-
CDST_LT: General concepts
FullscreenNeed for Specimen Collection and Transportation [SCT]
ContentThe National TB Elimination Programme (NTEP) has strengthened sputum collection and transport for laboratory testing by building capacity for decentralised collection.
- The provision for sputum collection and transport is used to establish linkages for giving diagnostic access to patients who cannot reach the health facilities.
- To increase access and coverage of services, designated sputum collection centres are also established for collecting and transporting sputum to the nearby laboratory.
Presumptive TB patients attending Peripheral Health Institutions (PHIs) other than Designated Microscopic Centres (DMCs) are either referred to the nearest DMC for sputum examination or their sputum specimens are collected and transported to the DMC as per guidelines.
- The patient is given these options as per their convenience to minimise the possible delay in diagnosis and initiation of treatment or avoid repeated visits by the patient.
- In case the patient is not able to travel to the DMC, then the spot sample is collected at the nearest health facility/ sputum collection centre/ sputum collection booths and transported to the DMC.
The need for sample collection can be categorized broadly into two categories:
1. When samples are sent to the DMC
- Some PHI/ sub-centres/ health and wellness centres function as sample collection centres.
- Sputum collected from referring health facilities needs to be transported to the nearest DMC.
2. When DMC has to collect and transport samples for testing/ follow-up/ resistance testing to higher laboratories
- If the Nucleic Acid Amplification Testing (NAAT) facility is not available at the health facility, the DMC may also need to collect and send the sample to the nearest linked facility for NAAT testing.
- If NAAT is available at DMC and Mycobacterium tuberculosis (MTB) is detected, the second sample needs to be transported to the Culture and Drug Susceptibility Testing (C&DST) lab. If the NAAT result suggests rifampicin-sensitive, a second sample is sent for First-line Line Probe Assay (FL-LPA) to look for H-mono/ poly resistance.
- Extrapulmonary samples have to be appropriately collected at the PHI/HF and transported for testing to NAAT/ culture facilities.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Specimen collection and transportation system minimises patient inconveniences and diagnostic delays.
True
False
1
Specimen collection and transportation system minimises patient inconveniences and diagnostic delays.
Yes Yes Process of SCT
Content- Good quality sputum collection is of paramount importance in tuberculosis diagnosis.
- Once a person is identified as presumptive TB, s/he is referred to the Designated Microscopy Centre (DMC) for sputum collection.
- For TB diagnosis two sputum samples are collected - one is the supervised ‘spot’ sample collected at the DMC (labelled Specimen A) and the other is the early morning sample collected by patient themselves at his/her home (labelled Specimen B).
- If the patient is coming from a long distance or s/he is unlikely to return to give the second specimen, two spot specimens may be collected with a gap of at least one hour.

Figure: Flowchart for Sputum Collection and Transport
Sputum Collection
- The (NTEP) request form required for the biological specimen examination need to be filled.
- The Lab Technician (LT) should instruct the patient to thoroughly rinse the mouth with clean water and demonstrate to inhale deeply 2-3 times and cough out the sputum from the depth of the chest into a sterile 50 ml sputum container, in a well-ventilated space.
- After collecting the sputum, close the lid of the containers and wipe the surface of the tube with 5% phenol to disinfect and allow it to air dry.
- Label the tubes with patient details, date and time of collection, specimen identification, lab no. using a permanent marker.
Sputum Transport
- The sputum collected should be transported immediately to the Nucleic Acid Amplification Testing (NAAT)/ Culture and Drug Susceptibility Testing (C&DST) laboratory. In case of any unavoidable delays, the sample should be refrigerated.
- The programme mandates triple layer packaging for the transport of the sputum specimens.
- Firstly, seal the joint between the cap and neck of the sputum containers with a parafilm strip (primary receptacle package).
- Wrap the sputum containers individually in absorbent cotton, place them in a zip lock pouch and secure them with a rubber band (secondary receptacle package).
- Fold and place the NTEP request form in another zip lock pouch.
- Place the zip lock pouch with sputum containers in a thermocol box along with two pre-frozen coolant gel packs and the zip lock pouch with the NTEP request form is placed on top.
- The dimensions of the thermocol box used for sputum transport are: thickness - 2.5 cm; Outer dimensions: length - 18.5 cm, breadth - 13 cm, height - 12 cm (without lid), height -14 cm (with lid); Inner dimensions: length - 14.5 cm, breadth - 8 cm, height - 12 cm (without lid), height - 13 cm (with inner part of lid).
- The coolant gel packs should be conditioned in the deep freezer in a temperature between -20 to -15o C for a minimum 48 hours to a maximum 72 hours before use so that they can maintain a temperature between 12-20o C for up to approximately 48 hours in tightly packed thermocol boxes while the average outside temperature is 35o C.
- Seal the thermocol box with duct tape and affix ‘To’ and ‘From’ address, biohazard sticker on the box (tertiary receptacle package).
- Weight of the fully packed consignment box should be up to 400 grams and the thermocol boxes and gel packs should not be reused.
- Transport the box through NTEP identified courier/ speed post service.
Resources
- Training Module (1-4) for Programme Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug-resistant TB in India, 2021.
- Module for Laboratory Technician, CTD, MoHFW, India, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test After collecting the specimen, the falcon tubes should be wiped and disinfected with which solution? 5% Iodine 5% Glycerine 5% Phenol 5% Sodium 3 After collecting the specimen, the falcon tubes should be wiped and disinfected with 5% phenol solution. Yes Yes NTEP mandates triple layer packaging for the transport of the sputum specimens
True False 1 Triple layer packaging prevents spills and leakages during transportation of sample. Yes Yes
-
CDST_LT: Good quality specimen collection
FullscreenAccepting the request for testing
ContentThree things are received by the Lab Technician- Sample for testing, request form, and request for test in Nikshay.
Accepting the request for testing includes the following steps:
1. The LT verifies details on the request form that has eleven parts.
- The first part contains details on the name of referring facility, name of the patient, complete address, age & gender of the patient, date of referral, type of presumptive TB, the key population to which the patient belongs and site of disease.
- The second part contains details of referring facilities, Nikshay ID, and the names of State, district and TB units.
- The third portion is for the diagnosis and follow-up of TB.
- The fourth portion is for the diagnosis and follow-up of drug-resistant TB.
- The fifth portion is to indicate the required tests with the details of the person requesting the test.
- Parts six to eleven are used for reporting test results.
2. The LT verifies the quality of the sample received.
3. LT captures details on the patient, reasons for testing, test requested, and the visual appearance of the sample in the TB Laboratory register.
4. LT verifies the test request generated in Nikshay against the test ID requested (Figure).

Figure: Test Details Added in Nikshay by the Referring Health Facility; Source: Guidelines for PMDT in India, 2021.
5. LT initiates the test requested.
Resources
- Guidelines for PMDT in India, NTEP, 2021.
- Training Modules (1-4) for Programme Managers and Medical Officers, NTEP, 2020.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
For all new presumptive TB cases, Nikshay ID is generated by the referring facility.
True
False
1
For all new presumptive TB cases, Nikshay ID is generated by the referring facility.
Yes
Yes
Spot and early morning sputum sample
ContentPresumptive pulmonary TB patients are subjected to sputum smear microscopy (Ziehl Neelsen (ZN)/ Florescence Microscopy (FM)). Two consecutive sputum specimens will identify the vast majority (95–98%) of smear-positive TB patients
Two specimens are collected:
- One Spot and one early morning sample OR
- Two supervised spot specimens collected at least one hour apart, and smears made from both the samples.
If one or both smears are positive, the patient is diagnosed as a microbiologically confirmed pulmonary TB case.
- The spot specimen collected is labelled as 'a'.
- While the patient is given a labelled container with instructions to cough out sputum into the container early in the morning after rinsing the mouth with water. This is the early morning specimen. This is labelled as specimen 'b'.
- If the health facility is not a Designated Microscopy Centre (DMC), then the patient is given a sputum container with the instructions to collect an early morning specimen and go with the sputum specimen to the DMC where the spot specimen can be collected.
- In case the patient is not able to travel to the DMC, then the spot specimen could be collected at the nearest health facility or sputum collection centre and transported to the DMC.
- These two samples should be collected within a day or two consecutive days.
- Two supervised spot samples may be collected one hour apart if patient is too sick, coming from a long distance or likelihood of not giving a second sample is significant.
- Collection of early morning specimens is preferred because of the overnight accumulation of secretions. However, spot samples collected at any time for patients is also suitable if productive sputum is expectorated after deep cough.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The spot specimen collected for sputum microscopy is labelled as 'a'.
True
False
1
The spot specimen collected for sputum microscopy is labelled as 'a'.
Yes
Yes
Educating patient on Sputum collection and dispensing Sputum cup
ContentEducating patients on collection is essential to have good quality sputum. The healthcare worker (HCW)/ medical officer (MO) or the laboratory technician (LT) can educate patients on how to collect and dispense sputum.
The HCW/MO/LT provides a new sputum cup with the Laboratory Serial Number written on its side to the patient. They should explain that sputum should be collected in an open place or in a well-ventilated room; it should not be collected in closed rooms, toilets and ill-ventilated rooms
A specimen collected under supervision is likely to yield better results. Supervising person has to demonstrate how to collect good sputum and dispensing it:
- Using a laboratory sputum cup, demonstrate how to open the lid of the specimen container and place it conveniently within their reach, so they can close it immediately after collecting sputum and also how to screw the cap on the cup tightly so it doesn't leak.
- Demonstrate how to bring up sputum, beginning with rinsing their mouth as food particles may give false positive results.
- Demonstrate deep inhalation (2–3 times) and let the patient know that this will initiate the cough reflex in most individuals.
- Demonstrate how individuals can place their palms on the waist, squat or sit and continue deep breathing again. Tapping or thumping of the back
may encourage expectoration (Sitting and placing hands on the waist fixes the shoulder and pelvic muscles and brings the intercostal muscles of ribcage and diaphragm into action) - After deep inhalation and coughing deeply, the sputum should come up in their mouth. The sputum is retained in the mouth and allowed to fall from their tongue into the pre-labeled container. Patient should be encouraged not to spit into the container. The patient can also be encouraged to cough directly into the cup.
- The patient’s mouth should not touch the container and the patient must ensure that sputum does not touch the outside of the container.
- The patient should not open the sputum cup till they are ready to use it
- They should not rub off the number written on the side of the container
- They should not touch the inside of the container with their fingers or tongue while collecting sputum
The person collecting the specimen should make sure that no one stands in front of the individual who is trying to cough up the sputum. When an individual has only coughed up saliva or has not coughed up at least 2 ml of sputum, they should be encouraged to give good specimen that is of appropriate quantity.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
When collecting sputum into containers, which of the following should not be done?
Cough deeply and directly into the sputum cup.
Sputum can sometimes touch the outside of the container.
Always close the lid tightly, after putting the sputum into the cup.
None of the above
2
When dispensing sputum into the sputum container, sputum must never touch the outside of the container.
Yes
Yes
Steps to Ensure a Good Quality Sputum Sample
ContentThe Healthcare Worker (HCW) needs to carefully explain how to collect a good quality sputum specimen. He/she needs to demonstrate how to bring up sputum from the chest, what a good sputum specimen looks like, and the quantity of sputum required.
Characteristics of a Good Sputum Sample
- Thick (semi-solid) muco-purulent (yellowish) in consistency, coughed out deeply from the lungs
- Sufficient in amount i.e., 2 to 5 ml (or enough to cover the size of a fingernail at the bottom of the container)
- It should not be blood-stained (brownish colour) as far as possible.
Steps to Ensure Good Quality Sputum Sample
- Explain to the patient the characteristics of sputum - that it is thick and mucoid as compared to saliva which is thin and watery.
- The patient should preferably rinse his/her mouth to get rid of any food particles which may give false-positive results.
- One should demonstrate to the patient by action how s/he should take deep breaths and bring up the sputum.
- The patient is instructed to inhale deeply (2–3 times), which will initiate the cough reflex in most patients.
- The sputum is retained in the mouth and spit into the pre-labelled container without spilling.
- Some patients may not be able to expectorate with deep breathing, in which case HCW should demonstrate to them how they should place their palms on the waist, squat or sit and continue deep breathing again.
- Tapping or thumping of the back may encourage expectoration. (Sitting and placing hands on the waist fixes the shoulder and pelvic muscles and brings the intercostal muscles of the ribcage and diaphragm into action).
- When a patient has only coughed up saliva or has coughed up less than 2 ml (the size of a fingernail at the bottom of the container) of sputum, the patient should be encouraged to provide a better specimen.
Resources
-
CDST_LT: Specimen transport
FullscreenTransportation of Biological Specimens
ContentAll efforts must be made to have decentralized local arrangements for transporting the specimens to the Designated Microscopy Centre (DMC)/ Culture Drug Susceptibility Test (CDST) and molecular Nucleic Acid Amplification Test (NAAT) and Line probe assay (LPA) labs through human volunteers/courier/speed post. The specimens collected should be carefully packed in a box to avoid any spillage or contamination.
The following points must be considered for the transportation of biological specimens:
- Samples need to be transported to DMC or CDST laboratory within 72 hours (in a cold chain if sent to CDST and molecular laboratories)
- The health care worker must inform and coordinate with the sample transportation agency to transport the sample to the necessary laboratory
- The accompanying dispatch list present along with the biological specimen must tally with the total number of sputum specimens collected, and must specify the details of the referring health facility collecting the specimen
- The Specimen Identification Number on the specimen container and the accompanying dispatch list must match
- For each patient, one biological requisition form is required and all necessary details should be filled
- All specimens transported to the laboratory must be accompanied by the request form in hard and soft copy formats
- Triple packing system should be utilized for transportation
- The box containing the specimen samples to be transported to the National TB Elimination Program (NTEP) certified laboratory should be labelled with a “BIO-HAZARD” sticker
- The date of dispatch must be marked by the health worker on the dispatch list and the same must be attached outside the box containing the specimen
Triple Layer Packaging
As per NTEP guidelines, the sputum specimen is packaged in triple layers for transportation to avoid spills and leakage and pose no hazard to the transporter. This includes:
1. Primary Container: This is a watertight, leak-proof, unbreakable tube containing the specimen. The tube is packaged with enough absorbent material to absorb all fluid in case of breakage or leakage.


Figure 1: Primary container; Source: PMDT Guidelines, 2021
2. Secondary Packaging: This is watertight, leak-proof packaging to enclose and protect the primary container. Several primary containers may be placed in one secondary packaging.


Figure 2: Secondary Packaging; Source: PMDT Guidelines, 2021
3. Outer Packaging: Secondary packaging is placed in rigid outer packaging to protect the contents from physical damage during transport. Gel packs should be kept inside the box to maintain temperature along with suitable absorbent/ cushioning material

Figure 3: Outer Packaging; Source: PMDT Guidelines, 2021
The transporter needs to ensure the following:
- The outer packaging is not damaged and is properly sealed
- Biohazard label is pasted on the outer packaging
- “From” and “To” addresses are clearly labelled
- Contact details (name, phone no.) of the receiver is pasted
- Upright symbol (

 ) is pasted appropriately
) is pasted appropriately - The temperature is maintained at 2-80 C for the entire transportation period
- Transport at the earliest (to reach the destination within 72 hours)
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Modes of Transportation in SCT
ContentAll efforts must be made to have decentralised local arrangements for transporting the specimens to the TB detection centre (TDC). If a proper transport mechanism for collected specimen is in place, it spares the patients from travelling to the laboratory.
The sputum sample is packaged in triple layers in such a manner that it arrives at the destination in good condition and presents no hazard to the transporter.
Transporter/ personnel transporting the sample should be sensitized by the National TB Elimination Programme (NTEP) staff prior to engagement.
- Sensitisation would be provided on Symptoms of TB disease and its transmission, precautions to be taken to prevent exposure, hand hygiene requirements and spill management.
The different modes of sample transport include:
1. Post: Postal department services available pan-district can be engaged to transport sputum samples.
2. Courier: Local logistics courier companies can also be identified and hired to transport samples.
3. Volunteers/ human carriers: Community volunteers/members of NGOs can be trained as human carriers to collect and transport samples.
District TB Centre or Medical Officer TB Control (MO-TC) should ensure feasibility and financial measures required for .such arrangements for sputum transport.
Systems can be established for transportation of various biological samples (not only TB) from referring centre/peripheral centre to laboratories in a hub and spoke model in a integrated manner.
Resources
- Specimen Transportation - A How-to Guide.
- Health and Safety Guidelines for Staff/ Workers involved in Sputum Transportation, CTD.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Sputum can be transported through post/ courier/ human carriers.
True
False
1
Sputum can be transported through post/ courier/ human carriers.
Yes Yes Requirements for Packaging a biological Specimen
ContentPeripheral Laboratories in the NTEP need to send biological samples (such as sputum) to nearby Culture and Drug Susceptibility Testing (CDST) laboratories for advanced testing (eg Drug Resistance Testing). The samples need to be safely packaged and transported such that there is no spillage or contamination.
The items required for safe packaging biological specimens are:
- Falcon tubes
- Three-layer packing materials like thermocol box
- Ice gel pack (pre-frozen at -20oC for 48 hours)
- Request for CDST forms
- Polythene bags
- Tissue paper roll for absorbent packing
- Parafilm tapes
- Brown tape for packing the thermocol box
- Permanent marker pen
- Labels with 'To' and 'From' address and blank labels for sample details
- Biohazard sticker
- Scissors
- Spirit swab
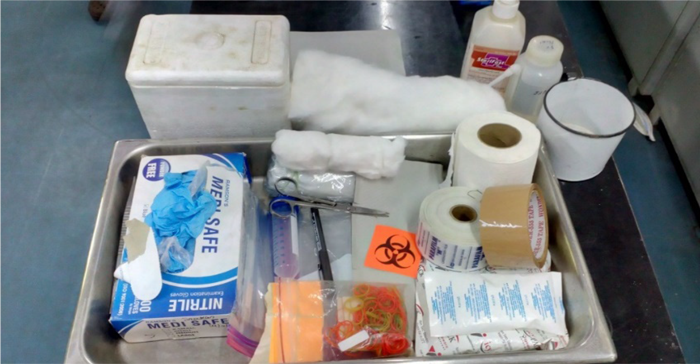
Figure: Items needed for packaging of biological specimens for CDST laboratory; Source: Guidelines for Programmatic Management of Drug Resistant TB in India, 2021
Video fileResources
SOP for Packaging Specimens during the Transportation of Biological Specimens for TB Diagnosis
ContentStandard Operating Procedure (SOP) for Packaging Specimen during the transportation of biological specimens for TB diagnosis, also known as Triple Layer Packaging, is as follows:
The Lab Technicians (LTs) at the Designated Microscopy Centres (DMCs) should be trained to carefully pack the sputum samples in the cold box to avoid spillage of the samples. Before packing, personal protection measures such as wearing hand gloves (double gloves preferred), goggles and masks are to be followed by the LTs to prevent contracting the infection.
Table: Steps in Standard Operating Procedure (SOP) for Packaging Specimen during Transportation for TB Diagnosis
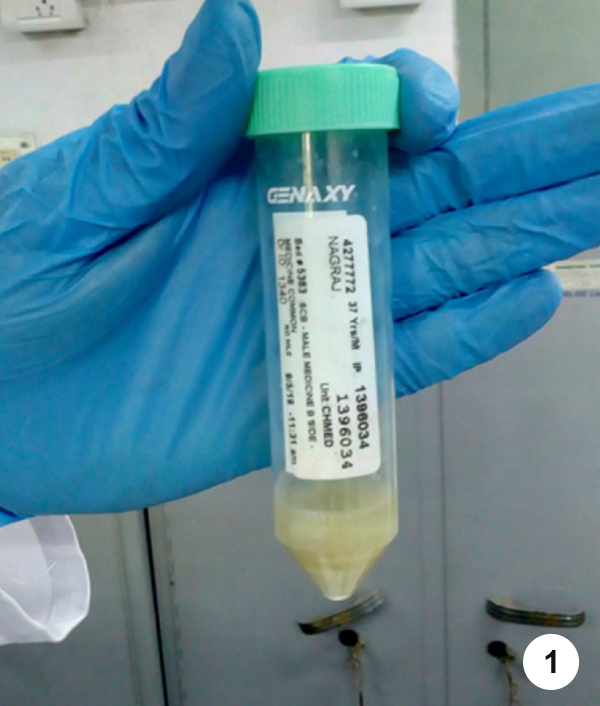
- Step 1. Make sure that the specimen collection tube is tightly closed after the sample has been collected from the patient.

- Step 2. Wipe the outer surface of the 50 ml conical tube with 5% phenol followed by absorbent tissues and allow it to air dry.

- Step 3. Write the patient details on the opaque area (white area) of the specimen collection tube using a permanent marker pen, clearly in capital letters.
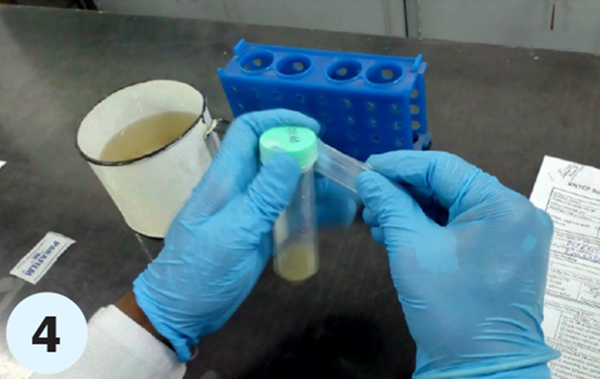
- Step 4. Cut the parafilm strip and wrap one of the strips at the joint between the cap and the neck of the specimen collection tube such that a secured seal is formed. (Primary receptacle/ package

- Step 5. Open the absorbent cotton roll and spread it out on the workbench; separate the cotton into two equal layers. Roll the specimen collection tube containing the sample tightly in the absorbent cotton such that the tube is covered completely.
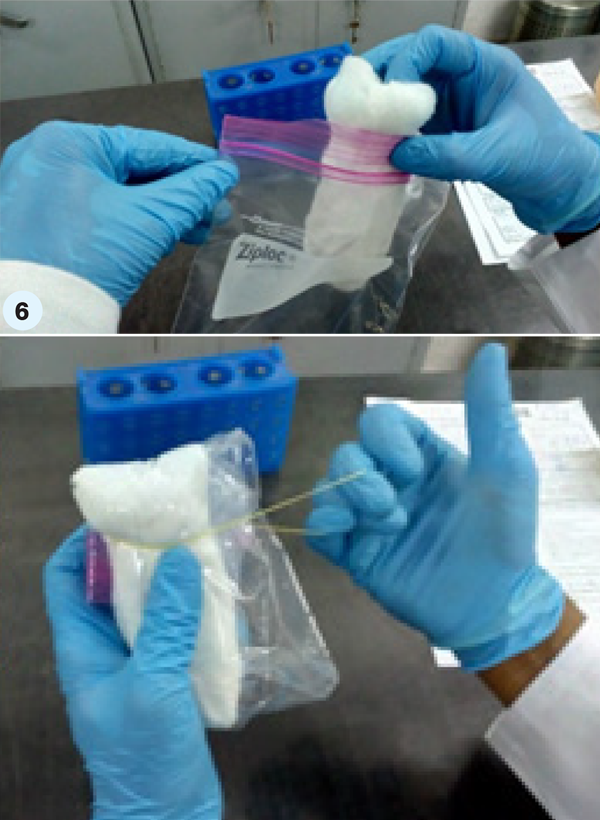
- Step 6. Put this roll containing the specimen collection tube into the ziplock pouch. Roll the whole into a tight bundle, ensuring that there is no air in the pouch. This bundle should be secured with rubber bands. (Secondary receptacle/ package)


- Step 7. Repeat steps 5–7 for the second sample of the patient.

- Step 8. Insert the Test Request form printed from Nikshay into the ziplock pouch after ensuring that the details on the form and the sample tubes match, with the writing facing outside (details visible through the package). Seal the ziplock on the pouch.

- Step 9. Place the cooled gel packs into the thermocol box, place the sample tubes packed in ziplock pouches on the frozen gel packs (frozen for 48 hours at -40°C) and also keep the pouch containing the Test Request form printed from Nikshay on top. Stick the BIOHAZARD sign over the lid and “To and From” stickers on the exterior of the thermocol box or box used to pack the specimen. Close the lid of the box and wrap it tightly with brown duct tape. (Tertiary receptacle/ package)

- Step 10. Complete the ‘From’ and ‘To’ addresses on the stickers, using a permanent marker pen.
The LT of the DMC should promptly inform the sample transport agency like a courier/ speed post service, or a human carrier to collect and transport the samples.
Video fileResources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug-resistant TB in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Specimen Carriers
ContentTechnical specifications for TB diagnosis specimen carriers are as follows:
Thermocol Box:
- Thickness of box - 2.5 cm
- Outer dimensions: Length - 18.5 cm, breadth - 13 cm, height - 12 cm (without lid), height - 14 cm (with lid); Inner dimensions: Length - 14.5 cm, breadth - 8 cm, height - 12 cm (without lid), height - 13 cm (with inner part of lid).
Gel Pack:
- Number of gel packs required: 2
- Weight of fully packed consignment box: 400 grams
- Gel packs maintain a temperature between 12-20ºC for up to approximately 48 hours in tightly packed thermocol boxes (average outside temperature 35ºC)
- Conditioning in the deep freezer (temperature between -20 to -15ºC) for a minimum 48 hours to a maximum 72 hours before use, is required.
This is a one-time use carrier since the thermocol boxes and the gel packs are not reused.

Figure: TB Diagnostic Specimen Carrier - Thermocol Box with Gel Packs; Source: NTEP Guidelines for Programmatic Management of Drug Resistant TB in India, 2021
Resources
- Training Module (1-4) for Program Managers and Medical Officer, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug Resistant TB in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Technical Specifications for Labelling Specimen Transport Carriers
ContentThese are the technical specifications for labelling specimen transport carriers:
- Specimen containers need to be labelled legibly with details such as the patients’ name, date and time of specimen collection, TB detection centre/ District Tuberculosis Centre (DTC), lab no., specimen A or B.


Figure: Details to be filled on the specimen containers

- Dispatch list with details of each specimen transported and the request form for examination of biological specimen for each specimen should be put in an envelope and attached to the outside box.
- As per the national guidelines for biomedical waste management, the containers used for transporting specimen samples to the National TB Elimination Programme (NTEP) - certified laboratory should be labelled with a “BIOHAZARD” sticker.

- Other than the Biohazard sticker it is mandatory to add 'To' and 'From' stickers on the specimen transportation carrier and fill in the necessary details.
- Specimen transport carriers should be labelled legibly with all the necessary details as listed above.
- A specimen may be rejected at the receiving laboratory if the specimen transport carriers are unlabelled or mislabelled.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Cold Chain Requirement for the Transport of TB Diagnostic Specimens
ContentA cold chain is a system of transporting and storing TB specimens at optimum temperature while being transported from the peripheral health institutions to the diagnostic labs to reduce the growth of contaminating endogenous respiratory organisms.
Cold chain requirements for transportation of TB diagnostic specimens are (Figure 1):
- A thick thermocol box (one-time use box), which has a thickness of about 2.5 cm. Outer dimensions of the box - Length: 18.5 cm, Breadth: 13 cm, Height: 12 cm. Inner dimensions of the box - Length: 14.5 cm, Breadth: 8 cm, Height: 10 cm.
- Two gel packs to maintain a temperature between 12-20°C for up to approximately 48 hours in tightly packed thermocol boxes (average outside temperature 35°C). Gel packs to be conditioned in the deep freezer (temperature between -20 to -15°C) for a minimum of 48-72 hours before use.

Figure 1: Technical specifications of transport box for sputum specimen transportation in cold chain
Specimen Transport Steps - Cold Chain (Figure 2)
- Place the gel pack into the thermocol box.
- Place the sample tubes (in zip-lock pouches) on the frozen gel packs.
- Keep the pouch containing the Test Request form on top before placing another gel pack on top.
- Close the lid of the box and wrap tightly with brown duct tape to maintain the cold chain.

Figure 2: Steps for specimen transportation in cold chain
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Dispatching the sample to a C&DST Lab
ContentFor presumptive Drug-resistant TB (DR-TB) patients’, the health facility staff arranges for specimen collection from patient, packs samples as per the Standard Operating Procedure (SOP) for triple layer packaging and dispatches it for transportation in cool chain to the linked Culture and Drug Susceptibility Testing (C&DST) laboratory.
All necessary materials for specimen collection and modality for transportation need to be made available/ arranged at the Designated Microscopy Centre (DMC)/ Peripheral Health Institute (PHI) by the District TB Officer (DTO).
Steps in sample dispatch include:
- Add test details in Nikshay (Figure) to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
- This will enable instant online intimation about the upcoming specimen to the C&DST laboratory.
- Complete test request form for biological specimen by adding:
- Patient information
- Details on name and type of referring facility
- Health establishment ID
- Reason for testing and test requested
- Patient ID and Test ID
- Put the appropriate address of receiving C&DST laboratory and address of health facility form where samples are dispatched on the transportation box.
- Lab Technician (LT) of the health facility should promptly inform the transport agency (courier/ speed post service) or human volunteers to collect and transport the samples.

Figure: Adding Test Details in Nikshay for Diagnosis of DR-TB; Source: Nikshay Diagnostics Training Content.
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, NTEP, 2020.
- Guidelines for PMDT in India, 2021.
- Nikshay Zendesk, Nikshay Knowledge Base, Diagnostics.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Test details are added in Nikshay to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
True
False
1
Test details are added in Nikshay to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
Yes
Yes
- Add test details in Nikshay (Figure) to generate test requests (Test ID) for the patient’s episode ID in Nikshay.
-
CDST_LT: Specimen receipt
FullscreenProcess of Specimen Receipt at the NTEP Laboratory
ContentThere is a specific procedure prescribed under the National TB Elimination Program (NTEP) during receipt of the specimen for NTEP Laboratory.
At the laboratory, all specimens need to be received in the registration area.
To minimize the risk of infection, the following procedures should be applied:
- The specimen box received needs to be opened only in a biosafety cabinet inside the laboratory. (Do not open on an open bench at the lab reception).
- Before opening the package, one should inspect the delivery box for signs of breakage or leakage; if there is gross leakage evident, one needs to discard the package following biomedical waste management.
- If on gross inspection there is no leakage, one needs to proceed with opening the sample.
- One needs to open the package carefully and re-check for any leakage. In case of leakage, again it should be discarded (the entire contents) following biosafety precautions. The laboratory needs to inform about rejection/ leakage of samples to the respective District TB Officers immediately to enable re-collection of specimens.
- Laboratory needs to check the labelling of specimens in the specimen container and the test request form.
- It should register the samples in Laboratory Information Management System (LIMS) and proceed for processing by the appropriate method.
- Each laboratory needs to document the date of the receipt of the specimen, patient name, age, sex and address, the name of the referring health centre, the reason for testing and volume of the specimen in the Culture & Drug Susceptibility Testing lab register.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Criteria for Acceptance and Rejection of TB Specimens at TB Diagnostic Laboratories
ContentThe criteria for acceptance and rejection of TB specimens at TB Diagnostic Laboratories are listed below:
Acceptance Criteria:
- Sample received in triple layer with no leakage
- Sample is without any blood and food particles
- Sample is received within 48 hours expected timeline after collection in a cool chain
- Adequate label and information about the specimen
- Identical name on the specimen and in the test request
- Sample is in adequate quantity
Rejection Criteria:
- Unlabeled or mislabeled specimens
- Specimen sent without the test request ID in Nikshay
- Mismatch in the name on the specimen and the test request in Nikshay
- If the container is full up to the lid with the specimen
- Sample is not collected in an appropriate container
- Specimen container is broken
- Leakage of specimen
- Sample received after two days of collection
Resources
- Training Module (1-4) for Program Managers and Medical Officer, NTEP, MoHFW, 2020.
- Guidelines for Programmatic Management of Drug Resistant TB in India, 2021.
Kindly provide your valuable feedback on the page to the link provided HERE
Precautions to be Undertaken for TB Diagnostic Specimen Collection, Transport and Receipt
ContentAll healthcare workers need to take the following precautions while undertaking the TB diagnostic specimen collection, transportation and receipt of samples as TB is an airborne infection:
- There is the risk of transmission of tuberculosis infection occurring in health care facilities, including the laboratory when patients remain undiagnosed and untreated for tuberculosis.
- The specimen needs to be collected in an aseptic environment, preferably in open space.
- The collection area should be well-ventilated with adequate air exchanges (>12 per hour).
- The Information, Education and Communication (IEC) material related with cough etiquette needs to be displayed in all TB related setting including Designated Microscopy Centres/ Nucleic Acid Amplification Testing Centres/ Culture Drug Susceptibility Testing laboratories.
- Use of closed room, toilet and Outpatient Department area should be avoided while collection of sputum sample by a patient.
- The slides once used should not be reused; same is applicable for thermocol boxes.
- The contaminated materials should be safely disposed off.
- The Triple Layer Packaging needs to be monitored at all levels; at point of exit and point of reception.
- Leakage and broken thermocol boxes should be avoided and samples should be discarded in case of leakage.
- While transportation, cold chain needs to be maintained.
- The sample handlers should use proper personal protection equipment, including gloves, masks and gowns.
- Use of mask, preferably N95, while receipt of samples and during processing in laboratories.
- Healthcare workers should be provided with adequate waste disposal bins as per Biomedical Waste Guidelines and it should be utilized on regular basis.
- Soap, sanitizer and handwash should be available to all dealing with collection, transportation and receipt of samples.
- Those with symptoms suggestive of TB should not be involved in these key activities and should undergo a complete diagnostic workout.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Receiving a biological specimen at the Laboratory
ContentBiological specimen/ samples collected on reaching a TB testing laboratory needs to be formally received. The sample may be handed over by agents(couriers, health staff/ volunteers, patient representatives) or by patients themselves. The formal receipt of sample enables further processes such as testing and communication of results back to the patient. If the sample is successfully received, the appropriate testing process is initiated using the sample, else it is rejected and a fresh sample requested.
The designated Lab Technician (LT) at the Laboratory is responsible for receiving the sample. To initiate the receipt, the sample along with the patient, or the sample along with accompanying test request is required. If it is available, following are the steps to complete sample receipt:
- Compare patient information (Patient Name and Patient ID / Sample ID/ Test ID) to the test request that has been made.
- This is performed by searching the patient ID in Nikshay under the Diagnostics menu and comparing and matching the patient details and the label on the sample.
- If a physical form (Request for examination of Biological specimen for TB) is available, the details on the sample label and form needs to be compared and matched as well.
- If the details are matched the LT then checks the quality (mucopurulent, non-blood stained), quantity (adequate to perform the requested test) of the sample and ensures that there is no leakage.
- If the above checks are passed, then the LT Accepts sample. If not the LT rejects sample with a request to get a new sample. The rejection of the sample and request to obtain a new sample is recorded on Nikshay and is communicated to the patient and the person who requested the test.
- To register the receipt by accepting/Check in the sample for testing in Nikshay. The relevant information may also be captured in the TB Laboratory Register. Ensure that the Test ID (if not already present) / Lab serial number from the Lab register is labelled on the sample container.
Sample receiving in Nikshay

Figure: Sample Journey Tracking in Nikshay; Source: Nikshay Diagnostic Training Content.
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, NTEP, 2020.
- Nikshay Zendesk, Nikshay Knowledge Base, FAQs.
- Nikshay Zendesk, Nikshay Knowledge Base, Diagnostics.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The process of receiving a sample does not involve the following
Requesting a test
Matching test request information and container label.
Checking in a sample on Nikshay
Rejecting an inappropriate sample. 1 Request for testing and sample transportation to the laboratory has to happen before a sample can be received.
Matching request information and checking in a sample on Nikshay and rejecting inappropriate sample are steps in the receiving process.
Yes Yes - Compare patient information (Patient Name and Patient ID / Sample ID/ Test ID) to the test request that has been made.
Storing a sputum sample
ContentStorage conditions of sputum sample can effect the test results.
- Sputum samples should be transported to the laboratory as soon as possible after collection.
- It is the responsibility of Laboratory Technician (LT) and Senior TB Laboratory Supervisor (STLS) to ensure proper storage and transport of sputum specimens.
- Sputum is stored to preserve the specimen quality.
- The stored sputum samples should not be frozen.
Storage of Sputum Samples
For microscopy
- For smear microscopy, sputum specimens should be examined on the same day and not later than 2 days after collection.
- If delay is unavoidable, the sputum collected should be stored in a cool place/ refrigerated at 4°C to inhibit the growth of unwanted microorganisms.
- Stored sputum samples should be protected from light and heat to prevent liquefaction of the sample, else it makes the selection of mucopurulent part of the sample difficult.
- Samples received over holidays/weekends should be stored in a cool place/ refrigerated at 4°C.
For liquid culture
- Sputum should be stored in a cool place/ refrigerated at 4°C to inhibit the growth of unwanted microorganisms; not later than 3 days after collection.
For molecular tests - Nucleic Acid Amplification Test (NAAT) and Line Probe Assay (LPA)
- Sputum must be stored by refrigerating at 4°C to inhibit the growth of unwanted microorganisms and transported in cool chain to the nearest molecular laboratory. It should not be stored beyond one week at 4°C.
Resources
Technical and Operational Guidelines; Chapter 3: Case finding and diagnosis strategy
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The sputum collected expecting a time delay for processing should be stored in cool place/ refrigerated at 4°C.
True
False
1
The sputum collected expecting a time delay for processing should be stored in cool place/ refrigerated at 4°C.
Yes
Yes
Fullscreen

