External Quality Assurance for CBNAAT
ContentExternal Quality Assurance (EQA) ensures that high-quality testing can be carried out efficiently and without interruption. It involves Proficiency Testing (PT) and On-site Evaluation (OSE).
PT is an important component of EQA for Cartridge-based Nucleic Acid Test (CBNAAT) under the National TB Elimination Program (NTEP) and guarantees accurate and reproducible results.
Importance of EQA/ PT for CBNAAT
- Gives assurance to users that the instrument is functioning properly
- Checks to verify that users can correctly interpret and report results
- Verifies that there are no major errors in the process control system and that samples are identified, tested and reported correctly
- Helps to recognize major problems with an instrument or user and take remedial action
Process of EQA for CBNAAT
- EQA of CBNAAT is done using dried tube panels consisting of Mycobacterium tuberculosis (MTB) strains that are RIF resistant/ sensitive/ Non-tuberculous Mycobacteria (NTM)/ negative.
- Coordination of the EQA activity, manufacture and validation of the panels is undertaken by the National TB Institute, Bangalore.
- The process of manufacturing and validating the Dried Tube Specimen (DTS) for EQA panels involves:
- Culturing the mycobacterial strain in the liquid culture system followed by inactivation of the cultures using sample reagents
- Further incubating for 84 days for confirmation of inactivation followed by preparation of DTS panel cultures and their validation.
- Once validated, the DTS panels are dispatched to the CBNAAT sites
Each CBNAAT site receives a set of 5 tubes/ machine.
Ideally, EQA/ PT for CBNAAT is done thrice in a year/ per module/ per site.
Based on the performance of the CBNAAT laboratory, corrective actions are taken by the supervising authority such as Intermediate Reference Laboratory (IRL)/ National Reference Laboratory (NRL). During the on-site visits by the IRL and the NRL, quality indicators for CBNAAT are evaluated and corrective actions are suggested.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Monitoring of CBNAAT Quality Indicators
ContentMonitoring of quality indicators is an essential component of quality assurance for the Cartridge-based Nucleic Acid Amplification Test (CBNAAT).
- Routine monitoring of quality (performance) indicators:
- Critical element of quality assurance for any diagnostic test
- International Organisation of Standardisation (ISO) requirement
- Each testing site should collect and analyze quality indicators monthly.
- Document and investigate any unexplained change in quality indicators, such as:
- Increase in error rates
- Change in Mycobacterium tuberculosis (MTB) positivity rate or Rifampicin (Rif) resistance rate
- Significant change in volume of tests conducted
- Error rates > pre-determined threshold (>5%)
- Test turnaround time
- All unexpected trends should be reviewed by the laboratory manager and linked to corrective actions.
- Standard set of quality indicators should be used for all CBNAAT testing sites.
- System should be in place for centralized reporting of monthly quality indicators.
- Each instrument should be monitored monthly, using the following minimum set of indicators to evaluate proper use:
- Number of tests performed per month
- Number and proportion of MTB detected; Rif-resistance not detected
- Number and proportion of MTB detected; Rif-resistance detected
- Number and proportion of MTB detected, Rif-indeterminate
- Number and proportion of MTB not detected
- Number and proportion of errors
- Number and proportion of invalid results
- Number and proportion of no results
- Median time to result after receipt of the specimen
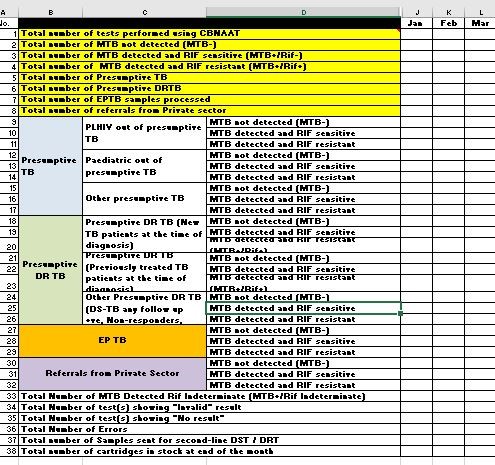
Figure: Monthly Quality Indicators for CBNAAT
Where possible, disaggregated data according to the tested population group (HIV positive, Multi-drug resistance (MDR) risk, extrapulmonary or paediatric TB) is collected.
Quality Indicator Monitoring: Troubleshooting Aid
Identifying the number and type of various errors can help with troubleshooting since certain errors may be associated with processing, instrumental or environmental conditions
The following analyses may be performed:
- The number of errors occurring by user
- The number of errors occurring by instrument module
- The number of tests lost due to power outages or surges
- The number, duration and causes of routine interruptions in the CBNAAT testing service
Video fileVideo : Monitoring of CBNAAT Quality Indicators
Resources
- Routine monitoring of quality (performance) indicators:
Fullscreen