Specimen Processing for CBNAAT
ContentThe CBNAAT system integrates and automates sample processing with amplification and detection of the target sequences
For sample processing, sample reagent is provided in CBNAAT kit, 8ml volume pack per each cartridge
- The sample reagent solution is clear but may range from colourless to golden yellow
Processing of clinical specimens should be performed as per laboratory biosafety standards
- Treat all sputum specimens as potentially infectious
- Wear protective gloves and laboratory coats when handling specimens and reagents
Sample processing from direct sputum and decontaminated sputum sediments is described in the Video given below -

Video 1: Sample processing from direct sputum; Source: Challenge TB: Sample Processing Sputum
Video fileVideo : Specimen Processing for CBNAAT
Resources
CBNAAT Sample Processing for Other Body Fluids
ContentThe following procedures are recommended when processing various body fluids with the Cartridge-based Nucleic Acid Amplification Test (CBNAAT):
Bronchoalveolar Lavage (BAL):
Processing of BAL for CBNAAT assay is given here. However, it is important that each laboratory optimizes this protocol to minimize the error rate.
If the BAL volume is sufficient (approx. 5 ml), centrifuge and dissolve sediment into 1 ml sterile phosphate buffer/ saline, then add sample reagent in a 1:2 ratio.
If the BAL volume is less (less than 5 ml), take 1 ml and add 2 ml of sample reagent.
- If the BAL is mucoid or has more than 0.5% blood contamination, decontaminate using N-acetyl-l-cysteine–sodium hydroxide (NALC-NaOH) treatment.
- Decontamination of BAL should also be carried out if the error rate is more than 2%.
Pericardial/ Ascitic/ Synovial Fluid:
If the sample volume exceeds 5 ml, centrifuge and dissolve sediment into sterile phosphate buffer/saline to make volume 1 ml, then add sample reagent in a 1:2 ratio.
If the sample volume is less than 5 ml, take 1 ml and add 2 ml of sample reagent.
- Pleural fluid is a suboptimal sample, and pleural biopsy is preferred.
- A positive CBNAAT result in pleural/body fluid can be treated as TB, a negative result should be followed by other tests.
- Decontaminate/concentrate using standard NALC-NaOH treatment if bloody (more than 0.5% blood), thick and/or clots are present.
Pus/ Abscess/ Aspirates/ Semen:
- Liquid/ slightly viscous specimen: Use sample-to-sample reagent 1:2 ratio, mix well and follow routine CBNAAT protocol.
- If very thick, viscous or bloody specimens: Add 2 ml sample reagent to 0.2 - 0.3 ml pus, mix well to the vortex, and increase the incubation time, if required.
- Decontaminate, if required (>0.5 % blood).
*Each laboratory must optimize these protocols to minimize the error rate.
Resources
CBNAAT Cartridge Loading
Content- Use the sterile transfer pipette provided in the CBNAAT kit to draw liquefied sample into the transfer pipette
- The minimum amount to be loaded into the cartridge is 2 ml
- Do not process the sample if there is insufficient volume
- Open the cartridge lid
- Transfer the sample into the open port (Figure 1) of the CBNAAT cartridge and dispense slowly to minimize the risk of aerosol formation
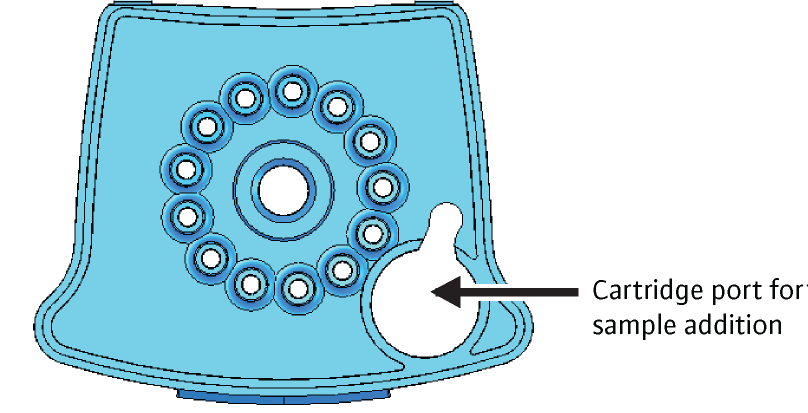
Figure 1: CBNAAT cartridge (top view) to show the open port
(SOP for GeneXpert MTB/RIF)- Discard the transfer pipette
- Snap the lid shut to close firmly
- Turn on the CBNAAT instrument
- Open the instrument module door, which displays the blinking green light
- Load the cartridge and close the door of the module firmly
- The test should be started within 30 minutes of adding liquefied sample to the cartridge
- The remaining liquefied sample may be kept for up to 12 hours at 2-8°C (for repeat testing)
Video fileVideo 1: CBNAAT loading
Video fileVideo 2: Sample Loading (Demonstration)
Resources
- Use the sterile transfer pipette provided in the CBNAAT kit to draw liquefied sample into the transfer pipette
Fullscreen