Regimen, Duration and Dosage for Isoniazid [H] Mono/Poly DR-TB Regimen
Isoniazid (H) mono/ poly Drug-resistant TB (DR-TB) regimen has the following regimen, duration and dosage of drugs.
Regimen: (6 or 9) Lfx R E Z
Dosage
Isoniazid (H) mono/ poly Drug-resistant TB (DR-TB) regimen has the following regimen, duration and dosage of drugs.
Regimen: (6 or 9) Lfx R E Z
Dosage
Pre-treatment evaluation for any TB patient must include a thorough clinical evaluation by a doctor with:
No additional investigations (except the basic evaluations mentioned above) are required for Isoniazid (H) mono/ poly Drug-resistant TB (DR-TB) patients unless clinically indicated.
The table below showcases the adverse drug events that may be caused by drugs used for longer oral Multi (M)/ Extensively Drug-resistant TB (XDR-TB) regimen. In these situations, replacement drugs are used instead of these drugs.
|
ADVERSE DRUG EVENTS |
DRUGS |
|---|---|
|
QT prolongation |
Pre-treatment evaluation for patients on a longer oral Multi/ Extensively Drug-resistant TB (M/XDR-TB) regimen requires both clinical evaluation and laboratory-based evaluation as given below.
Clinical Evaluation
Laboratory-based Evaluation
In a situation where Drug-resistant TB (DR-TB) patients may have consumed anti-TB drugs for some duration from the private sector, such prior anti-TB treatment is not likely to be uniformly reliable as far as the quality or quantity and duration of drugs consumed is concerned.
Given that uncertainty, the basic principle is that the duration of the DR-TB regimen under the National Tuberculosis Elimination Programme (NTEP) need not be reduced.
Many times, Drug-resistant Tuberculosis (DR-TB) patients diagnosed in the private sector will wish to avail services from the public sector.
The National TB Elimination Programme (NTEP) strongly recommends bacteriological confirmation of any DR-TB patient before initiation of treatment and discourages any empirical treatment.
Drug Susceptibility Testing (DST) results available from private laboratories for such patients will be considered acceptable under the following situations:
The National TB Elimination Program (NTEP) makes an effort to provide Tuberculosis (TB) services to each TB patient, including services to Drug-resistant TB (DR-TB) patients notified/ referred from the private sector.
The requirements from institutions for establishing Nodal Drug-resistant Tuberculosis Centres (NDR-TBCs) are as follows:
The total duration of treatment in this regimen is 9-11 months with Intensive Phase (IP) at least 4 months and Continuation Phase (CP) for 5 months. Treatment extension of IP is done up to 2 months based on follow-up results and is indicated in the algorithm presented in the figure below.
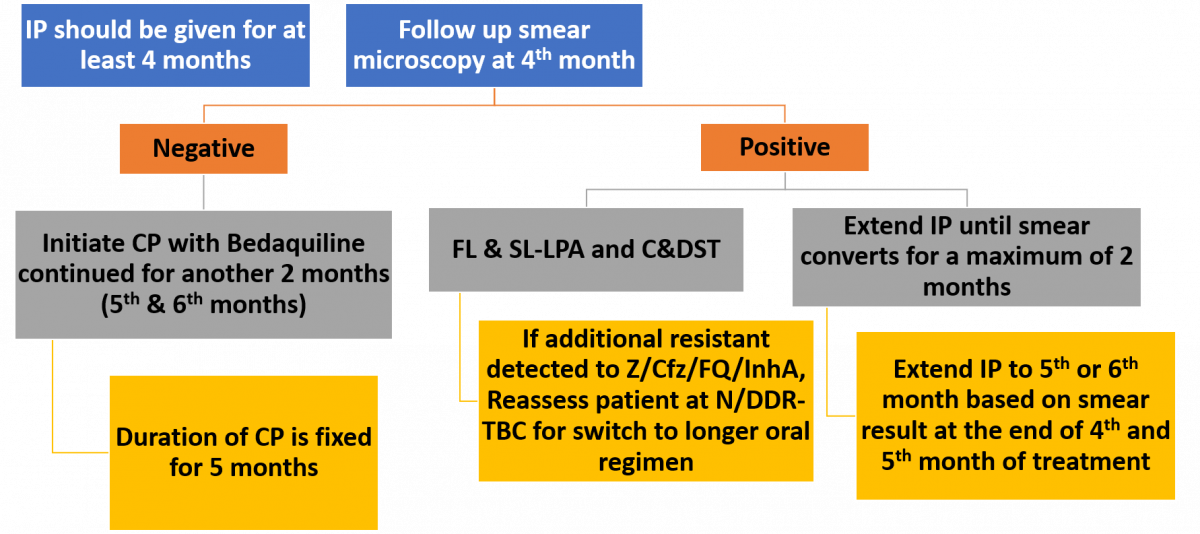
Figure: Treatment Extension/ Regimen Change Based on Follow up Smear/ Culture/ DST Results
Based on the World Health Organization (WHO) treatment guidelines, 2020 recommendations, the National TB Elimination Programme (NTEP) have decided to transition from the current shorter injectable-containing Multi-drug Resistant (MDR)/ Rifampicin-resistant TB (RR-TB) regimen to the shorter oral bedaquiline-containing MDR/RR-TB regimen in the year 2021.
Salient Features of the Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen