Preparation of Patient-wise Boxes [PWB] for DR-TB Patients
ContentUnder the National TB Elimination Program (NTEP), Drug-resistant TB (DR-TB) patients receive standardized DR-TB treatment boxes (called Patient-wise Boxes - PWB) for each month. These boxes are either with the health facility nearest to DR-TB patients (DOT centre) or with the treatment supporter who is involved in the care of DR-TB patients.
Steps involved in the preparation of second-line drug boxes for DR-TB patients:
- NTEP State Drug Store (SDS) receives quality-assured second-line drugs from the centre through the General Medical Stores Depot (GMSD) and/or Central Medical Services Society (CMSS).
- The loose medicines are utilized for individual patient-wise box creation at the SDS/DDS by the store staff.
- Each patient-wise second-line drug box is prepared according to the NTEP standard box preparation guidelines utilizing the logistics, labels and drugs, and the same details are entered in Ni-kshay Aushadi for maintaining accountability of all drug boxes.
- Each patient-wise box needs to clearly mention the batch number of different drugs it contains, the expiry of the drug box and individual drugs, and any specific instructions that need to be followed, e.g., temperature, humidity or local storage.
- Second-line drug regimen box for INH mono/ poly regimen can be prepared at the district level considering its 6-month duration with the selected 4 drugs while Bdq/ Dlm regimen-based boxes are usually prepared at SDS and provided to District Drug Stores (DDS).
The exercise of preparing the standard patient-wise boxes is done at the NTEP DDS under the guidance and supervision of the District TB Officer (DTO). In such cases, drugs to the district will be supplied in loose form through Nikshay Aushadhi.
Resources
Constituents of Monthly Patient-wise Boxes [PWB] for Shorter Oral Bedaquiline-containing MDR/RR-TB Regimen
ContentThe patient on shorter oral Bedaquiline-containing multi-drug resistant (MDR)/rifampicin resistant TB (RR-TB) regimen shall be put on Type A and Type B box when initiated on treatment on a monthly patient wise box (PWB). Bedaquiline (Bdq) needs to be issued separately and stopped after 6 months. The patient should be provided with only Type A boxes when started on continuation phase (CP).
Table 1: Constituents of monthly-type A and B PWB of shorter oral Bedaquiline-containing MDR/RR-TB regimen; Source : Guidelines for PMDT, India 2021, p.132-133 TYPE A BOX
(USE IN IP AS WELL AS CP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin#
250/500 mg
250 mg *30tab
250 mg *30tab + 500 mg *30tab
500 mg *60tab
500 mg *60tab
Tab. Clofazimine
50/100 mg
50 mg *30tab
100 mg *30tab
100mg *30tab
100 mg *60tab
Tab. Ethambutol
400/800 mg
400 mg *30tab
800 mg *30tab
400 mg *30tab + 800 mg *30tab
800 mg *60tab
Tab. Pyrazinamide
500/750 mg
750 mg *30tab
500 mg *30tab + 750 mg *30tab
500 mg *60tab + 750 mg *30tab
500 mg *30tab + 750 mg *60tab
Tab. Pyridoxine
50/100 mg
50 mg*30tab
100 mg*30tab
100 mg*30tab
100 mg*30tab
Type B Box
(Use in IP)
Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Isoniazid
100/300 mg
300 mg *30tab
300 mg *60tab
300 mg *90tab
300 mg *90tab
Tab. Ethionamide
125/250 mg
125 mg *30tab + 250 mg *30tab
250 mg *60tab
250 mg *90tab
250 mg *120tab
Bedaquiline bottle
100 mg
1 Jar (Jar of 188 tablets for full course)
# When moxifloxacin prescribed under exceptional condition instead of levofloxacin, the modified box with moxifloxacin (normal dose) can be prepared from standard box at district drug store (DDS)
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Constituents of Patient-wise Boxes [PWB] for Isoniazid [H] Mono/Poly DR-TB Regimen
ContentStandard Patient-wise Boxes (PWBs) are constituted for TB patients initiated on Isoniazid (H) Mono/ Poly Drug-resistant TB (DR-TB) regimen.
This regimen has no segregation in terms of Intensive Phase (IP) or Continuation Phase (CP), hence drugs are provided in a single type of PWB.
Table: Constituents of standard PWB (6 months) of H mono/ poly DR-TB regimen; Source: Guidelines for PMDT, India 2021, p134
STANDARD PWB
CONTINUE FOR COMPLETE TREATMENT Drugs
Strength
16-29 kg
30-45 kg
46-70 kg
>70 kg
Tab. Levofloxacin
250/ 500 mg
250 mg *180 tab
250 mg *180 tab + 500 mg *180 tab
500 mg *360 tab
500 mg *360 tab
Tab. Rifampicin
150/ 300/ 450 mg
300 mg *180 tab
450 mg *180 tab
300 mg *360 tab
300 mg *360 tab + 150 mg *180 tab
Tab. Ethambutol
400/ 800 mg
400 mg *180 tab
800 mg *180 tab
400 mg *180 tab + 800 mg *180 tab
800 mg *360 tab
Tab. Pyrazinamide
500/ 750 mg
750 mg *180 tab
750 mg *180 tab + 500 mg *180 tab
750 mg *180 tab + 500 mg *360 tab
750 mg *360 tab + 500 mg *180 tab
Tab. Pyridoxine
50/100 mg
50 mg *30 tab
100 mg *30 tab
100 mg *30 tab
100 mg *30 tab
*No separate box for IP and CP.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Central TB Division, MoHFW. Training Modules (5-9) for Programme Managers & Medical Officers, 2020.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, RNTCP.
- Technical and Operational guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Guidelines for Issuing Bedaquiline and Delamanid to the Patient
ContentLoose drugs will be supplied to the Nodal (N)/ District DR-TB Centre (DDR-TBC) from the State Drug Stores (SDS)/ District Drug Stores (DDS) as per the consumption pattern of the previous month and requested via Nikshay Aushadhi.
- On discharge, the patient will be handed over the drugs for one month of consumption for transit.
- The senior DR-TB TB-HIV supervisor under the guidance of the N/DDR-TBC, shall guide the senior treatment supervisor (STS) for arranging the treatment supporter.
- The entire course available with the patient shall be handed over to the treatment supporter.
- For the provision of newer drugs, the table below shows the protocol to be followed for different regimens.
Table: Protocol for New Drug Provision for DR-TB Patients
PATIENT CATEGORY PROTOCOL Patient put on Bedaquiline (Bdq)-containing regimen The entire bottle of Bdq (188 tablets) shall be earmarked for each enrolled patient and handed over to the treatment supporter under the supervision of the senior DR-TB TB-HIV supervisor in every district. Patients put on the Delamanid (Dlm)-containing regimen The entire course of Dlm treatment will be earmarked at the DDS level. However, drugs will be supplied on a monthly basis to the TB Unit (TU) and further to the Health Facility (HF). Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Management of Patient-wise Boxes in Different Scenarios
ContentThere are different scenarios regarding patient-wise box (PWB) management. These are described below.
Scenario 1: Modification in regimen
In this scenario, the nodal and district drug-resistant tuberculosis centre (N/DDR-TBC) committee decides on a regimen modification.
- The district drug store (DDS) prepares modified PWBs from standard available boxes and loose drugs and arranges the supply of these PWBs to the treatment supporter (TS).
- The district TB officer (DTO) ensures that the drugs are supplied as per the modified regimen for all subsequent months.
Scenario 2: Extension of intensive phase
If intensive phase (IP) is extended:
- N/DDR-TBC informs the DTO
- DTO informs the health facility (HF) doctor and respective TB unit (TU)
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
When the patient is switched to continuation phase (CP) in case of a shorter regimen:
- DTO, again, informs HF doctor and respective TU
- HF releases 1-month PWB to the respective treatment support centre from where the patient is taking treatment
In the case of a longer regimen, after completion of 6-8 months of treatment, Linezolid (Lzd) should be reduced to 300 mg following N/DDR-TBC directives.
All patients must complete their monthly boxes before switching to the subsequent box provided.
Scenario 3: Change in DR-TB regimen
If N/DDR-TBC decides to change the DR-TB regimen of a patient, the DDS supplies a new treatment regimen box from PWB/ loose drugs. Unused drugs including Bdq containers should be sent back to the DDS. In this scenario, the patient should be immediately switched to the new regimen designed by N/DDR-TBC.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the State Drug Store [SDS] in the Constitution of Patient-wise Boxes
ContentThe State Drug Store (SDS) shall constitute drug boxes for:
- Shorter oral Bedaquiline (Bdq)-containing Multi-drug resistant (MDR)/ Rifampicin-resistant Tuberculosis (RR-TB) regimen (Type A and B)
- Longer oral Multi (M)/ Extensively Drug-resistant Tuberculosis (XDR-TB) regimen
- Isoniazid (H) mono/ poly DR-TB regimen
Boxes will be prepared manually only and not through Nikshay Aushadhi software.
From Nikshay Aushadhi drugs will be supplied in loose to districts.
Based on the requirement of districts SDS will supply boxes to respective districts by a hired transport agency or other mechanisms.
Loose drugs will also be supplied from SDS to the District Drug Store (DDS) for modification and preparation of new boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Role of the District Drug Store [DDS] in the Constitution of Patient-wise Boxes
ContentWhen there is a modification in the Drug-resistant TB (DR-TB) regimen as suggested by the Drug-Resistant Tuberculosis Centre (DR-TBC), the district drug storekeeper/ pharmacist prepares the modified boxes from loose Second-line anti-TB Drugs (SLD) supplied from the State Drug Store (SDS).
- The state shall provide the necessary support for capacity building of the District Drug Store (DDS) for carrying out the entire exercise of preparing standardized/ modified patient-wise drug boxes at the DDS level.
- Whenever oral regimens are modified during the course of treatment, the DDS needs to ensure that the change in the regimen should be incorporated into the supply of subsequent boxes.
- A full-time DDS storekeeper/ pharmacist must be mandatorily recruited/ placed for a successful decentralized system of preparation of drug boxes at the DDS level.
- District TB Officer (DTO) and Medical officer-DTC usually perform a random check to understand if there is any challenge in the constitution of patient-wise boxes.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Patient-wise Drug Boxes: Packing Instructions
ContentPackaging of loose drugs into monthly patient-wise boxes should be done under the guidance of those in charge of drug logistics at the state and district levels.
Patient-wise drug box preparation should be done for the following Drug-resistant TB (DR-TB) regimens:
- Shorter oral Bedaquiline-containing Multidrug-resistant/ Rifampicin-resistant TB (MDR/RR-TB) regimen
- Longer oral Multi (M)/ Extensively Drug-resistant TB (XDR-TB) regimen
- Isoniazid (H) mono/poly DR-TB regimen
Packaging Instructions
Patient-wise drug boxes are prepared as per the type of DR-TB regimen and weight bands. One monthly pouch of the capsule Cycloserine (Cs) and tablet Ethambutol (E) each, should be made from a plastic bag with a zip lock facility in which 1 gm pouch of silica gel desiccant should be kept.

Figure 1: Isoniazid (H) Mono/ Poly Regimen DR-TB Treatment Box
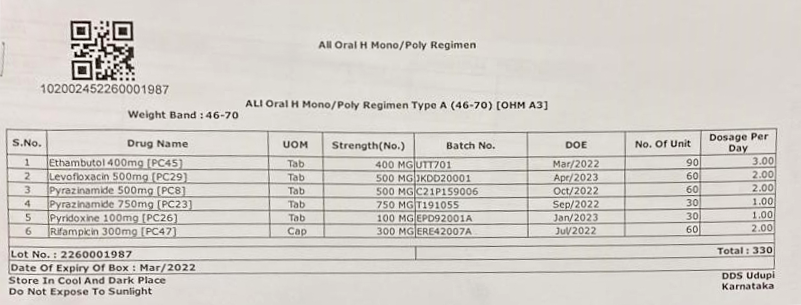
Figure 2: Isoniazid (H) Mono/ Poly Regimen DR-TB Label
In each Type A box, one pouch of silica gel desiccant of 4 gm weight should also be kept.
Labels for the boxes should be developed with the following information (Figures 2 and 3):
- Item-wise name of drugs with a quantity of each drug in the box
- Batch number and Date of Expiry (DOE) of the individual drugs
- DOE of boxes, with expiry date of the drug having the shortest expiry date
- Date of issue of the box from the State Drug Store (SDS)
- Serial number of the box
- Storage instructions on the box for ensuring adequate precautions in storage of the drugs, especially at the treatment supporter level. Some suggested messages are:
- “Store in a cool and dark place, preferably in a clean cupboard”
- “Do not expose to direct sunlight”
- “Keep away from children/unauthorized persons”
- “Box to be closed properly every time after the withdrawal of drugs”.
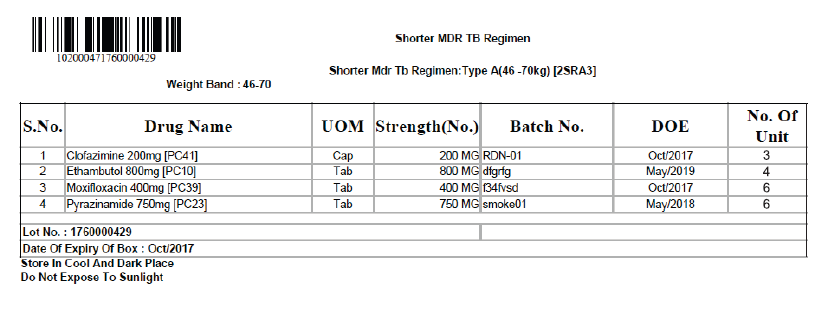
Figure 3: Prototype of a Label (Type A box for shorter DR-TB regimen for the weight band - 46 to 70 kg)
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, MOHFW, GOI.
Kindly provide your valuable feedback on the page to the link provided HERE
Recording box preparation in Nikshay Aushadhi
ContentRecording Box Preparation in Ni-kshay Aushadhi
As per the National TB Elimination Programme (NTEP) guidelines, CTD procures loose medicines for the treatment of DR-TB patients. These loose medicines are packed into Monthly Patient-Wise Boxes (PWB). PWB are prepared only at the State Drug Store (SDS) and District Drug Store (DDS) levels. Depending upon the availability of drugs, two types of boxes can be prepared in Ni-kshay Aushadhi.
Complete box – contains all the required drugs as per the regimen
Incomplete box – has less than the required number of drugs as per the regimen
ImageFigure 1: Overview of the process of recording box preparation in Ni-kshay Aushadhi
Detailed stepwise procedure:
Box Preparation
Step 1: Go to the Ni-kshay Aushadhi website and click on login. Enter your User ID, Password, and then the captcha text and click login.
Step 2: Reach the ‘Box Preparation Window’ following the path Home-menu > Services > Packing/Repacking > Box preparation and select the appropriate options as per the regimen from the drop-down menus of different sections given in the window and then press the ‘Go’ tab.
Image
Figure 2: Box completion window in Ni-kshay Aushadhi
Source: Ni-kshay Aushadhi portal
Step 3: Select the drug from the checkbox. The system will auto-fill the quantity required as per the box preparation quantity. Batch of near expiry date will be highlighted in pink colour. This is to be noted that the expiry date of the nearest expiry drug in the box will be the expiry date of the box itself. You can select any batch as per your requirement.
Image
Figure 3: Entering details of the box
Source: Ni-kshay Aushadhi portal
Step 4: After selecting the batch, click on ‘Add’ button to add the drug to the box. The system will highlight the added drug into the table with green colour. Repeat the process for all the drugs in the regimen. When all the drugs are added into the box as per the required quantity, the system will highlight them in green.

Figure 4: Details of drugs added to the box
Source: Ni-kshay Aushadhi portal
Step 5: Enter the ‘Remarks’ if required and click on the ‘Save’ button. The system will generate the voucher. You can save or print the voucher.

Figure 5: Voucher generated
Source: Ni-kshay Aushadhi portal
Note: If the available number of drugs is less than required, the selection could still be saved, and a voucher generated. Such boxes will be shown in blue in the view boxes section and are called as incomplete boxes.
Box Completion
This process involves adding the remaining drugs to the prepared incomplete boxes and can also be done at the receiving store. The process can be repeated multiple times depending upon the availability of the remaining drugs.
Steps to be followed in Ni-kshay Aushadhi:
Step 1: Reach the ‘Box Completion’ window following the path Menu > Services > Packing/Repacking > Box completion and enter your store name.
Step 2: On selecting the regimen from Tuberculosis (TB) subcategory, those batches with incomplete boxes will be displayed within the ‘Batch’ section. On selecting the batch from the Drug Details section will get displayed.
Step 3: Select a drug from the Drug Details table. Once selected, the batch details table of that drug will appear. Select the batch and add the drug quantity into the ‘To Be Added Qty' column.

Figure 6: Box completion window
Source: Ni-kshay Aushadhi portal
Step 4: Click on the 'Add' button. The selected row in the drug details gets coloured in green, which means that the given drug has been successfully added.
Step 5: Repeat steps 3 and 4 to fill more drugs and complete the box. Click on 'Save' to complete the process and generate the voucher.
Resource
Ni-kshay Aushadhi Manual-Central TB Division, Ministry of Health and Family Welfare, Government of India.
Assessment
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Where can box preparation be done? SDS only SDS and DDS only
TU
DDS only
2
This activity can be done at the SDS and DDS levels only.
Yes
Yes
Reconstitution of Bedaquiline [Bdq] Bottle
ContentThe reconstitution of Bedaquiline (Bdq) bottle should be done at the State Drug Store (SDS).
In case of lost to follow-up/ death/ transferred-out/ treatment stopped patients, Bdq bottle shall be brought back from the treatment centre to the Health Facility (HF) to the TB Unit (TU) to the District TB Centre (DTC) within the shortest possible time.
- A partially used Bdq bottle shall be sent back to the SDS for repackaging.
- The remaining tablets in the bottle received back shall be accounted for in the stock register and Nikshay-Aushadhi at the SDS.
- Upon reconstitution, the bottle shall be accounted for in the stock register (loose tablets to be mentioned in the remarks column) to be issued as per First-expire-first-out (FEFO) principles.
- When reconstitution is done, tablets of the same expiry can be considered using the same container to a maximum of 188 tablets.
- These reconstituted containers shall be used for the treatment of subsequent patients found eligible for Bdq.
- All such drugs that are taken from the new containers shall be collected as a group of 188 tablets of the same expiry and put in a light-resistant container as per the advice from the manufacturer.
- The actual expiry of tablets should be mentioned over the container.
- In the event of the SDS falling short of 188 tablets from an expiry batch, reconstitution can still be done using a number of tablets to complete 188 tablets with another expiry batch.
- In such a case, tablets of the respective expiry should be retained in their same respective containers and issued to patients and providers with counselling to consume the tablets with the nearest expiry first.
- If the expiry of the remaining tablets is less than six months, the same shall be consumed at the Nodal/ District Drug-resistant TB Centre (N/DDR-TBC) for admitted patients. It will be adjusted from the new long expiry bottle on discharge.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Standard Operating Procedure Manual - Procurement & Supply Chain Management, MOHFW, GOI.
Kindly provide your valuable feedback on the page to the link provided HERE
Storage of Drugs
ContentGood storage conditions and safe custody of drugs is important to ensure quality of drugs. Temperature and humidity control systems should be appropriately designed, installed, qualified and maintained, to ensure that the required storage conditions are maintained.
The State TB Officer (STO)/ District TB Officer (DTO) must ensure that the pharmacist/ store-keeper adheres to the following guidelines on proper storage of drugs.
Temperature
- The storage temperature should be 250C and maintained with an air conditioner wherever applicable.
Humidity
- Humidity levels should be below 60% and maintained with a dehumidifier, wherever required
Power Supply
- Regular power supply should be available for cooling devices (AC, ceiling fans etc) and dehumidifiers.
Drug Safety
Storage Room
- Should be cleaned and disinfected regularly, should be dry, well-lit and well-ventilated
- Should be free from any water penetration, rodent and pests
- Should have fire safety equipment in place
- The drugs should not be exposed to direct sunlight.
Stacking
- The shelves should be placed in such a way that there is sufficient space around for air circulation and free movement of personnel.
- Similar boxes should be stored adjacent to each other and stacked as per their expiry dates.
- The drugs expiring early should be placed closer to the ground and as those expiring late should be place at higher levels.
- Expired drugs should be segregated, sealed and stored in a separate part of the store so as to avoid issue to patients
Labelling
- The identification label, expiry date & manufacturing date of the anti-TB drugs should be marked with a bold marker pen on the visible side of the carton.
Resources
- WHO Technical Report Series, No. 908, 2003; Annex.9: Guide to Good Storage Practices for Pharmaceuticals.
- Procurement, Supply Chain Management & Preventive Maintenance, Module 6, CTD, MoHFW, India.
- Standard Operating Procedure Manual Procurement & Supply Chain Management, CTD, MoHFW, India, 2018.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
What is the maximum acceptable humidity levels in a drug store to maintain drug efficacy?
10%
25%
45%
60%
4
Humidity levels should be below 60% and maintained with a dehumidifier wherever required in order to ensure shelf life and efficacy of the drugs.
Yes
Yes
Fullscreen