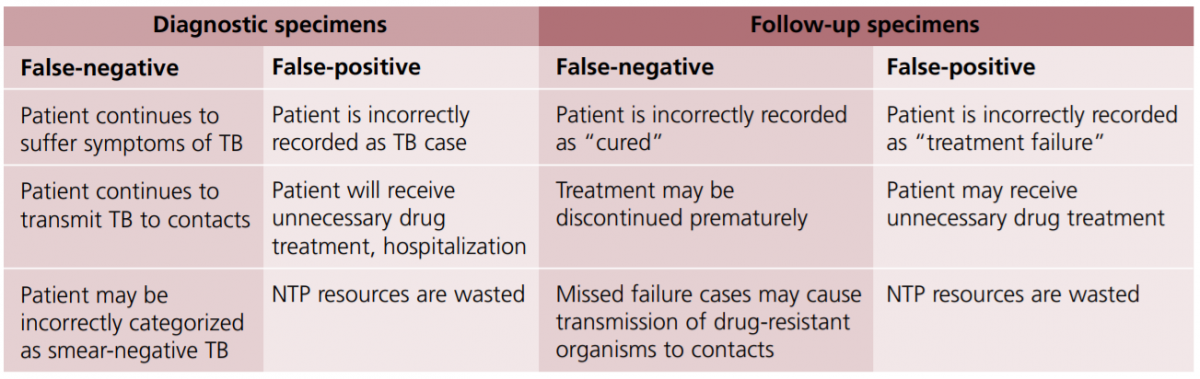
External Quality Assurance (EQA) ensures that high-quality testing can be carried out efficiently and without interruption. It involves Proficiency Testing (PT) and On-site Evaluation (OSE).
PT is an important component of EQA for Cartridge-based Nucleic Acid Test (CBNAAT) under the National TB Elimination Program (NTEP) and guarantees accurate and reproducible results.
Importance of EQA/ PT for CBNAAT