LPA for DR-TB Diagnosis
ContentAs per the Integrated Drug Resistant Tuberculosis (DR-TB) Diagnostic Algorithm:
- Nucleic Acid Amplification Test (NAAT) are preferred for initial detection of Rifampicin (R) resistance.
- Line Probe Assay (LPA) test is preferred for detection of Isoniazid (H), Fluroquinolones (FQ) and second-line injectable (SLI) drugs resistance.
- When Rifampicin resistance is detected by NAAT (Figure):
- Offer first-line (FL) and second line (SL) LPA
- Direct LPA performed on smear positive specimen or indirect LPA is performed on culture isolate when smear is negative
- When Rifampicin resistance is not detected by NAAT (Figure):
- Offer FL-LPA to detect H resistance
- Direct LPA performed on smear positive specimen or indirect LPA is performed on culture isolate when smear is negative
- If H resistance is detected, SL-LPA is performed to check FQ resistance.
- When Rifampicin resistance is detected by NAAT (Figure):

Figure: Flow Chart to decide FL/SL-LPA after NAAT Results
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Line Probe Assay [LPA] in DR-TB Diagnosis
ContentAs per the Integrated Drug-resistant Tuberculosis (DR-TB) Diagnostic Algorithm:
- Nucleic Acid Amplification Tests (NAAT) are preferred for the initial detection of Rifampicin (R) resistance
- Line Probe Assays (LPA) are preferred for the detection of Isoniazid (H), Fluoroquinolones (FQ) and second-line injectable (SLI) drugs resistance.
When rifampicin resistance is detected by NAAT (see figure below):
- Offer first-line (FL) and second line (SL) LPA
- Direct LPA performed on smear positive specimen or indirect LPA is performed on culture isolate when the smear is negative
When Rifampicin resistance is not detected by NAAT (see figure below):
- Offer FL LPA to detect H resistance
- Direct LPA performed on smear positive specimen or indirect LPA is performed on culture isolate when the smear is negative
- If H resistance is detected; SL LPA is performed to check FQ resistance
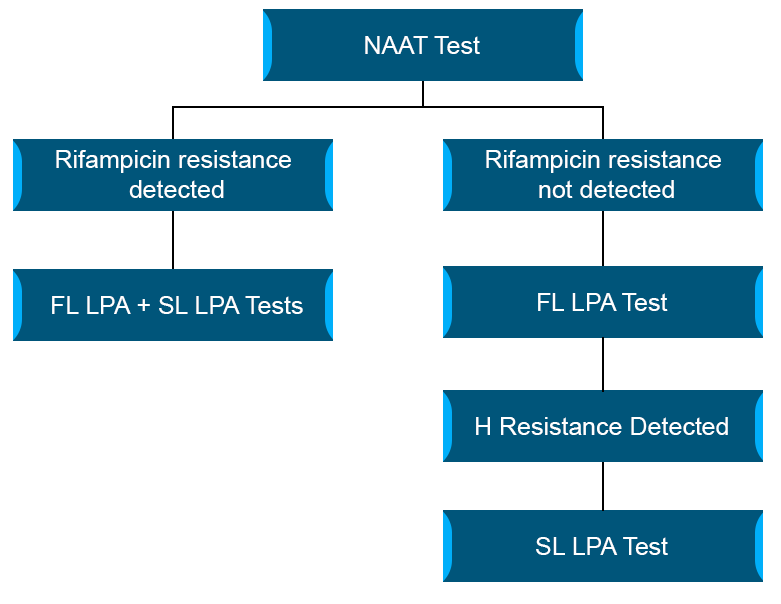
Figure: Flowchart to decide FL/SL-LPA after NAAT results
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
First Line LPA [FLLPA]
ContentFirst Line (FL) LPA
- Performed using GenoType MTBDR plus ver 2.0 kit
- GenoType MTBDR plus (Figure) identifies:
- Mutations in the Rif-resistance Determining Region (RRDR) of rpoB gene (from codon 505 to 533) to detect Rifampicin resistance
- Mutations in the inhA promoter (from -16 to -8 nucleotides upstream) and the katG (codon 315) regions to identify Isoniazid resistance.

Figure: GenoType MTBDR plus FL-LPA strip to determine Wild Type genes and mutations responsible for resistance to Rifampicin and Isoniazid; Source: GLI LPA Guidance Document.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Second Line LPA [SLLPA]
ContentSecond-line Line Probe Assay (SL-LPA) is a LPA to detect resistance to the second-line anti-TB drugs. This test is recommended for identifying TB patients with Multidrug-resistance (MDR) or rifampicin-resistance and those who can be placed on the shorter MDR-TB regimen.
The SL-LPA produces results in just 24-48 hours.
- It is performed using GenoType MTBDRsl Version 2.
- This is a DNA-based test that identifies genetic mutations in MDR-TB strains, making them resistant to fluoroquinolones and injectable second-line TB drugs.
- GenoType MTBDRsl Version 2 (Figure) identifies:
- Quinolone-resistance determining region (QRDR) of gyrA (from codon 85 to 96) and of gyrB (from codon 536 to 541) genes for detection of resistance to fluoroquinolones
- rrs (nucleic acid position 1401, 1402 and 1484), eis promoter region (from -37
to -2 nucleotides upstream, low level kanamycin resistance) for detection of resistance to SLI drugs.
- Single mutation in gyrA confers low level fluoroquinolone resistance.
- Concurrent mutations in gyrA or both gyrA and gyrB confer high level fluoroquinolone resistance.
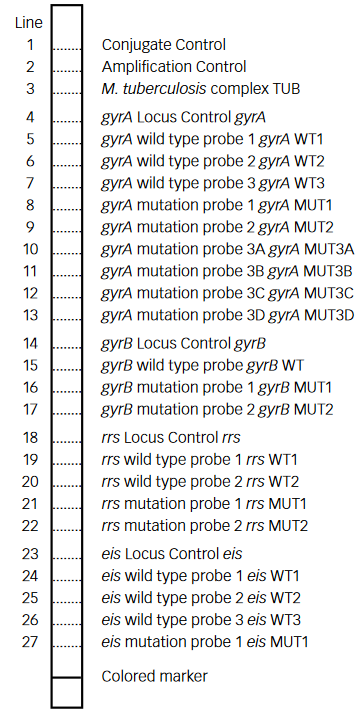
Figure: GenoType MTBDRsl Version 2 SL-LPA strip to determine Wild Type genes and mutations responsible for Resistance to Fluoroquinolones and Second-line Injectable Drugs; Source: GLI LPA Guidance Document.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen