Testing for TB Infection
ContentFor TB infection, there are two recommended tests which can be used to identify such patients.
Tuberculin Skin Test (TST)
The skin test is done by injecting a small amount (0.5 ml) of TB antigens into the top layer of skin on your inner forearm. If one has ever been exposed to TB bacteria (Mycobacterium tuberculosis), there will be a reaction indicated by the development of a firm red bump (induration) >= 10 mm at the site within 2 days.
Image
Figure: Tuberculin Skin Test
Interferon-gamma release assay (IGRA)
IGRA is a Blood test. If one has been exposed to TB bacteria, the white blood cell in the blood will release a substance called gamma interferon when the cells are exposed to specific TB antigens.
Image
Figure: Interferon-gamma release assay (IGRA)
Resources:
- Latent Tuberculosis Infection Guideline
- Guideline for Programmatic Management of Tuberculosis Preventive Treatment in India
Kindly provide your valuable feedback on the page to the link provided HERE
TB Preventive Therapy
ContentTPT treatment options recommended under NTEP include:
- 3-month weekly Isoniazid and Rifapentine (3HP)
- 6-months daily isoniazid (6H)

Table 1: TPT Options for Target Population; Source: (Guidelines for Programmatic Management of Tuberculosis Preventive Treatment)

Table 2: TPT dosage based on age and weight band recommended by NTEP; Source: Guidelines for Programmatic Management of Tuberculosis Preventive Treatment
Resources
- Guidelines for Programmatic Management of Tuberculosis Preventive Treatment
- National Strategic Plan for TB Elimination
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test TPT options recommended under NTEP include which of the following? 3-month weekly Isoniazid and Rifapentine (3HP) Rifampicin 6-months daily isoniazid (6H) 1 and 3 4 TPT options recommended under NTEP include 3-month weekly Isoniazid and Rifapentine (3HP) and 6-months daily isoniazid (6H). Yes Yes Eligibility for TPT
ContentThe eligibility for TB Preventive Treatment (TPT) relies on ruling out active TB among individuals and groups who are known to have a high risk of acquiring TB.
Prioritization of the target population for TPT is based on elevated risk of progression from infection to TB disease or increased likelihood of exposure to TB disease: At-risk populations include:
1. Expanded eligible group including children >5 years, adolescents and adult Household Contacts (HHC) of pulmonary* TB patients notified in Nikshay from public and private sector (*bacteriologically confirmed pulmonary TB patients will be prioritized for enumeration of the target population for TPT)
Table 1: Target Population (Expanded Eligible Groups); Source: Guidelines for Programmatic Management of Tuberculosis Preventive Treatment.

(*bacteriologically confirmed pulmonary TB patients will be prioritized for enumeration of the target population for TPT)
TPT reduces the overall risk for TB by 60-90% among People Living with HIV (PLHIV). Adults and children (>12 months) living with PLHIV should be screened for TB using a four-symptom complex and TPT can be provided to those without symptoms or after ruling out active TB in those with TB symptoms.
All HHC of pulmonary TB patients is at substantially higher risk for progression to active TB than the general population. Hence, all HHC of pulmonary TB patients, regardless of their age, should be given TPT after ruling out TB. In children HHC under 5 years of age, TPT will be offered after ruling out active TB, without testing for TB infection. In children, HHC >5 years and adults, chest X-rays and testing for TB infection would be offered wherever available.
- Expanded to other risk groups
Individuals in other risk groups include those on immunosuppressive therapy, having silicosis, on anti-TNF treatment, on dialysis, and preparing for organ or haematologic transplantation.
Systematic TB infection testing and treatment are not recommended for people with diabetes mellitus, malnutrition, smoking, or harmful alcohol abuse unless they have other risk factors for TB, such as HIV infection or a history of contact with TB patients within their household.
Table 2: Target Population (Other Risk Groups); Source: Guidelines for Programmatic Management of Tuberculosis Preventive Treatment.

Resource
Guidelines for Programmatic Management of Tuberculosis Preventive Treatment.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test TPT reduces the overall risk for TB by 60-90% among People Living with HIV (PLHIV). True False 1 TPT reduces the overall risk for TB by 60-90% among People Living with HIV (PLHIV). Cascade of Care for TPT
ContentIn the cascade of care approach, all target populations (People Living with HIV (PLHIV), Household Contacts (HHCs) and other such groups) who are at risk of developing TB disease are systematically reached out, screened for TB disease and after ruling out active TB disease, provided TB Preventive Treatment (TPT) as a part of the continuum of care.
The cascade of care approach among TPT target populations is shown in Figure 1.
Image
Figure 1: Cascade of TB Preventive Treatment; Source: Guidelines for Programmatic Management of Tuberculosis Preventive Treatment, p3.
Resources:
Guidelines for Programmatic Management of Tuberculosis Preventive Treatment in India.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of the following is the correct TPT cascade of care? Offer upfront CBNAAT to all at-risk populations, then offer TPT based on the results. Identify at-risk populations, then offer TPT to all the people that have been identified. Identify target populations at risk of developing TB, screen them, rule-out active TB disease, and provide TPT to eligible populations. None of the above 3 The TPT cascade of care is: Identify target populations at risk of developing TB disease, screen them for TB disease, rule-out active TB disease, and provide TPT to eligible populations. Approaches for TPT implementation
ContentThere are two programmatic approaches for Tuberculosis Preventive Therapy (TPT) implementation:
1. Test-and-treat approach – This approach aims to detect TB infection among key groups for implementing TPT.
- After ruling out active TB, the beneficiary is tested for TB infection.
- TPT is offered only to those with a positive test (Interferon Gamma Release Assay (IGRA)/ Tuberculin Skin Test (TST)/ Cutaneous TB (C-TB))
2. Treat-only approach – For certain groups, like People Living with HIV (PLHIV) and House Hold Contacts (HHC) < 5 years old, detecting TB infection is not required. Hence, this approach is given.
- After ruling out active TB, TPT is offered without testing for TB infection.
Test and treat approach*
- HHC of sputum positive Pulmonary TB >/= 5 years old
- Individuals on:
- Immunosuppressive therapy
- Having silicosis
- On anti-TumourTNF treatment
- On dialysis
- Preparing for solid organ or haematopoietic stem cell transplantation
Treat-only approach
- HHC of sputum positive Pulmonary TB (PTB) < 5 years old
- PLHIV#
*All efforts should be made to make IGRA available. However, TPT should not be withheld in case of non-availability of IGRA.
#PLHIV < 1 year old are offered TPT only if they are a household contact of an active TB case.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which of the following category of TPT beneficiaries is offered TPT without IGRA testing?
Household contacts of sputum positive PTB >/= 5 years old
PLHIV
Patient on dialysis
Silicosis patient
2
PLHIV and HHC of sputum positive PTB < 5 years old are offered TPT without testing for IGRA. This is called Treat-only approach.
Yes
Yes
Counselling for IGRA/TST
ContentInterferon Gamma Release Assay (IGRA) and Tuberculin Skin Tests (TST) are performed on individuals who are ruled out for active TB disease.
However, positive and negative tests in IGRA and TST do not necessarily mean the patient does or does not have Tuberculosis Infection (TBI) as the possibility of false positives and false negatives cannot be ruled out in these tests.
Importance of Counselling in IGRA/ TST
- All patients who undergo IGRA/ TST are already aware that they do not have an active TB disease and hence counselling is important to help them make informed decisions about undergoing IGRA/ TST for detecting TBI.
- Additionally, at the time of receiving positive IGRA/ TST results, they may be symptom-free or otherwise healthy. In such cases, resistance/denial to receive a prophylactic treatment like TB Preventive Therapy (TPT) is higher as its treatment course duration also is relatively longer.
- Counselling in IGRA/ TST is of utmost importance when the respective person belongs to the high-risk population and needs to be necessarily initiated on TPT and thus needs to be counselled for the same.
Components of Counselling in IGRA/ TST
- Information on TBI
- Need for undergoing IGRA/ TST
- Importance of initiating TPT post-IGRA/ TST tests
- If initiated on treatment, then schedule of medication
- Medication adherence support
- Follow-up
- Importance of completing the TPT course, adverse events
Resources
- Guidelines for Programmatic Management of Tuberculosis Preventive Treatment in India, CTD, MoHFW, India, 2021.
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, CTD, MoHFW, India, 2021.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Counselling for IGRA/ TST should necessarily include which of the following?
Counselling on DR-TB
Counselling on TB Infection (TBI)
Counselling on DBT
None of the above
2
Counselling for IGRA/ TST should necessarily include ‘Counselling on TB infection (TBI)’.
Yes
Yes
Counselling for TPT
ContentCounselling is of paramount importance for TB Preventive Treatment (TPT) initiation and completion as most of the target population screened and found eligible would know that they do not have TB disease, would be symptom-free or otherwise healthy and would not feel the need to take any treatment, especially Household Contacts (HHC).
Stakeholders Involved in Counselling for TPT (Figure below)

Figure: Stakeholders involved in counselling for TPT
Abbr: HWCs: Health and Wellness Centres; PHC: Primary Health Centre; ICTC: Integrated Counselling and Testing Centres; ART: Anti-retroviral Therapy; PLHIV: People Living with HIV
Components of Counselling for TPT
While counselling the person and family members, the treating doctors/ staff must follow the steps outlined in the table below for an effective counselling session.
Component
Actions to be taken
Confidentiality
Ensure confidentiality when seeking a person’s commitment to complete the course before initiating TPT.
Information
Provide information on:
- TB infection
- Need for TPT and protective benefits to the individual, household and wider community
- TPT is available free of charge under National Tuberculosis Elimination Programme (NTEP)
- TPT regimen prescribed, including duration, schedule of medication collection, and directions on how to take the medications
- Potential side-effects and adverse events involved and what to do in the event of various side-effects. People treated with rifamycins should be alerted in advance about the pink discolouration of secretions due to this medicine
- Importance of completing the full course of TPT
- Reasons and schedule of regular clinical and laboratory follow-up for treatment and monitoring
- Signs and symptoms of TB and advise on steps if they develop them
Medication adherence support
Agree on the best way to support treatment adherence, including the most suitable location for drug intake and the need for a treatment supporter, if required.
Family support
Involve family members and caregivers in health education when possible.
Openness
Invite clarification questions and provide clear and simple answers.
Information, Education and Communication materials
- Provide information materials in the local language and at the appropriate literacy level of the person concerned.
- Reinforce supportive educational messages at each contact during treatment.
Call support (in case of emergencies)
Provide a telephone number of the HCW staff/ TB Health Visitors and Senior Treatment Supervisors concerned to call for other queries or a need to contact health services for advice.
The National TB Elimination Programme (NTEP) national call centre (NIKSHAY SAMPARK – Toll-free number 1800116666) may be provided to index TB patients, those initiated on TPT and family members to serve as a resource for information, counselling and troubleshooting as required to enable TPT initiation, follow-up monitoring and completion.
Resources:
Guidelines for Programmatic Management of Tuberculosis Preventive Treatment in India.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which of the following people are involved when counselling for TPT?
Index TB patients
Caregivers
Family members
All of the above
4
When counselling persons eligible for TPT, it is best to involve the index TB patients, their families and caregivers.
Yes Yes Monitoring adherence to TPT
ContentTo achieve high treatment completion rates and the desired epidemiological impact of the TB Preventive Treatment (TPT), monitoring TPT treatment adherence, including management of missed doses and Adverse Drug Reactions (ADRs), is of paramount importance under the National TB Elimination Programme (NTEP).
Significance of Monitoring Adherence to TPT
Adherence to the TPT course and treatment completion are important determinants of clinical benefit, both at the individual and population levels as:
- Irregular or inadequate treatment reduces the protective efficacy of the TPT regimen.
- Poor adherence or early cessation of TPT can potentially increase the risk of the individual developing TB, including drug-resistant TB.
- Efficacy of TPT is greatest if at least 80% of the doses are taken within the duration of the regimen. The total number of doses taken is also a key determinant of the extent of TB prevention.

Figure: Strategies to Promote Adherence
Prevent TB India App and Integration with Nikshay as a Monitoring Tool
- Currently, under the NTEP, the person’s lifecycle approach and TB treatment episode level are recorded in Nikshay.
- TPT information management is integrated with this existing Nikshay approach. This includes information on screening, testing, eligibility assessment, TPT initiation, adherence monitoring and follow-up till treatment completion.
- The NTEP has adapted the World Health Organisation (WHO) Prevent TB India app and hosted it on Nikshay as an interim solution till the Nikshay TPT module is developed and fully functional.
- Health workers or treatment supporters will make entries directly into the app.
- The TPT monitoring dashboard can be accessed by various levels of supervisors using their respective Nikshay login ids using a link provided in the Nikshay Reports section on TPT Reports.
- A web-based comprehensive dashboard for Prevent TB initiative is also available at https://ltbi.nikshay.in/ltbi-generic-new/#/
Table: Roles of Stakeholders in Monitoring Adherence to TPT Role
Stakeholder
Treatment support and adherence monitoring including entry of daily doses taken in the Prevent TB India app/ Nikshay TPT module.
Community volunteers (TB survivors/ champions, Accredited Social Health Activists (ASHAs) and Anganwadi Workers)
- Regularly undertake home visits or tele/ video calls to monitor TPT adherence.
- Identify treatment interruptions at the earliest (Dashboards of Prevent TB India app/ Nikshay TPT module may be checked every week along with pill counting).
- HWCs/ sub-centre/ urban health posts (Community Health Officers (CHOs), Auxillary Nurse Midwives (ANMs), multipurpose workers and other field staff)
- Primary Health Centres (PHCs)/ Urban PHCs/ Private clinic (Medical Officers (MO), staff nurse)
- Adherence support and clinical monitoring through the concerned PHC/ sub-centre.
- Supportive supervision and handholding support to field level facilities and frontline workers, ASHAs and community volunteers on digital recording, using Prevent TB India app and monitoring TPT and follow-up examinations.
TB Unit (MO, Laboratory Technicians (LTs), staff nurse, pharmacist, counsellor (if available), Senior Treatment Supervisors (STS), Senior TB Laboroary Supervisors (STLS), TB Health Visitors (TBHV))
Ensuring adherence support for People Living with HIV (PLHIV) on TPT through mechanisms such as outreach workers, PLHIV networks, peer support groups, etc.
Anti Retroviral Therapy (ART) centre/ Link ART centre (MO, pharmacist, (institutional) staff nurse, counsellor, care coordinator)
Monitor and support adherence to TPT.
Tertiary care/ Medical colleges/ Corporate hospitals/ District hospitals/ Dialysis/ Cancer facilities (doctors, staff nurses)
Review data updating in Prevent TB India app/ Nikshay TPT module wherever available, check the quality of data regularly and provide feedback to TPT treatment supporters and for retrieval of TPT interrupters.
Supervisory staff at all health facilities including the State/ District TB cell (State TB Officers (STO), District TB Officers (DTO), State/ District Programme Coordinators)
Resources:
- Guidelines for Programmatic Management of Tuberculosis Preventive Treatment.
- Prevent TB Dashboard.
- Prevent TB India Mobile App.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which tools are used to monitor TPT adherence under the NTEP?
Video calls
Counting empty blisters
Directly asking the patient
Options 1 and 2
4
TPT adherence monitoring tools include direct observation of drug intake, 99DOTS/ MERM, counting empty blisters, tele/ video calls and refill monitoring.
Which of the following apps are currently used by NTEP to monitor TPT adherence?
TB Aarogya Sathi
Prevent TB India App
TPT app for NTEP
None of the above
2
NTEP has adapted the WHO Prevent TB India app and hosted it on Nikshay to monitor the entire TPT care cascade, including TPT adherence.
Preventive Treatment for Contacts of DR-TB Patients: WHO Recommendations
ContentThe World Health Organisation (WHO) recommends Tuberculosis Preventive Treatment (TPT) among contacts exposed to Multidrug-resistant TB (MDR-TB) with Fluoroquinolone (FQ)-sensitive, or Isoniazid (H)-resistant with Rifampicin (R) sensitive DR-TB patients.
Points to be Considered Before TPT Initiation
- The preventive treatment should be individualized after a careful assessment of the intensity of exposure of contact with the index case.
- Confirm the source patient and her/his drug resistance pattern bacteriologically.
- Ascertain Latent TB Infection (LTBI) using Interferon Gamma Release Assay (IGRA) or Tuberculin Skin Tests (TST).
Table: WHO's TPT Regimen Recommendation
TPT REGIMEN
ELIGIBLE POPULATION
6Lfx (six-month Levofloxacin)
Among contacts exposed to patients with known MDR and FQ-sensitive TB
6H (six-month Isoniazid)
Among contacts of H-susceptible TB in confirmed R-resistant TB index patient
4R (four-month Rifampicin)
Among contacts with known H-resistant and R-sensitive TB
Regardless of whether treatment is given or not, clinical follow-up should be done for two years.
Any emergent signs and symptoms suggestive of TB should be actively investigated and curative regimens started, as needed.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Consolidated Guidelines on Tuberculosis: Module 1: Prevention: Tuberculosis Preventive Treatment, 2020.
- Latent TB Infection: Updated and Consolidated Guidelines for Programmatic Management, WHO, 2018.
Kindly provide your valuable feedback on the page to the link provided HERE
Screening among Household Contacts of DR-TB Patients
ContentThe National TB Elimination Programme (NTEP) follows an integrated algorithm for screening and ruling out active Tuberculosis (TB) among Household Contacts (HHCs) of Drug-resistant Tuberculosis (DR-TB) patients.
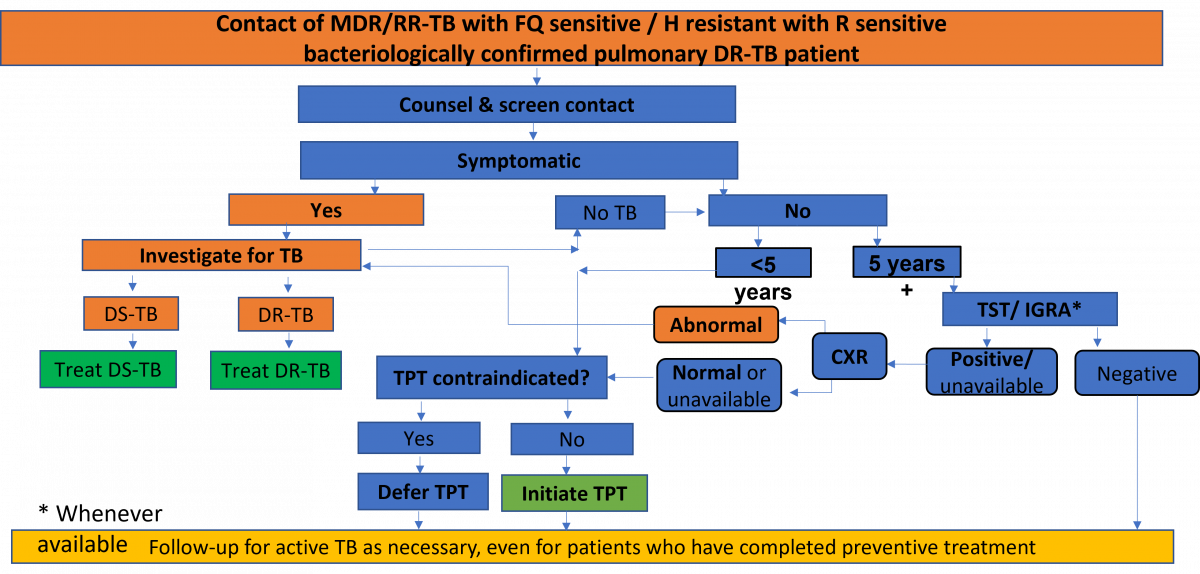
Figure: Integrated Algorithm for Screening and Ruling out Active TB among HHCs of DR-TB Patients; Source: Guidelines for PMDT in India, 2021, p118.
Abbr: MDR/RR-TB: Multidrug-resistant/ Rifampicin-resistant TB; FQ: Fluoroquinolone; h: Isoniazid; R: Rifampicin; DR-TB: Drug-resistant TB; DS-TB: Drug-susceptible TB; TPT: TB Preventive Treatment; CXR: Chest X-ray; TST: Tuberculin Skin Tests; IGRA: Interferon Gamma Release Assay.
Salient Features of the Screening Algorithm
- Once a DR-TB patient is identified, all HHCs are counselled, screened and evaluated to rule out active TB.
- Nucleic Acid Amplification Test (NAAT) will be used up front among contacts with symptoms or abnormal Chest X-ray (CXR) to diagnose TB.
- If the result is Mycobacterium tuberculosis (MTB) detected with no resistance, the treatment for Drug-sensitive TB (DS-TB) is initiated.
- If the result is MTB detected with Isoniazid (H) and/or Rifampicin (R) resistance, manage as per DR-TB guidelines.
- If the result is MTB not detected, in HHC <5 years of age, assess for TB Preventive Treatment (TPT) and check for any contraindications.
- If the result is MTB not detected, in HHC >5 years of age with Latent TB Infection (LTBI) test positive or unavailable and CXR is normal or unavailable, check for any contraindications.
- If contraindications to TPT drugs exist, defer TPT and if no contraindication exists, offer TPT regimen as appropriate based on the Drug Susceptibility Testing (DST) pattern of the index patient
Follow-up for active TB as necessary, even for patients who have completed preventive treatment irrespective of TPT offer.
Footnotes
- HIV-positive contact: If <10 years of age, any one of current cough, fever, history of contact with TB, reported weight loss, confirmed weight loss >5% since last visit or growth curve flattening or weight for age <-2 Z-scores. Asymptomatic infants, <1 year, with HIV are only treated for LTBI if they are household contacts of a TB patient. TPT or Interferon Gamma Release Assay (IGRA) may identify People Living with HIV (PLHIV) who will benefit most from preventive treatment. CXR may be used in PLHIV on Anti-retroviral Treatment (ART), before starting TPT.
- Symptomatic HHC: Any one of cough or fever or night sweats or haemoptysis or weight loss or chest pain or shortness of breath or fatigue. Children <5 years should also be free of anorexia, failure to thrive, not eating well, decreased activity or playfulness to be considered asymptomatic.
- Other high-risk groups: Includes silicosis, dialysis, anti-TNF agent treatment, preparation for transplantation or other vulnerable risk groups where testing must precede TPT.
- Contraindication to TPT: Include acute or chronic hepatitis; peripheral neuropathy (if Isoniazid is used); regular and heavy alcohol consumption. Pregnancy or a previous history of TB are not contraindications.
- Selection of TPT regimen: Regimen is chosen based on considerations of age, strain (drug-susceptible or otherwise), risk of toxicity, availability and preferences.
- CXR may have been carried out early on as part of intensified case finding.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- WHO Consolidated Guidelines on Tuberculosis: Module 1: Prevention: Tuberculosis Preventive Treatment, 2020.
- Latent TB Infection: Updated and Consolidated Guidelines for Programmatic Management, WHO, 2018.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen