Biosafety Requirements in Line Probe Assay [LPA] Laboratory
ContentDNA extraction for Line Probe Assay (LPA) from clinical specimens can be performed in either a BSL-2 or BSL-3 laboratory, while DNA extraction from mycobacterial cultures must be performed in a BSL-3 laboratory. Only after heat-killing of the organism and DNA isolation can the sample be considered non-infectious and moved to the LPA laboratory.
The subsequent steps, i.e., amplification and post-amplification, only require a BSL-1 laboratory.
Aspects of biosafety in LPA laboratory include:
- Facility Design
- Workflow description
- Rooms required to complete LPA procedures
- Access to authorized personnel only
- Electricity and backup power supply
- Laboratory Equipment
- Equipment operation
- Equipment maintenance
- Safe Lab Practices
- Cleaning and disinfecting work areas
- Use of Personal Protective Equipment (PPE)
- Disinfection and decontamination
- Biomedical waste management
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
- Facility Design
Biosafety Requirements in LPA Facility Design
ContentThere are two types of risk while setting up a Polymerase Chain Reaction (PCR) facility for Line Probe Assay (LPA):
- Biohazard risk: The potential that a laboratory worker will become infected when working with live M. tuberculosis.
- Bio-risk: The potential that specimens or reagents will become contaminated with DNA, amplified products (amplicons) or exogenous contaminants that lead to false-positive PCR results.
Precautionary measures to reduce the biohazard and bio-risk in DNA molecular procedures are critical and drive the general facility design.
LPA Facility Design and Layout
There should be four separate rooms for:
- DNA extraction
- Reagent preparation for PCR - the pre-amplification step
- PCR amplification
- Hybridization and detection
In case of space constraints, one room can be used for both hybridization and amplification.
Movement in the LPA Facility (Figure 1 and 2)
- There should be a unidirectional flow of lab staff, as well as the tools used.
- Never reverse the flow.
For example:
- In the morning, commence work in the reagent preparation room as it is “the ultra-clean room”. From this room, one may proceed to either the specimen preparation room, the PCR amplification room, or the PCR amplification and detection room.
- If one starts work in the specimen preparation room they should not enter the reagent preparation room, but they may work in the PCR amplification room, or the PCR amplification and detection room.
- If one starts working in the amplification or detection room they should not enter the reagent preparation room.
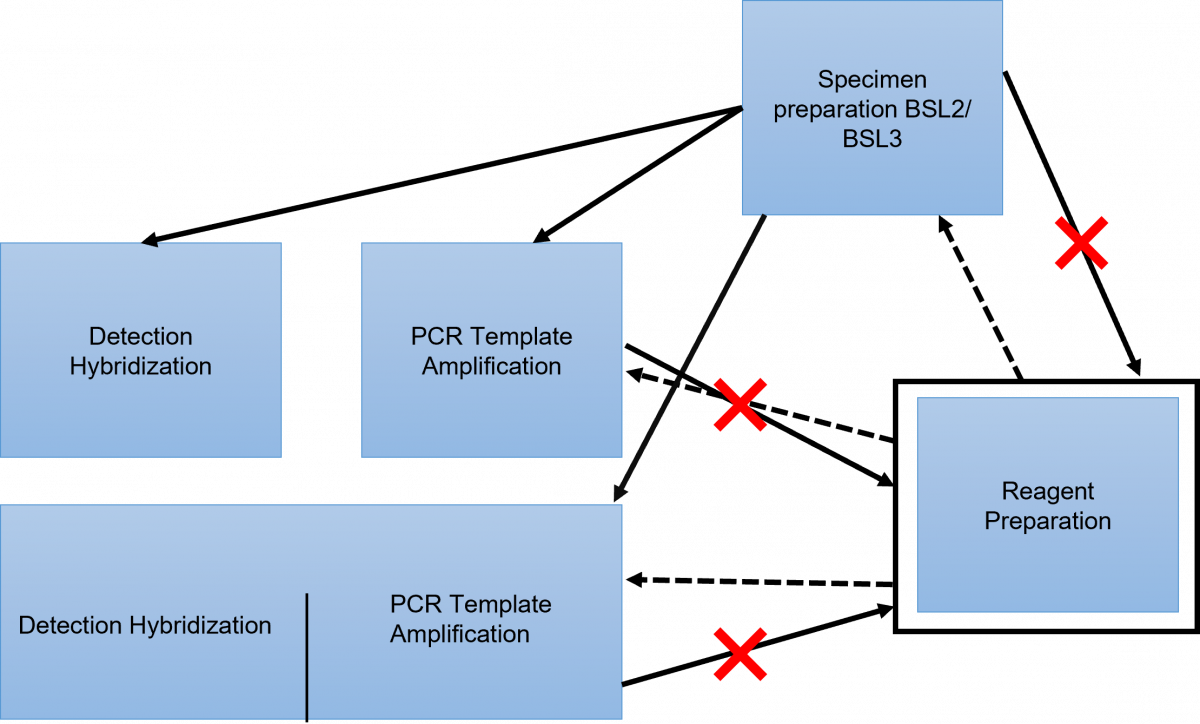
Figure 1: LPA Facility Layout showing Maintenance of Unidirectional Movement and Flow of Tools

Figure 2: Requirements to Maintain the Unidirectional Flow of Tools and Staff in the LPA Laboratory
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Electrical Supply and Back-up Power Requirements in LPA Facilities
ContentImportance of Electrical Supply and Backup Power in the Line Probe Assay (LPA) Facility
- Reagents used in LPA are to be stored at 4°C or -20°C. Hence, require uninterrupted electricity supply/ backup power.
- Amplification and hybridization procedures must be conducted under closely monitored temperature conditions.
- Uninterrupted Power Supply (UPS) connection is required during PCR amplification and use of the automated hybridization systems to avoid interruption of the procedure and subsequent loss of results.
Features of Electrical Fittings, Wiring, Power Points, Sockets, Fixtures and Electrical Boxes in the LPA Facility
- All the electrical fittings in the LPA laboratory areas should be suitable for clean room application and sealed (all conduits, outlets) with silicon sealant, leak-proof and capable of withstanding chemical exposures during fumigation.
- Necessary wiring and power points (at least six in each room) should be provided for all equipment.
- Modular type, power points of 5A/15A required at various locations on the wall as per placement of equipment.
- Power backup of about 125 KVA capacity is required for LPA laboratory.
- Provision should be made for backup power supply to the critical components and equipment through a UPS and diesel power generator set.
- There should be two outlets, one for direct line and one for UPS backups, planned for each lab equipment.
- The sockets meant for UPS should be screen printed as (UPS) for ease of operation and identification. Marked wires and cables used should be copper wire of standard make (ISI marked) and manufacturer.
- Light fixtures should be placed flush with roof, with gasket or otherwise sealed with silicon sealant.
- Accessibility for replacement of light fixtures should be provided from the roof side.
- Circuit breakers must be able to accommodate the electrical demand needed to operate the various laboratory equipment for LPA.
- All electrical boxes and wiring should be certified to avoid surges that might render the equipment inoperable.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Equipment Required in Different Rooms in the LPA Lab Facility
ContentThe design of the LPA facility includes the availability of different rooms to perform different steps. Each room has a set of specific equipment.
There should be a minimum of 4 separate rooms to carry out procedures. The details of these rooms and equipment are elaborated below:
1. DNA Extraction Room: For DNA extraction from decontaminated samples. Table 1 shows the equipment in the DNA extraction room.
Table 1: Equipment in the DNA Extraction Room
BIOSAFETY CABINET MICROLITER CENTRIFUGE VORTEX MIXER 


HOT AIR OVEN REFRIGERATOR 

2. Master Mix/ Reagent Preparation Room: For the preparation of reagents required in the master mix for PCR. Table 2 shows the equipment required in this room.
Table 2: Equipment in the Master Mix/Reagent preparation room
PCR HOOD
PICO-FUGE
-20ºC FREEZER



3. DNA Amplification Room: For PCR amplification of DNA. Table 3 shows the equipment in this room.
Table 3: Equipment in the DNA Amplification Room
PCR HOOD
THERMAL CYCLER
REFRIGERATOR



4. Hybridization – detection Room: For hybridization of amplicons with probes and detection of bands. Table 4 shows the equipment required in this room.
Table 4: Equipment in the Hybridization-detection Room
TWINCUBATOR
GT BLOT


REFRIGERATOR
WATER BATH


Resources
- Molecular Detection of Drug-resistant Tuberculosis by Line Probe Assay.
- GLI Training Package on LPA.
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Biosafety Essentials: Procedural Biosafety Measures
ContentProcedural biosafety measures include:
- Biosafety measures taken during the Line Probe Assay (LPA) procedures
- Aerosol generation and preventive measures
Manipulations that produce aerosols in DNA Extraction Room are:
- Pipetting
- Centrifugation
- Vortexing
- Discarding micropipette tips after use
Measures to Prevent Aerosol Generation
- Always open and manipulate specimens and cultures in a certified Biosafety Cabinet (BSC).
- Work over absorbent material (sheet/ paper) soaked in a disinfectant.
- Use centrifuge safety cups/ aerosol tight rotor and open them in the BSC.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
LPA Biosafety Essentials: Waste Disposal
ContentBiomedical Waste Management is an essential biosafety component of TB containment laboratories. All infectious materials should be decontaminated, incinerated, buried or autoclaved.
The following materials are suitable for waste disposal by autoclaving:
- All positive TB cultures
- Mycobacterial cultures
- All infectious materials from TB-containment laboratories where mycobacterial culture is performed
In laboratories where the risk of infection with TB is low, plastic sputum containers, cartridges used for molecular analysis - Nucleic Acid Amplification Test (NAAT) and Line Probe Assay (LPA) - (CBNAAT cartridges, TrueNAT chips, used tips, droppers, reagents, other consumables) and wooden applicator sticks, should be removed from the laboratory in sealed disposal bags and incinerated.
Important considerations for waste disposal
- Any materials that are reused must be decontaminated with a suitable disinfectant or autoclaved before being removed from the laboratory.
- All infectious waste should be soaked in appropriate disinfectant (5% phenol/ 1% sodium hypochlorite) and discarded in the bio-safety disposal bins.
- The bio-medical waste should be segregated into containers.
- Colored plastic bags should be used as per the Programmatic Management of Drug Resistant TB, 2021 Guidelines, at the point of generation.
- The authorized person should maintain records related to the generation, collection, reception, storage, transportation, treatment, disposal or any other form of handling of bio-medical waste, for a period of five years.
- Waste should be disposed off according to the Bio Medical Waste Guidelines, 2019.
Resources
- Guidelines for PMDT in India, 2021.
- Central Pollution Control Board: Waste Management and Handling Rules.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen