-
STLS: General concepts in quality assurance
FullscreenStoring slides at a DMC for QA
ContentIt is important to store slides, after reading as these will be required for Quality assurance - internal rechecking by the supervisor, and for External Quality Assurance (EQA) as per National TB Elimination Programme (NTEP) guidelines.
- Prior to storage slides need to be dried by gently placing the slide gently face down on the tissue paper or by wrapping it in the tissue paper and leaving it overnight to remove excess oil (Figure 1).

Figure 1: Using Tissue Paper to Drain Oil
- The slides are to be stored in the slide box in the same order as they are listed in the laboratory register (Figure 2).

Figure 2: Keeping the Slides in Same Order as Listed in the Laboratory Register
- One blank place must be left behind the first slide from a patient suspected with TB this allows the second slide of the same patient to be added after reading. This process will keep results consistent with the laboratory register (Figure 3).

Figure 3: Keeping Blank Spaces Between Slides for Consistency
- Always store slides in closed slide boxes away from sunlight (Figure 4).

Figure 4: Storing Slides in Closed Slide Boxes

Resources
- Manual for Sputum Smear Fluorescence Microscopy, RNTCP, MoHFW, 2007.
- Laboratory Diagnosis of Tuberculosis by Sputum Microscopy, GLI Initiative.
Kindly provide your valuable feedback on the page to the link provided HERE
False Positivity and False Negativity in Microscopy
ContentFalse result: It’s a situation where the test result is different from the truth.
Types of False Results
- False Positive: When the test result is positive but the patient does not have TB.
- False Negative: When the test result is negative but the patient has TB.
Both cases will lead to wrong categorization and incorrect treatment. The consequences of false results are shown in the figure below:
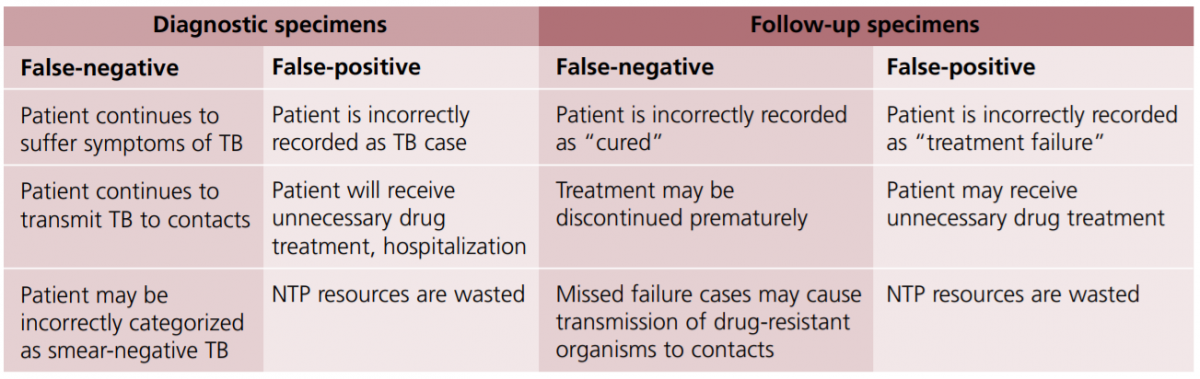
Figure: Consequences of False Results in Sputum Smear Microscopy; Source: Quality Assurance of Sputum Microscopy in DOTS Programmes.
Resources
- RNTCP Module for Laboratory Technicians, CTD, MoHFW, 2005.
- Quality Assurance of Sputum Microscopy in DOTS Programmes Regional Guidelines for Countries in the Western Pacific, WHO, 2003.
Kindly provide your valuable feedback on the page to the link provided HERE
Need for Quality Testing and Control
ContentQuality Assurance (QA) is a systematic process used to determine the quality standards of TB laboratories.
Poor quality diagnosis may results in failure to diagnose TB or unnecessary treatment of non-TB case. Thus an effective QA mechanism is important for reliability of laboratory diagnosis.
QA in National Tuberculosis Elimination Programme (NTEP) consists of:
- Internal Quality Control (QC)
- External Quality Assessment (EQA)
- Continuous Quality Improvement (QI)
QC or Internal Quality Assurance is a systematic internal monitoring of working practices, technical procedures, checking equipment, new lot of reagents, smear preparation, grading etc
EQA is a process to assess laboratory performance by comparing laboratory results with a laboratory in higher tier i.e.Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL) through on-site evaluation, panel testing and random blinded rechecking of slides. Figure 1 depicts an eg of on-site evaluation with roles of various stakeholders and frequency of this activity.

Figure 1: On-site evaluation (RNTCP Laboratory Network Guidelines)
QI is continuous monitoring of laboratory performance, finding non compliance and taking remedial measures to prevent recurrence of problems
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Quality Improvement mostly relies on effective on-site evaluation visits?
True
False
1
Quality Improvement mostly relies on effective on-site evaluation visits.
Yes
Yes
Measures for Quality Assurance in Microscopy
ContentSputum smear microscopy is an integral part of the National TB Elimination Programme's (NTEP’s) diagnostic services and can majorly impact the success of the programme.
It is essential to have a credible and well-established quality assurance system to assess the performance of the microscopy service.
NTEP has implemented a multi-level quality assurance network system of sputum smear microscopy in the country consisting of:
- Internal Quality Control (IQC)
- External Quality Assessment (EQA)
- Quality Improvement (QI)
- Internal Quality Control is a process of effective and systematic internal monitoring of the working practices.
- It includes technical procedures, checking instruments, quality of new batches of staining solution, smear preparation, grading, equipment infection control measures, waste management, etc.
- For the purpose of IQC, it should be ensured that all staining reagents’ dye content is mentioned on the bottle, concentrated acids are stored carefully in separate containers and stains are filtered before use.
- Further, after each new batch of reagents is made, Quality Control Positive (QCP) and Quality Control Negative (QCN) slides should be prepared by the Senior TB Lab Supervisor (STLS) for quality control.
- QCP slides should be prepared by pooling 3+ grade sputum samples, while QCN slides are to be prepared by pooling negative sputum samples with an adequate number of pus cells (≥10 pus cells/ field).
- One set of QCP-QCN slides should be stained by STLS and another set should be given to the Designated Microscopic Centre (DMC) Lab Technician (LT) along with the reagent, and the results of both should be entered in the batch register/ IQC document.
- All quality control slides should be stored for a maximum period of Four months. For the microscope, the lens should be cleaned with tissue paper after examining each slide and stored inside a microscope box at the end of the day.
- The microscope box should contain silica gel and an electric bulb of 10-15 watts for desiccation to prevent fungal growth on the lens. The silica gel should be dehydrated periodically under direct sunlight. All microscopes should be covered under the Annual Maintenance Contract (AMC) with routine preventive maintenance.
- External Quality Assessment (EQA) is a process to assess the performance of the peripheral laboratories by a more competent laboratory, like the intermediate or the national reference laboratory.
- EQA has 3 components:
1) Onsite evaluation of peripheral laboratories by the supervisors from the reference laboratory under actual working conditions, in order to review the internal quality control mechanisms in place.
2) Panel testing of the slides (unstained and stained) received from the reference laboratory by the peripheral laboratory LT to evaluate the smear microscopy process. This helps to determine whether the LT can adequately perform Acid-fast Bacilli (AFB) smear microscopy.
3) Random blinded re-checking of a small sample of routine slides (both positive and negative) in the peripheral laboratory by the reference laboratory to assess the accuracy of the reading.
- Quality Improvement (QI) is a process by which all components of smear microscopy diagnostic services are continuously monitored and carefully analysed with the aim to identify the problems and implement corrective actions (including retraining) to prevent the recurrence of the problems.
Resources
- Training Module (1-4) for Program Managers and Medical Officers, NTEP, MoHFW, 2020.
- Guidelines for Quality Assurance of Smear Microscopy for Diagnosing Tuberculosis, RNTCP Lab Network, CTD, MoHFW, India, 2005.
Assessment
Question Answer 1 Answer 2 Answer 3 Answer 4 Correct answer Correct explanation Page id Part of Pre-test Part of Post-test Which of these is systematic internal monitoring of working practices that includes technical procedures, staining solution, smear preparation, grading, equipment infection control measures and waste management? Onsite evaluative (OSE) External Quality Assessment (EQA) Internal Quality Control (IQC) Quality Improvement (QI) Internal Quality Control (IQC) Internal Quality Control (IQC) is systematic internal monitoring of working practices that includes technical procedures, staining solution, smear preparation, grading, equipment infection control measures and waste management. Yes Yes Quality control slides should be stored for a maximum period of how many months? 3 6 5 2 1 All quality control slides should be stored for a maximum period of three months.
Yes
Yes
Quality assurance of Stains and Internal Quality Control
-
STLS: External quality assurance
FullscreenGood Laboratory Practices
ContentGood laboratory practices help to maintain biosafety in TB lab settings. However, it is good to keep in mind that:
- NOTHING can totally eliminate the safety risk associated with the TB laboratory.
- Good laboratory practices with an emphasis on biosafety, significantly reduce the risk of laboratory-acquired infection.
- Specialized equipment aids good laboratory practice but does NOT replace it.
Good Laboratory Practices
- Biohazard signs (Figure 1) should be posted at the entrance to laboratories performing work on infectious agents and hazardous chemicals.

Figure 1: Biohazard Sign
- Laboratory access should be limited to essential staff.
- No eating, drinking, or smoking (Figure 2).
- No mouth pipetting (Figure 2).

Figure 2: No eating or mouth pipetting in lab
- No placing pencils or pens in the mouth.
- Keep hands away from eyes and face.
- Always wash hands before leaving the lab.
- Remove gloves before handling phones, instruments or computers.
- Minimize the use of mobile phones.
- Lab coats must be decontaminated and laundered regularly (never take them home for laundering!)
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Panel Testing
ContentPanel testing is a method of External Quality Assurance (EQA) that is used to determine the adequacy of a laboratory technician to perform Acid-fast Bacillus (AFB) smear microscopy. This method evaluates individual performance in staining and reading and not the other laboratory activities.
- Panel testing under National TB Elimination Programme (NTEP) is used for Intermediate Reference Laboratories (IRLs) and District TB Centres (DTCs) during on-site evaluation, because these institutions do not have routine slides for blinded rechecking.
- A panel consists of a batch of stained and/or unstained smears that are sent out by the higher-level reference laboratory to the peripheral laboratories for processing, reading, and reporting of results
- Panel testing is not performed as a routine in DMCs, as they will have regular on-site evaluation and blinded rechecking.
Uses of Panel Testing
- Supplement re-checking programmes
- Provide information on the capabilities of the peripheral laboratories prior to
implementing a re-checking program - Assess status level of performance or to quickly detect problems associated with very poor performance
- Evaluate proficiency of laboratory technicians following training
- Monitor performance of individuals when adequate resources are not available to implement a re-checking program.
Resources
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Panel testing is useful to evaluate the proficiency of laboratory technicians following training.
True
False
1
Panel Testing is useful to evaluate the proficiency of laboratory technicians following training.
Yes
Yes
Onsite Evaluation[OSE]
ContentA field visit is an ideal way to obtain a realistic assessment of the conditions and skills practiced in the laboratory. Under National TB Elimination Programme (NTEP), On-site Evaluation (OSE) of Intermediate Reference Laboratories (IRLs) and District TB Centres (DTCs)/ Designated Microscopy Centres (DMCs) is therefore an essential component of a meaningful Quality Assurance (QA) programme.
OSE should be performed a tleast once a year by personnel from a higher-level laboratory (IRL/ National Reference Laboratory (NRL)) in order to evaluate the overall operational conditions in the microscopy centers.
Laboratory Supervisors must be knowledgeable in all operational and technical elements of AFB smear microscopy and have sufficient expertise to observe technicians performing routine tasks.
Importance of OSE
- Ensures that Standard Operating Procedures (SOPs) are in place and are displayed in all DMCs, internal Quality Check (QC) as per RNTCP is performed, and a functional binocular microscope is available
- Provides an opportunity for immediate problem-solving corrective action and on-site retraining.
Frequency of OSE (Figure)
- At least once a month visit by Senior TB Lab Supervisor (STLS) to the DMC, is required with re-checking of five positive and five negative slides.
- Regular on-site evaluation by District TB Officer (DTO) is important to assure recording and reporting of results; assessing operational conditions, safety, supplies, equipment and total workload.
- At least once a year visit by laboratory supervisors is recommended for IRLs by NRLs and for DTCs by IRLs.
- When poor performance has been identified through OSE, blinded rechecking or panel testing, additional visits by trained laboratory personnel from a higher-level laboratory (the IRL or NRL laboratory) supervisor are mandatory to perform a comprehensive evaluation of all laboratory procedures, implement corrective action, and provide training.
Components of OSE (Figure)
The visit includes:
- Comprehensive assessment of laboratory safety including infection control measures; conditions of equipment, adequacy of supplies as well as the technical components of Acid-fast Bacillus (AFB) smear microscopy. Sufficient time must be allotted for the visit to include observation of all the work associated with AFB smear microscopy, including preparing smears, staining and reading of smears.
- Examining a few stained positive and negative smears to observe the quality of smearing and staining as well as condition of the microscope
- Facilitating quality improvement through on the spot problem solving and suggestions for corrective action, wherever needed.
The NRLs provide training to all IRL personnel responsible for OSE. Additionally, non-laboratory personnel (e.g., DTOs) should acquire working knowledge of routine laboratory operations, including proper NTEP procedures, appropriate supplies, laboratory safety, basic microscope operations, and requirements of panel testing or rechecking programmes.

Figure: Frequency and Components of On-site Evaluation; Source: RNTCP Laboratory Network Guidelines
Checklist for IRL OSE and NRL OSE
On-site quarterly evaluation report of DTCs visited by IRL team to be given to concerned State TB Officer (STO), IRL and NRL using an IRL OSE Checklist while OSE checklist for NRL Laboratory Personnel to IRL is prepared using a NRL OSE Checklist.
Resources
RNTCP Laboratory Network Guidelines.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
On-site Evaluation provides an opportunity for immediate problem solving, corrective action and on-site retraining.
True
False
1
On-site Evaluation provides an opportunity for immediate problem solving, corrective action and on-site retraining.
Yes
Yes
OSE Feedback and action required
ContentDocumentation of any significant problems (technical, operational, others) and feedback for corrective action during On-site Evaluation (OSE) is necessary to formulate plans with the District TB Officer (DTO), Senior TB Laboratory Supervisor (STLS), Medical Officer (MO), Intermediate Reference Laboratory (IRL), National Reference Laboratory (NRL) and Laboratory Technician (LT) to improve the quality of smear microscopy.
Checklists for OSE
- OSE is done through comprehensive checklists.
- The checklist must be used by NRL, IRL and District TB Center (DTC) STLS and a shorter version by DTO/ Medical Officer TB Center (MOTC) during each OSE visit to document observations and corrective action.
- The checklists contain open, non-leading questions and recommended observations along with objective criteria for acceptable practices so that the supervisor can assess how well the technician understands proper procedures.
Feedback
Feedback is the process of communicating results of External Quality Assurance (EQA) to the original laboratory, including suggestions for possible causes of errors and remedies.
TB Unit On-site Evaluation (TU-OSE) Checklist and Feedback
The observations in TU-OSE Checklist, including observation of five positive and negative slides must be documented (Figure 1). Corrective actions must be discussed by the STLS with LT and checklist should be signed by MO of DMC.

1: Write smear and grade
2: Tick appropriate column
3: Tick if good; write ‘U’ if under-decolourized, ‘O’ if over-decolourized
4: Tick if good; write ‘B’ if too big, ‘S’ if too small
5: Tick if good; write ‘K’ if too thick, ‘N’ if too thin
6: Tick appropriate columnFigure 1: Review of five positive and five negative slides by STLS and instructions to fill observations in TU-OSE Checklist; Source: RNTCP Laboratory Network Guidelines
Action Required based on TU-OSE Checklist
The STLS should:
- Enter the summary of ‘action required’ in the Supervision Register before leaving the DMC
- Submit the summary report of DMCs under him to DTO on a monthly basis
- Maintain separate files for checklists including summary reports of each DMC in respective TU and submitted for review by higher level supervisors including on-site evaluation visits by the IRL to the district.
The DTO should:
- Give monthly feedback to the MO in-charge of the respective DMC
- Send summary of results of checklists for each DMC in the district once every quarter for analysis by the respective CMO/ DMO and IRL.
Checklist IRL OSE and NRL OSE and Feedback
- On-site quarterly evaluation report of DTCs visited by IRL team should be given to concerned State TB Officer (STO), IRL and NRL using an IRL OSE Checklist.
- OSE Checklist for NRL Laboratory Personnel to IRL is prepared using a NRL OSE Checklist.
- The IRLs will submit a consolidated quarterly report to the concerned STO and NRL. In turn, the NRL will submit a consolidated quarterly report to the Central TB Division.
- A comprehensive list of all operational elements to be observed will help to ensure consistency in laboratory evaluations and provide immediate feedback to the technicians to facilitate rapid corrective action, as well as serve as documentation of the visit and record of current conditions and actions needed.
- All potential sources of error should be investigated, including quality of stains and staining procedure, quality of microscopes, and administrative procedures that may contribute to recording errors and corrective actions provided (Figure 2).
- All problems contributing to errors must be resolved.
- Possible causes of errors, and suggested evaluation steps are provided in Annexure K.
- Remedial training must be provided for technicians unable to properly identify AFB in smears.

Figure 2: Documentation of corrective actions in IRL-OSE Checklist )RNTCP Laboratory Network Guidelines)
Action Required for IRL/NRL-OSE Checklist
A state level consolidated summary will be prepared by the respective IRL every quarter from the district summaries for submission to STO and NRL. The DTO/ CMO of the district is to submit an action taken report on the team’s recommendations to the STO within a month of the IRL visit.
Resources
RNTCP Laboratory Network Guidelines.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
The IRLs submit a consolidated quarterly report of on-site visits to the concerned State TB Officer and NRL.
True
False
1
The IRLs submit a consolidated quarterly report of on-site visits to the concerned State TB Officer and NRL.
Yes
Yes
Random Blinded ReChecking [RBRC] Process
ContentAnnual slide sample and the monthly sample for Random Blinded Rechecking (RBRC) is determined by the District TB Officer (DTO), assisted by the Intermediate Reference Laboratory (IRL), Statistical Assistant (SA) or Data Entry Operator (DEO) based on Lot Quality Assurance Sampling (LQAS) method.
Under the LQAS method, sample size depends on 3 components:
- Annual Negative Slide Volume (ANSV)
- Slide Positivity Rate (SPR)
- Sensitivity of picking up lower bacili count in microscopy
Process of RBRC (Figure 1)
- The LT at each DMC stores slides for RBRC in slide boxes.
- The laboratory must store slides in a way that allows for easy retrieval of every slide identified for the rechecking sample. Therefore, all slides are to be stored in the provided slide boxes in the same order as they are listed in the laboratory register. Slides are marked as ‘a’ and ‘b’ along with the lab serial number for first spot and early morning specimen.
- The result of the smear examination must not be written on the slide.
- Removal of immersion oil is to be done using tissue paper before storing slides in a slide boxSenior TB Lab Supervisors (STLS) of the district, informing them of the total number of slides to be collected every month from each DMC.
- The STLS then select the required number of slides from the Laboratory Register and the LT records the results as per Annexure B (Figure 2).
Collection of slides for RBRC explained with an example
-
- If the sample size is calculated to be 180 smears per year, 15 slides are to be collected during each monthly visit.
- If the STLS observes that the laboratory processed 82 slides since the last monthly visit, they could collect for example every fifth (82/15 = 5.4 or 5th) slide randomly to obtain the required 15 slides and may begin with any number between 1 to 5, say 3.
- The first random number may be selected by choosing last digit on any available currency note.
- The remaining slides are chosen by adding serially 5 till 15 slides are selected.
- In this example, the 3rd, 8th, 13th, 18th, 23rd, 28th, 33rd, 38th, 43rd, 48th, 53rd, 58th, 63rd, 68th and 73rd slides are selected to obtain 15 slides required for that month.
- Annexure B (Figure 2) is then put into an envelope and sealed. The number of slides packed is written on the top of the envelope.
- Both the slide box and the envelope are marked with the name of the respective DMC, the name of the TB Unit and district, and the month and year.
- The slide box and the sealed envelope are then taken by the STLS for handing over to the DTO.

Figure 1: Process of random blinded rechecking (RBRC) performed by (1) STLS during on-site visit to DMC and (2) at DTC (RNTCP Laboratory Network Guidelines)

Figure 2: Annexure B to document Smear Results Sheet for Blinded Rechecking (RNTCP Laboratory Network Guidelines)
The DTO is responsible for ensuring that the blinding process is strictly followed.
Resources
RNTCP Laboratory Network Guidelines.
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Who is responsible for ensuring that the blinding process is strictly followed?
LT
DTO
STS
STLS
2
The DTO is responsible for ensuring that the blinding process is strictly followed.
Yes
Yes
Random Blinded ReChecking [RBRC] Process at DTC
ContentRandom Blinded Rechecking (RBRC) is the process of re-reading a statistically valid sample of routine slides from a designated microscopy centre (DMC) based on lot quality assurance strategy (LQAS) in a blinded manner to assess the laboratory performance.
RBRC Process
- The district TB officer (DTO) sends information to all senior TB lab supervisors (STLS) of the district on the number of slides to be collected every month from each DMC
- The STLS then selects the required number of slides from the TB Laboratory Register and marks the RBRC selected slides in the laboratory register with a circle
- The laboratory technician (LT) fills out Annexure B (Figure 1) for the selected slides
- LT seals the filled Annexure B in an envelope and marks the slide box with the Serial No. of slides, name of the DMC and TB unit (TU), month and year
- The STLS hands over sealed envelopes and the slide box to the DTO

Figure 1: Annexure B for Blinded Rechecking of DMC Slides (RNTCP Laboratory Network Guidelines)
- The DTO with the help of the statistical assistant (SA) removes all the identifying attributes of the selected slides (including the test results). This process is called Blinding.
- Blinded re-examination of selected slides is done by the STLS of another TB Unit (TU) within the respective district. The STLS (controllers) must have demonstrated proficiency with the Ziehl-Neelsen (ZN) staining and reading method (as seen by panel testing done by Intermediate Reference Laboratory (IRL)).
- Smears may be evaluated for specimen quality (sputum versus saliva), appropriate size and thickness, and quality of staining (as per Annexure C, Figure 2).

Figure 2: Worksheet for Blinded Rechecking of DMC Slides as per Annexure C (RNTCP Laboratory Network Guidelines)
- Problems detected by the controller are noted on the form, as this information may be very useful to supervisors responsible for providing feedback to the peripheral technicians, assessing possible reasons for high false positive or false negative results, and implementing plans for retraining and corrective action.
- The DTO with the help of the statistical assistant compares the results provided by the STLS against the original results provided by the LT. This process is called Unblinding.
- The discrepant slides are sent for umpire (second controller) reading.
- All discrepant slides are re-stained and re-examined by the second controller, as there is likelihood of fading of carbol fuchsin. This rechecking of discordant slides by a second controller also acts to evaluate the performance of the first controllers.
Feedback after RBRC
- Regular and timely feedback to the DMC is essential to improve performance
- Feedback and remedial actions are provided at the end of each sampling period i.e., completion of rechecking of the annual sample
- Also, feedback is given on a monthly basis to the respective DMCs using the form in Annexure D (Figure 3) during the monthly on-site evaluation visit by the STLS responsible for the respective DMC
- Feedback includes the return of slides with discordant results to be re-read by the original LT of the respective DMC
- Potential sources of errors are investigated during the on-site evaluation visit
- Appropriate corrective actions and/or remedial training are provided within one month

Figure 3: Quality Assurance Report on Sputum Microscopy as per Annexure D (RNTCP Laboratory Network Guidelines)
Resources
RNTCP Laboratory Network Guidelines
Assessment
Question
Answer 1
Answer 2
Answer 3
Answer 4
Correct answer
Correct explanation
Page id
Part of Pre-test
Part of Post-test
Who is responsible for the blinded re-examination of selected slides?
STLS of another TU
DTO
STS
LT
1
Blinded re-examination of selected slides is done by an STLS of another TU within the respective district.
Yes
Yes
External Quality Assurance for CBNAAT
ContentExternal Quality Assurance (EQA) ensures that high-quality testing can be carried out efficiently and without interruption. It involves Proficiency Testing (PT) and On-site Evaluation (OSE).
PT is an important component of EQA for Cartridge-based Nucleic Acid Test (CBNAAT) under the National TB Elimination Program (NTEP) and guarantees accurate and reproducible results.
Importance of EQA/ PT for CBNAAT
- Gives assurance to users that the instrument is functioning properly
- Checks to verify that users can correctly interpret and report results
- Verifies that there are no major errors in the process control system and that samples are identified, tested and reported correctly
- Helps to recognize major problems with an instrument or user and take remedial action
Process of EQA for CBNAAT
- EQA of CBNAAT is done using dried tube panels consisting of Mycobacterium tuberculosis (MTB) strains that are RIF resistant/ sensitive/ Non-tuberculous Mycobacteria (NTM)/ negative.
- Coordination of the EQA activity, manufacture and validation of the panels is undertaken by the National TB Institute, Bangalore.
- The process of manufacturing and validating the Dried Tube Specimen (DTS) for EQA panels involves:
- Culturing the mycobacterial strain in the liquid culture system followed by inactivation of the cultures using sample reagents
- Further incubating for 84 days for confirmation of inactivation followed by preparation of DTS panel cultures and their validation.
- Once validated, the DTS panels are dispatched to the CBNAAT sites
Each CBNAAT site receives a set of 5 tubes/ machine.
Ideally, EQA/ PT for CBNAAT is done thrice in a year/ per module/ per site.
Based on the performance of the CBNAAT laboratory, corrective actions are taken by the supervising authority such as Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL). During the on-site visits by the IRL and the NRL, quality indicators for CBNAAT are evaluated and corrective actions are suggested.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Monitoring of CBNAAT Quality Indicators
ContentMonitoring of quality indicators is an essential component of quality assurance for the Cartridge-based Nucleic Acid Amplification Test (CBNAAT).
- Routine monitoring of quality (performance) indicators:
- Critical element of quality assurance for any diagnostic test
- International Organisation of Standardisation (ISO) requirement
- Each testing site should collect and analyze quality indicators monthly.
- Document and investigate any unexplained change in quality indicators, such as:
- Increase in error rates
- Change in Mycobacterium tuberculosis (MTB) positivity rate or Rifampicin (Rif) resistance rate
- Significant change in volume of tests conducted
- Error rates > pre-determined threshold (>5%)
- Test turnaround time
- All unexpected trends should be reviewed by the laboratory manager and linked to corrective actions.
- Standard set of quality indicators should be used for all CBNAAT testing sites.
- System should be in place for centralized reporting of monthly quality indicators.
- Each instrument should be monitored monthly, using the following minimum set of indicators to evaluate proper use:
- Number of tests performed per month
- Number and proportion of MTB detected; Rif-resistance not detected
- Number and proportion of MTB detected; Rif-resistance detected
- Number and proportion of MTB detected, Rif-indeterminate
- Number and proportion of MTB not detected
- Number and proportion of errors
- Number and proportion of invalid results
- Number and proportion of no results
- Median time to result after receipt of the specimen
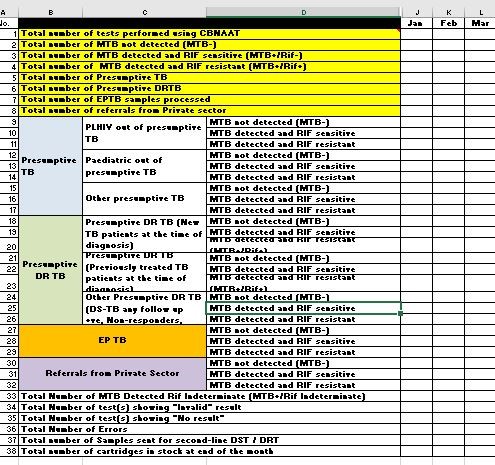
Figure: Monthly Quality Indicators for CBNAAT
Where possible, disaggregated data according to the tested population group (HIV positive, Multi-drug resistance (MDR) risk, extrapulmonary or paediatric TB) is collected.
Quality Indicator Monitoring: Troubleshooting Aid
Identifying the number and type of various errors can help with troubleshooting since certain errors may be associated with processing, instrumental or environmental conditions
The following analyses may be performed:
- The number of errors occurring by user
- The number of errors occurring by instrument module
- The number of tests lost due to power outages or surges
- The number, duration and causes of routine interruptions in the CBNAAT testing service
Video fileVideo : Monitoring of CBNAAT Quality Indicators
Resources
- Routine monitoring of quality (performance) indicators:
External Quality Assurance of TrueNAT
ContentExternal Quality Assurance (EQA) ensures quality of Truenat laboratory is maintained by comparing their results through retesting and panel testing by a higher level laboratory (Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL)).
EQA for Truenat laboratory is done by:
- Onsite supervision: Visits conducted at regular intervals by the IRL/ NRL/ Central TB Division (CTD)
- Proficiency Testing or Panel Testing (PT): Inter-laboratory comparisons conducted by IRL/ NRL to assess the performance of the laboratory supervised by them; re-checking of the standard panel provided by the IRL/ NRL.
- Random Blinded Re-checking (RBRC): Re-examination of randomly selected samples by the IRL/ NRL.
Plan for Truenat EQA
- Truenat EQA visits need to be conducted annually.
- Panel is provided by the National Tuberculosis Institute (NTI), Bangalore, to the participating sites.
- Participating site to conduct panel testing and report results to NTI as per timelines.
- Feedback/ corrective action provided by NTI to participating sites.
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Monitoring Quality Indicators of Truenat
ContentMonitoring of key quality indicators or performance indicators of Truenat is important to assess the functioning of the instrument, calibration/ service requirements of the instrument, competency of the technician performing the assays and inventory management. Overall quality indicators should assess:
- Total tests done
- Type of samples tested
- Test failure
- Availability of consumables
- Turnaround time
- Specimen rejected
- External Quality Assurance (EQA) results
- Instrument downtime
Quality performance indicators should be reviewed weekly by personnel in charge of Truenat laboratory, and monthly by the Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL)/ Central TB Division (CTD).
Under the National Tb Elimination Programme (NTEP) the following quality indicators are to be reported monthly:
- Total number of tests performed using Truenat (including EP-TB and private sector)
- Total presumptive TB tested
- Total number of presumptive DR TB tested
- Total number of Rif indeterminate, invalid, error results
- Total number of re-tests done and results
- Total number of samples sent for First Line-Line Probe Assay (FL-LPA) and Second Line-Line Probe Assay (SL-LPA)
- Total number of chips (MTB, MTB Rif) in stock
Resources
- Practical Guide to Implementation of Truenat™ Tests for the Detection of TB and Rifampicin Resistance.
- MolBio Diagnostics Pvt. Ltd.
Kindly provide your valuable feedback on the page to the link provided HERE
Fullscreen
