Active TB Drug Safety Monitoring [aDSM]
ContentActive Drug Safety Monitoring (aDSM) is a proactive effort made to elicit adverse events from patients by directly asking the patient, screening records, and checking laboratory and clinical tests. All adverse events are captured systematically at all patient visits (during treatment initiation, treatment review and extra visits).
Drug-resistant Tuberculosis (DR-TB) management requires a robust system of monitoring and reporting Adverse Drug Reaction (ADR)-related information to build guidelines for the safe use of drugs, newer drugs, a newer combination of drugs in the regimen, high dose formulations etc. Hence, aDSM is essential.
The National Tuberculosis Elimination Programme (NTEP) in collaboration with PvPI (Pharmacovigilance Program of India) and with support from the World Health Organisation (WHO) India, developed the comprehensive aDSM system for monitoring the ADRs for all types of DR-TB patients.
- aDSM follows the patient pathway from registration to the treatment outcome.
- For every patient, an aDSM treatment initiation form needs to be filled out. For all patients with Serious Adverse Events (SAE), the aDSM treatment review form needs to be filled out.
- The aDSM mechanism was initially used only at the Nodal DR-TB Centre (NDR-TBC) involved in Bedaquiline (Bdq - new drug) - containing regimens, but now this mechanism has been expanded to all DR-TB centres.
- A drug safety monitoring committee periodically monitors the occurrence of Adverse Events (AEs) or SAEs, including deaths of patients while on new drugs containing regimens for necessary signalling and guidance to the program on their safety and efficacy.
- aDSM reports should be uploaded in a timely manner on Nikshay.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
Pharmacovigilance in NTEP
ContentPharmacovigilance is defined by the World Health Organisation (WHO) as the science and activities relating to the detection, assessment, understanding and prevention of adverse effects or any other drug-related problem.
- It is a fundamental public health surveillance activity to ensure patient safety measures in healthcare.
- Good pharmacovigilance will identify the risks within the shortest possible time after medicines have been marketed and help establish or identify risk factors.
Importance of Pharmacovigilance
Pharmacovigilance allows for intelligent, evidence-based prescribing with the potential for preventing many Adverse Drug Reactions (ADRs). Pharmacovigilance will help in:
- Improving patient care by assessing both the harms and benefits received from drugs (anti-tubercular treatment).
- Strengthening patient safety, safeguarding the patient’s interests and ensuring adherence to prescribed drug regimens.
- Preventing antimicrobial resistance.
Pharmacovigilance ultimately helps each patient in receiving optimum therapy at a lower cost to the health system.
Conducting Pharmacovigilance under the National TB Elimination Programme (NTEP)
The Pharmacovigilance Programme of India (PvPI) was set up by the Ministry of Health and Family Welfare, Govt. of India, in July 2010. PvPI is India’s national programme for surveillance of ADR-related information.
NTEP in collaboration with PvPI, and with support from WHO India, developed the comprehensive active Drug Safety Monitoring and Management (aDSM) system for ADR monitoring. Pharmacovigilance is prioritised in Drug-resistant TB (DR-TB) centres for drug-resistant cases.
Adverse events reporting for pharmacovigilance is done as follows:
- DR-TB centres are linked with ADR Monitoring Centres (AMC) established in medical colleges to initiate reporting of ADR in a systematic manner.
- Serious adverse events are reported to AMCs and Central TB Division (CTD) within 24 hours. This is done via a standardized suspected ADR reporting form (Annexure-11) which is filled by the treating doctor.
- The data is entered in Nikshay on a regular basis by statistical assistants at the nodal DR-TB centre and senior DR-TB TB-HIV supervisors at the district DR-TB centre.
- From Nikshay, the information is directly communicated to PvPI through a connecting bridge called Vigiflow.
- The ADR data submitted to Vigifloware is analysed by PvPI and shared with CTD on a regular basis.
Resources
- Training Modules (1-4) for Programme Managers and Medical Officers, 2020.
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Practical Handbook on the Pharmacovigilance of Medicines used in the Treatment of Tuberculosis, WHO, 2012.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Which of the following is true concerning pharmacovigilance in NTEP?
PvPI is India’s national programme for surveillance of ADR-related information.
ADR-related information flows between Nikshay and PvPI via Vigiflow.
Pharmacovigilance assesses both the harms and benefits received from anti-TB drugs.
All of the above
4
PvPI is India’s national programme for surveillance of ADR-related information, which flows between Nikshay and PvPI via Vigiflow. Pharmacovigilance assesses both the harms and benefits received from anti-TB drugs.
Yes Yes Adverse Drug Reactions(ADRs) to Second Line Treatment
ContentCommon Adverse events to second line treatment are as below
Figure: Adverse Drug Reaction to Second line drugs
Adverse events should be identified, monitored and be referred to
- Nearest treating doctor for minor symptoms or
- District DR-TB Centres for major symptoms
If required, hospitalization can be done at the District DR-TB Centers where inpatient facility is available or referred to a Nodal DRTB Centre for admission
Management of DR-TB ADR: Rash, Allergy and Anaphylaxis Reaction
ContentHypersensitivity reactions like rashes, allergies and anaphylactic reactions are common Adverse Drug Reactions (ADRs) to any of the second-line anti-TB drugs.
These ADRs are often reported by patients themselves.
Milder forms of this ADR present with a localised rash that is not associated with mucus membranes. In such cases, patients can be reassured and managed symptomatically at home or at the Peripheral Health Institution (PHI).
Serious cases might present with generalized patchy lesions and should be referred to the district/ nodal DR-TB centre.
Drugs that have been identified as the cause of these reactions must be noted in the Drug-resistant TB (DR-TB) treatment card.
Suspected Agent(s): Any second-line anti-TB drug
Management Strategies
- Eliminate other potential causes of allergic skin reactions (e.g., scabies or other environmental agents).
- Mild reaction: Reassure the patient and manage symptomatically with the doctor.
- Minor dermatologic reactions: Continue anti-TB medications and:
- Include antihistamines, hydrocortisone cream for localized rash
- Prednisone in a low dose of 10 to 20 mg per day for several weeks, if other measures are not helpful
- Use a moisturizing lotion for dry skin related itching (especially in diabetics)
- Serious allergic reactions: Stop all therapies pending resolution of reaction and refer the patient to a nodal DR-TB centre/ tertiary centre for further management.
- Anaphylaxis: Follow standard emergency protocols.
Points to Note
- History of previous drug allergies to be reviewed.
- Any known drug allergies are to be noted on the DR-TB treatment card.
- Flushing reaction to Pyrazinamide (Z) is usually mild and resolves with time.
- Hot flushes, itching, palpitations can be caused by Isoniazid (H) and tyramine-containing foods (cheese, red wine). If this occurs, advise patients to avoid foods that precipitate reactions.
- Any of the drugs can cause hives (urticaria). To identify the drug, introduce drugs one at a time and in case of hives, a desensitization attempt can be made.
Once the rash resolves, reintroduce the remaining drugs, one at a time with the one most likely to cause the reaction last. The order of reintroduction will be Isoniazid (H), Pyrazinamide (Z), Ethionamide (Eto), Ethambutol (E), Fluoroquinolone (FQ).
Consider not reintroducing, even as a challenge, any drug that is highly likely to be the cause of anaphylaxis or Stevens-Johnson syndrome and suspend any drug identified to be the cause of a serious reaction permanently.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Diarrhoea and/or Flatulence
ContentAdverse Drug Reactions (ADRs), such as diarrhoea and/or flatulence, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s): Para Aminosalicylic Acid (PAS), Ethionamide (Eto)
Suggested Management Strategies
- Motivate patients to tolerate some degree of loose stools and flatulence.
- Encourage fluid intake.
- Treat uncomplicated diarrhoea (no blood in stool and no fever) with Cap Racecadotril 1 stat followed after 8 hours.
- Check serum electrolytes (especially potassium) and dehydration status if diarrhoea is severe.
- Fever and diarrhoea and/or blood in the stools indicate that diarrhoea may be secondary to something other than a simple adverse effect of anti-TB drugs.
Points to Note
- Consider other causes of diarrhoea, such as:
- Pseudo-membranous colitis: It is related to broad-spectrum antibiotics (such as FQ), is a serious and even life-threatening condition, and shows warning signs such as fever, bloody diarrhoea, intense abdominal pain and increased white blood cells.
- Parasites and common waterborne pathogens in the area of the patient: Evaluate and treat these.
- Lactose intolerance: Especially if the patient has been exposed to new foods in the hospital which is not normally part of his/ her diet.
- Consider using Loperamide in children over two years of age.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer- Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Hepatitis
ContentHepatitis is a common adverse drug reaction caused by some Drug-resistance TB (DR-TB) drugs. It is monitored by measuring the Alanine transaminase (ALT) and Aspartate aminotransferase (AST) levels.
If there is jaundice (yellowing of the skin or eyes), field-level health care workers must immediately refer the patient to the nearest higher health centres - District/ Nodal DR-TB Centre/ Sub-district or district hospital, medical college hospitals, etc.
Suspected agent(s): Pyrazinamide (Z), Isoniazid (H), Rifampicin (R), Ethionamide (Eto), P-aminosalicylic Acid (PAS), Bedaquiline (Bdq)
Suggested Management Strategies
- In cases where the patient is very sick, i.e., having meningitis and sputum smear is grade 3+, administer Anti-TB Treatment (ATT), e.g., Streptomycin, Fluoroquinolone (FQ) and Cycloserine (Cs).
- Where the patient is not seriously ill and one can wait, the introduction of ATT can be done once enzyme levels are near normal.
- If enzymes are more than five times the upper limit of normal, stop all hepatotoxic drugs and continue with at least three non-hepatotoxic medications (for example, the injectable agent, FQ and Cs). If hepatitis worsens or does not resolve with the three-drug regimen, then stop all drugs.
- If hepatitis worsens or does not resolve with the three-drug regimen, then stop all drugs.
- Eliminate other potential causes of hepatitis (viral hepatitis and alcohol-induced hepatitis being the two most common causes) and treat any that are identified.
- Once enzyme level improves, reintroduce remaining drugs, one at a time with the least hepatotoxic agents first, while monitoring liver function by testing enzymes every three days.
- If the most likely agent causing hepatitis is not essential, consider not reintroducing it.
Points to Note
- Any history of previous drug-induced hepatitis should be carefully analysed to determine the most likely causative agent(s); these drugs should be avoided in future regimens.
- Viral serology should be done to rule out other aetiologies of hepatitis if available, especially, for hepatitis A, B and C.
- Alcohol use should be investigated and addressed if found.
- Generally, hepatitis due to medications resolves upon discontinuation of the suspected drug.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Giddiness
ContentAdverse Drug Reaction (ADR), such as giddiness, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s) - Amikacin (Am), Ethionamide (Eto), Fluoroquinolone (FQ) and/or Pyrazinamide (Z)
Suggested Management Strategies
- Whenever a patient complains of giddiness, over-sleepiness or poor concentration, they should be provided counselling.
- If severe, the offending drug should be identified by administering drugs individually and observing the response.
- The dose of the offending drug identified may be adjusted or the offending drug terminated if required.
- Aminoglycosides, especially in the elderly age group must be kept in mind for giddiness as it may be an early sign of 8th nerve toxicity.
Point to Note: In cases of severe giddiness, the patient may be referred to the neurologist for further management as per the standard protocols.
Resources
Management of DR-TB ADR: QT Prolongation
ContentQT prolongation is a condition in which repolarization of the heart after a heartbeat is affected.
It results in an increased risk of an irregular heartbeat which can result in shortness of breath or chest pain, fainting, seizures or cardiac arrest.
If patients experience such signs or symptoms, health workers need to refer such patients to the nearest health facility where Electrocardiogram (ECG) can be done and further management initiated.
Suspected agent(s): Bedaquiline (Bdq), Fluoroquinolone (FQ), Clofazimine (Cfz)
Suggested Management Strategies
- Values above Corrected QT Interval by Fridericia (QTcF) 450 ms in males and 470 ms in females are referred to as prolonged. Patients with prolonged QTcF are at a risk for developing cardiac arrhythmias like Torsades de Pointes, which can be life-threatening.
- Fluoroquinolone (FQ) may cause prolongation of the QTcF; Moxifloxacin (Mfx) and Gatifloxacin (Gfx) cause the greatest QTcF prolongation, while Levofloxacin (Lfx) and Ofloxacin (Ofx) have a lower risk.
- Currently, ECG monitoring prior to initiation and during Drug-resistant TB (DR-TB) treatment is only required with the use of Bdq or when two drugs known to prolong QTcF (e.g., Mfx, Cfz) are combined in the same regimen.
- Low serum levels of potassium, calcium and magnesium are associated with QTc prolongation. Electrolyte levels should be maintained in the normal range in any patient with an elevated QT interval.
- Avoid other drugs that increase the QT interval.
- QT prolongation can result in ventricular arrhythmias (Torsades de Pointes) and sudden death. It is therefore imperative that ECGs be used to monitor the QT interval regularly during the use of the suspected drugs.
- QTcF value between 450-480 ms: Rule out other causes of prolonged QTc, before deciding to withhold the suspected agents.
- Management of increased QTcF entails looking at the algorithm for the reintroduction of anti-TB drugs (Bdq/ Delamanid (Dlm)/ FQ/ Cfz) once prolonged QTc has normalised.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- WHO Consolidated Guidelines on Tuberculosis: Module 4 – Treatment: Drug-resistant TB Treatment, 2020.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Hematological Abnormalities
ContentAdverse Drug Reactions (ADRs), such as haematological abnormalities, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s): Linezolid (Lzd)
Suggested Management Strategies
- Stop Linezolid (Lzd) if myelosuppression (suppression of white blood cells, red blood cells or platelets) occurs.
- Consider restarting with a lower dose of Lzd (300 mg, instead of 600 mg) if myelosuppression resolves and if Lzd is considered essential to the regimen.
- Consider non-drug-related causes of haematological abnormalities.
- Consider blood transfusion for severe anaemia.
Points to Note
- Haematological abnormalities (leukopenia, thrombocytopenia, anaemia, red cell aplasia, coagulation abnormalities and eosinophilia) can rarely occur with several other anti-TB drugs.
- There is little experience with prolonged use of Lzd.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
- Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Hypothyroidism
ContentAdverse Drug Reactions (ADRs), such as hypothyroidism, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
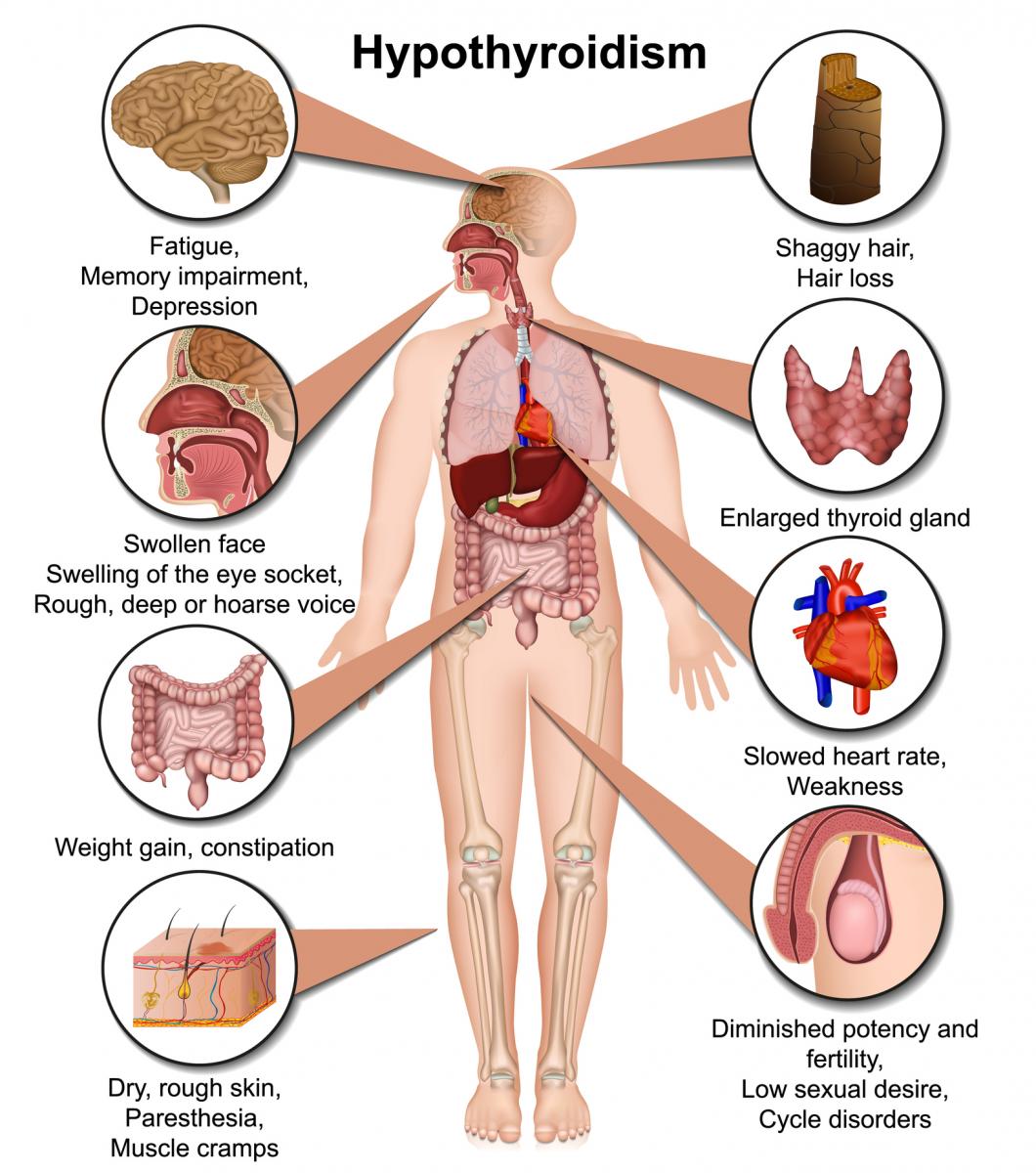
Figure: Symptoms of Hypothyroidism
Suspected agent(s): Ethionamide (Eto), Para Aminosalicylic Acid (PAS)
Suggested Management Strategies:
- In cases of hypothyroidism, the opinion of a general physician/ endocrinologist may be taken.
- Eto /PAS can be continued with the introduction of thyroxine.
Points to Note
- Symptoms of hypothyroidism include fatigue, somnolence, cold intolerance, dry skin, coarse hair and constipation, as well as occasional depression and inability to concentrate (Figure above).
- In cases with abnormal weight gain, hypothyroidism may be ruled out.
- It is completely reversible upon discontinuation of PAS and/or Eto.
- Combination of Eto with PAS is more frequently associated with hypothyroidism than when each individual drug is used.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Arthralgia
ContentAdverse Drug Reactions (ADRs), such as arthralgia, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s): Pyrazinamide (Z), Fluoroquinolone (FQ), Bedaquiline (Bdq)
Suggested Management Strategies
- Initiate with paracetamol in the beginning.
- Treatment with non-steroidal anti-inflammatory drugs (Indomethacin 50 mg twice daily or Ibuprofen 400 to 800 mg three times a day).
- Lower the dose of the suspected agent (most commonly Z) if this can be done without compromising the regimen.
- Discontinue the suspected agent if this can be done without compromising the regimen.
Points to Note
- Symptoms of arthralgia generally diminish over time, even without intervention.
- Uric acid levels may be elevated in patients on Z.
- There is little evidence to support the addition of Allopurinol for arthralgia. However, if gout is present, it should be used.
- If acute swelling, redness, and warmth are present in a joint, consider aspiration for diagnosis of gout, infections, autoimmune diseases, etc.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Peripheral Neuropathy
ContentSerious Adverse Drug Reaction (ADR), such as peripheral neuropathy, may occur during Drug-resistant TB (DR-TB) treatment.
Suspected agent(s): Linezolid (Lzd), Cycloserine (Cs), Isoniazid (H), Amikacin (Am), Fluoroquinolone (FQ), rarely Ethionamide (Eto), Ethambutol (E)
Suggested Management Strategies
- To prevent the occurrence of such adverse reactions, all patients on Multidrug-resistant TB (MDR-TB) medicines should receive pyridoxine daily.
- The commonest offending agent is Linezolid (Lzd), almost 60–70% of patients on Lzd 600 mg/day may develop neuropathy and pyridoxine does not help in preventing Lzd-induced neuropathy. Early recognition of neuropathy symptoms and early dose reduction of Lzd helps to prevent progression.
- If there is no improvement or symptoms worsen, Amitriptyline 25 mg will be added (to be avoided with Bdq).
- Correct any vitamin or nutritional deficiencies and increase pyridoxine to the maximum daily dose (100 mg per day).
- Consider whether the dose of Cs can be reduced without compromising the regimen. If H is being used (especially high dose Isoniazid (Hh)), consider stopping it.
Medical Treatment of Peripheral Neuropathy
- Non-steroidal anti-inflammatory drugs or acetaminophen may help to alleviate symptoms.
- Treatment with tricyclic antidepressants, such as amitriptyline (start with 25 mg at bedtime, the dose may be increased to a maximum of 150 mg), can be tried.
- Do not use tricyclic antidepressants with selective serotonin reuptake inhibitors and Bedaquilin (Bdq).
- Medication can be discontinued (rarely), only if an alternative drug is available and the regimen is not compromised.
Points to Note
- Patients with comorbid diseases (diabetes, HIV, alcohol dependence) are likely to develop peripheral neuropathy.
- Neuropathy associated with Lzd is common after prolonged use and may be irreversible. Thus, suspension of this drug should be strongly considered when neuropathy persists despite the above measures.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Psychotic Symptoms
ContentAdverse Drug Reactions (ADRs), such as psychotic symptoms, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s): Cycloserine (Cs), Isoniazid (H), Fluoroquinolone (FQ)
Suggested Management Strategies
- Stop the suspected agent for a short period (1-4 weeks) while the psychotic symptoms are brought under control.
- The most likely drug is Cs followed by high dose isoniazid (Hh). Lower the dose/ discontinue the suspected agent (if it can be done without compromising the regimen).
- If moderate to severe symptoms persist, initiate antipsychotic treatment (haloperidol).
- Hospitalize in a ward with psychiatric expertise if the patient is at risk to hurt himself/ herself or others.
- Once all the symptoms resolve and the patient is off Cs, antipsychotic treatment can be tapered off.
- If Cs is continued at a lower dose, antipsychotic treatment may need to be continued and any attempts of tapering off should be done after referring to a psychiatrist trained in the adverse effects of second-line anti-TB drugs.
Point to Note
- Some patients will be required to continue antipsychotic treatment.
- A previous history of psychiatric disease is not an absolute contraindication to Cs, but its use may increase the likelihood of psychotic symptoms that are found to be developing during treatment.
- Some patients will tolerate Cs with an antipsychotic drug, but this should be done in consultation with a psychiatrist as these patients will be required to be under special observation; this should be done only when there is no other alternative.
- Psychotic symptoms are generally reversible upon completion of DR-TB treatment or cessation of the offending agent.
- Always check creatinine in patients with new-onset psychosis. A decrease in renal function can result in high blood levels of Cs, which can cause psychosis.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Nephrotoxicity [Renal Toxicity]
ContentAdverse Drug Reactions (ADRs), such as renal toxicity, may occur from the Drug-resistant TB (DR-TB) treatment regimen.
Suspected agent(s): Amikacin (Am)
- Prior to starting the treatment, all patients will have their renal function evaluated.
- During the treatment of DR-TB, if the patient presents with symptoms and/or signs of renal impairment (oliguria, anuria, puffiness of face, pedal oedema), all the drugs should be withheld; renal function tests should be done and, if required, the opinion of a nephrologist should be sought.
- The re-introduction of drugs will be undertaken by the DR-TB centre committee in consultation with a nephrologist, along with frequent monitoring of renal parameters.
Suggested Management Strategies
- Discontinue the suspected agent.
- Consider using Capreomycin (Cm) if Aminoglycoside was the prior injectable drug in the regimen.
- Consider other contributing etiologies (non-steroidal anti-inflammatory drugs, diabetes, other medications, dehydration, congestive heart failure, urinary obstruction, etc.) and address as indicated.
- Follow creatinine (and electrolyte) levels closely, every 1–2 weeks.
- Consider dosing the injectable agent 2-3 times a week if the drug is essential to the regimen and the patient can tolerate it (close monitoring of creatinine).
- If creatinine continues to rise despite twice/ thrice a week dosing, suspend the injectable agent.
- Adjust all TB medication according to creatinine clearance in consultation with a nephrologist.
- Also, note that renal impairment may be permanent.
Points to Note
- During treatment, blood urea and serum creatinine should be done every month for the first three months after treatment initiation and then every three months thereafter whilst injection Am is being administered. Salient renal toxicity may be picked up by these routine follow-up biochemical examinations.
- If at any time, the blood urea or serum creatinine becomes abnormal, treatment should be withheld and further management is decided upon in consultation with the DR-TB centre committee.
- History of diabetes or renal disease is not a contraindication to the use of agents listed here, although patients with these comorbidities may be at an increased risk for developing acute kidney injury.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officer - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
- Technical and Operational Guidelines for TB in India, 2016.
Kindly provide your valuable feedback on the page to the link provided HERE
Management of DR-TB ADR: Vestibular Toxicity
ContentVestibular toxicity is one of the Adverse Drug Reactions (ADRs) that results in damage of the balance structure in the inner ear leading to loss of balance for the patient.
Suspected agent(s): Amikacin (Am), Cycloserine (Cs), Fluoroquinolones (FQs), Isoniazid (H), Ethionamide (Eto), Linezolid (Lzd)
Suggested Management Strategies
- If early symptoms of vestibular toxicity appear, there may be a need to change the dosing of the injectable agent to twice/ thrice a week.
- Consider using Capreomycin (Cm) if an aminoglycoside was the prior injectable in the regimen.
- If tinnitus and unsteadiness worsen with the above adjustment, stop the injectable agent.
- This is one of the few adverse events that may cause permanent intolerable toxicity and can necessitate the discontinuation of a class of agents.
Points to Note
- Ask the patient about tinnitus and unsteadiness every week, especially in elderly patients.
- Fullness in the ears and intermittent ringing are early symptoms of vestibular toxicity.
- A degree of disequilibrium can be caused by Cs, FQs, Eto, H or Lzd.
- Some clinicians will stop all drugs for several days to see if symptoms are attributed to these drugs.
- Symptoms of vestibular toxicity generally do not improve on withholding medications.
Resources
- Guidelines for Programmatic Management of Drug-resistant Tuberculosis in India, March 2021.
- Ready Reckoner for Medical Officers - Adverse Drug Reactions Associated with Anti-TB Drugs Identification and Management, 2019.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen